Structural Biochemistry/Proteins/Amino Acids
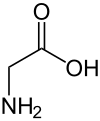
Proteins are polymers of multiple monomer units called amino acid, which have many different functional groups. More than 500 amino acids exist in nature, but the proteins in all species, from bacteria to humans, consist mainly of only 20 called the essential amino acids. The 20 major amino acids, along with hundreds of other minor amino acids, sustain our lives. Proteins can have interactions with other proteins and biomolecules to form more complex structures and have either rigid or flexible structures for different functions. Iodinated and brominated tyrosine are also amino acids found in species, but are not included in the 20 major amino acids because of their rarity: iodinated tyrosin is only found in thyroid hormones, and brominated tyrosine is only found in coral. The 20 main amino acids that are found in most but not all proteins are listed below:
Amino Acids
[edit | edit source]Amino acids are molecules which contain both a carboxylic acid and an amine group. In amino acid, the carboxyl group is more acidic than the carboxylic acid. 2-amino acids, also known as alpha-amino acids, are a specific type of amino acid that makes up proteins. These amino acids have many interesting properties which will be discussed in the next sections.
Amino acids play central roles both as building blocks of proteins and as intermediates in metabolism. Proteins are linear polymers formed by linking the a-carboxyl group of one amino acid to the a-amino group of another amino acid. This type of linkage is called a peptide bond or an amide bond. The formation of a dipeptide from two amino acids is accompanied by the loss of a water molecule. The equilibrium of this reaction lies on the side of hydrolysis rather than synthesis under most conditions. Hence, the biosynthesis of peptide bonds requires an input of free energy. Nonetheless, peptide bonds are quite stable kinetically because the rate of hydrolysis is extremely slow; the lifetime of a peptide bond in aqueous solution in the absence of a catalyst approaches 1000 years. Thus, the 20 amino acids that are found within proteins convey a vast array of chemical versatility. The precise amino acid content, and the sequence of those amino acids, of a specific protein, is determined by the sequence of the bases in the gene that encodes that protein. The chemical properties of the amino acids of proteins determine the biological activity of the protein. Proteins not only catalyze all (or most) of the reactions in living cells, they control virtually all cellular process. In addition, proteins contain within their amino acid sequences the necessary information to determine how that protein will fold into a three dimensional structure, and the stability of the resulting structure. The field of protein folding and stability has been a critically important area of research for years, and remains today one of the great unsolved mysteries. It is, however, being actively investigated, and progress is being made every day.
Amino Acid Subdivisions
[edit | edit source]There are twenty major amino acids which make up proteins. Each of them contains a unique functional group which gives rise to different properties. These properties include size, shape, charge, capacity for hydrogen bonding, hydrophillicity/hydrophobicity(hydrophobic interactions), and chemical reactivity. Amino acids can be broadly hydrophobic and hydrophilic, depending on the chemical properties of the R group side chain. In an aqueous environment, the hydrophobic amino acids are unable to participate in hydrogen bonding. They associate with one another and reside mostly inside the protein. On the other hand, hydrophilic amino acids tend to interact in the aqueous environment due to polarity. These amino acids are normally found on the exterior surface.
Zwitterion
[edit | edit source]An amino acid is in a zwitterionic state when the carboxylic acid group is deprotonated and the amino group is protonated, simultaneously. Zwitterions are dipole ions—meaning that these molecules have two charges, both a positive and a negative charge. The pH of the water solution is a factor determining the state of protonation. Such a state leaves the carboxylic end negatively charged (-COO-) and the adjacent amino end positively charged (-NH3+). The carboxyl group (-COO-) is deprotonated first because the pKa is about 2 and the pKa of the amine group (-NH3+) is about 9. The net charge for the protein in zwitterionic form is zero. [1] Molecules which behave in this fashion are called amphoteric. In solid state, the amine functionality deprotonates the carboxylic acid group, giving rise to the zwitterionic, dipolar entity. The charged state of an amino acid in aqueous solution depends largely on the pH. The major form of all amino acids at a pH of 2 to 9 is the zwitterionic form. In strong acid (pH < 2), the predominant form is the fully protonated cationic ammonium with the corresponding protonated form of the carboxylic acid. This species has a net charge of +1. In strongly basic solutions (pH > 9), the predominant form is the fully deprotonated aminocarboxylate anion. This species would have a net charge of -1. These forms interconvert by acid-base equilibria. This leaves a wide pH range wherein the zwitterion would play a large role as a contributing species. The pH at which the extent of protonation equals that of deprotonation is called the isoelectric pH or the isoelectric point (pI). At this pH, the amount of positive charge balances that of negative charge and the concentration of the charge-neutralized zwitterionic form is at its highest. When the side chain of the acid bears an additional acidic or basic function, the pH is either decreased or increased, respectively. Note that at most relevant physiological pH ranges, the zwitterion would be, by far, the species of the most abundance.
Histidine contains an imidazole ring with 2 nitrogen atoms: one is basic and the other is not. The basic nitrogen is involved in the delocalization which is important during enzyme catalysis.
Here is an example of L-amino acids forming zwitterion at neutral pH:

Optical Activity
[edit | edit source]All proteins or polypeptides are a series of linked amino acids. A typical α amino acid consists of a central carbon (which is the alpha carbon in this case) that is attached to an amino group (-NH2), a carboxylic acid (-COOH), a hydrogen atom, and a distinctive R group. The R group, usually referred to as a side chain, determines the properties of each amino acid. Scientists classify amino acids into different categories based on the nature of the side chain. A tetrahedral carbon atom with four distinct groups is called chiral. The ability of a molecule to rotate plane polarized light to the left, L (levorotary) or right, D (dextrorotary) gives it its optical and stereochemical fingerprint. All amino acids within polypeptides are configured in the L form. The L form corresponds to the absolute configuration of S, which is a system used to designate stereochemistry in the field of organic chemistry. Although D-amino acids (designated as R stereoisomers in the field of organic chemistry) exist naturally, they are not found in proteins. Thus far, scientists have not been able to come up with a hypothesis on the preference for the L amino acids in living organisms. It is clear, however, that all of the physiological mechanics downstream of the amino acids are geared towards recognizing and interacting with the specific L conformation. Note: Since the central carbon has four distinct groups attached, all of amino acids are chiral except for glycine, which is achiral. This is due to the fact that the central carbon atom in glycine contains only 3 unique substituents instead of 4 (R sidechain = H).
Modified Amino Acids
[edit | edit source]Within proteins, it is possible to find amino acids which do not correspond to the 20 standard types. Most of these come about by chemical modification of an already incorporated amino acid. For example, a hydroxylated form of proline exists within collagen protein. Also, a selenium analog of cysteine is known to occur in glutathione peroxidase enzymes. Pyrrolysines have also been isolated and characterized. These exceptions to the rule are dictated by and encoded within DNA and RNA and there are many more examples.
The Peptide Bond
[edit | edit source]Any discussion of amino acids is not complete without mentioning how each amino acid bonds to another. All amino acids bond to one another through a condensation reaction involving the amine group of one amino acid and the carboxylic acid group of another. The enzymatically-catalyzed reaction forms an amide entity: [R1-NH2 + R2-COOH ==> R1-NH-C(=O)-R2 + H2O]. The amide bond has special properties in that it has a resonance form which gives the bond a planar, rigid, double bond character: [R1-N-C(=O)-R2 <==> R1-N+=C(-O-)-R2]. Amino acids can link to each other in small units of only 2 or 3 amino acids called dipeptides and tripeptides, but can also connect in very large chains consisting of hundreds or even thousands of amino acids. Each complete peptide series has an N terminus (amino) and a C terminus (carboxylate). The overall, 4 atom angles involved in the peptide bond system are important to those who study proteins. In particular, the R-[N-C-C(=O)-N]-R group is called a phi torsion angle and the adjacent angle, the psi, φ, torsion angle, involves the R-[C-N-C-C(=O)]-R group. These angles are important to consider and the natural distribution of know peptide angles are summarized on the Ramachandran plot. Peptide bond is formed by condensation reaction and broken by hydrolysis (addition of water).

Tetraptide is a peptide that has four amino acids that are joined by peptide bonds.

Amino Acid Classification
- Non-polar Amino Acids
- Aliphatic : glycine, alanine, valine, isoleucine, leucine
- Aromatic : phenylalanine, tryptophan.
- Cyclic : Proline
- Polar Amino Acids
- Sulfur-Containing : cysteine, methionine
- Hydroxyl-Containing : serine, threonine
- Aromatic : tyrosine
- Acidic Amide : asparagine, glutamine
- Charged Amino Acids (at physiological pH)
- Acidic : aspartic acid, glutamic acid
- Basic : histidine, lysine, arginine
List of the 20 Amino Acids
[edit | edit source]Network Approach
[edit | edit source]- The network approach helps determine the role of a specific amino acid at a known position in the protein structure. Networks simplify complex system behaviors by splitting the system into a series of links. Links represent the neighboring positions of amino acids in protein molecules. Because proteins are linked in this way and protein structure networks are connected to each other by only a few other amino acid elements, we can determine folding probability. Proteins with denser protein structure networks fold more easily and the folding probability increases as the protein structure becomes more compact.
- The network approach can also be applied to the prediction of active centres in proteins. Active centres are protein segments that play key parts in the catalytic reaction of the enzyme function shown by their respective proteins. Scientists have used long-range network topology to create a network skeleton from which they can study only side chains which are essential in the flow of information for the whole protein. Network analysis has showed that active centres occupy a central position in protein structure networks, usually have many neighbors, give unique linkages in their neighborhood, integrate communication for the entire network, do not take part in wasteful actions of ordinary residues, and collect and coordinate most of the energy in the network.
Alanine - Ala/ A
[edit | edit source]
Structure Alanine, also known as 2-Aminopropanoic Acid, (abbreviated as Ala or A) is an α-amino acid with the chemical formula HOOCCH(NH2)CH3. It has a molar mass of 89.09 g/mol and a density of 1.424 g/cm3. The α-carbon atom of alanine is bound with a methyl group (-CH3), making it one of the simplest α-amino acids with respect to molecular structure and also resulting in alanine being classified as an aliphatic and amino acid. The methyl group of alanine is non-reactive and is thus almost never directly involved in protein function. Alanine is a nonpolar hydrophobic molecule. It is ambivalent, meaning it can be inside or outside of the protein molecule. The α-carbon of alanine is optically active; in proteins, only the L-isomer is found.
Features Alanine is a non-essential amino acid which means that it can be manufactured by the human body and does not need to be obtained directly through the diet. Alanine is found in a wide variety of foods, but is particularly concentrated in meats. It is a non-essential amino acid that occurs in high levels in its free state in plasma.
Functions Alanine is the primary amino acids for sugar and acid metabolism. It boosts up the immune system by producing antibodies, and provide energy for muscles tissues, brain, and the central nervous system. It is used in pharmaceutical preparations for injection or infusion. It is also used in dietary supplement and flavor compounds in maillard reaction products. In addition, it is a stimulant of glucagon secretion.
Chemical Synthesis Alanine can be manufactured in the body from pyruvate and branched chain amino acids such as valine, leucine, and isoleucine. Alanine is most commonly produced by reductive amination of pyruvate. Because transamination reactions are readily reversible and pyruvate pervasive, alanine can be easily formed and thus has close links to metabolic pathways such as glycolysis, gluconeogenesis, and the citric acid cycle. It also arises together with lactate and generates glucose from protein via the alanine cycle. Racemic alanine can be prepared via the condensation of acetaldehyde with ammonium chloride in the presence of potassium cyanide by the Strecker reaction.
Analysis Alanine can be identified via UV spectrometry, infrared spectroscopy (IR), nuclear magnetic spectroscopy, (NMR), and mass spectroscopy.
Arginine - Arg/ R
[edit | edit source]Structure Arginine, 2-Amino-3-carbamoylpropanoic acid, contained of a three-carbon aliphatic straight chain with the end of which is capped by a guanidinium group. Its molar mass is 132.12g/mol. With a pKa of 12.48, the guanidinium group is positively charged in neutral, acidic and even most basic environments. Therefore, arginine has basic chemical properties. Because of the conjugation between the double bond and the nitrogen lone pairs, the positive charge is delocalized and enables the formation of multiple H-bonds.
Features Arginine is an essential amino acid that plays important role in nitrogen metabolism. It is a chemical precursor to nitric oxide (a blood vessel-widening agent called a vasodilator. Nitric oxide is a powerful neurotransmitter that helps blood vessels relax and also improves circulation. Food that are rich in arginine include red meat, fish, poultry, wheat germ, grains, nuts and seeds, and dairy products.
Functions Arginine assists in wound healing and help in burn treatment. It is necessary in normal immune system activity by enhancing the production of T-cells. Studied show that arginine may help treat medical conditions that improve with increased vasolidation. Some conditions that are treated with arginine are chest pain, atherosclerosis (clogged arteries), heart disease or failure, erectile dysfunction, intermittent claudication/peripheral vascular disease, and vascular headaches (headache-inducing blood vessel swelling). Arginine also helps with bodybuilding, enhancing sperm production, and preventing tissue wasting in people with critical illnesses. Arginine hydrochloride has high chloride content and has been used to treat metabolic alkalosis.
Biosynthesis Arginine is synthesized from citrulline with the presence of cytosolic enzymes argininosuccinate synthetase and argininosuccinatelyase. This is energetically costly reaction. Therefore, the synthesis of each molecule of argininosuccinate will be coupling with hydrolysis of adenosine triphosphate (ATP) to adenosine monophosphate (AMP).
Synthesis of arginine in human body occurs principally via the intestinal–renal axis, wherein epithelial cells of the small intestine, which produce citrulline primarily from glutamine and glutamate, then join with the proximal tubule cells of the kidney, which extract citrulline from the circulation and convert it to arginine, which comes back to the circulation.
Arginine and Nitrogen Storage In order for a cell to grow, it needs nitrogen which can come from ammonia, nitrates, dinitrogen or amino acids. The PII protein is an ancient signaling protein that senses and integrates nitrogen and carbon abundance by binding 2 OG and ATP/ADP. The N-acetyl-L-Glutamate kinase (NAGK) stores nitrogen as arginine which it incorporates into arginine rich copolymers. Since arginine is nitrogen-rich, it is an ideal for nitrogen storage. The osmotic impact of arginine minimizes when arginine is incorporated into proteins. The PII protein binds to NAGK when nitrogen is abundant only in oxygenic phototrophs. But when nitrogen is scarce, 2-oxoglutarate binds to the PII protein with ATP leading to the dissociation of the PII-NAGK complex.
Arginine-insensitive NAGK is a homodimer containing a backbone of 16-stranded Beta sheets in both subunits. However, arginine-sensitive are hexameric and recent studies have shown that these enzymes are ring-like hexameric trimers of dimers. The ring is formed by the link between three E. Coli NAGK-like dimers and the N-terminal alpha-helix. In arginine-sensitive NAGK, the arginine is connected by interlaced N-helices. The helices are needed for making NAGK an arginine-operated switch showing a sigmoidal of the arginine inhibition kinetics. The PII protein is homotrimers having a βαββαβ subunit topology with the alpha helices looking outward and the beta sheet inward. The T-loop is large and flexible loop that contain the phosphorylation and uridylylation sites in cyanobacteria and proteobacteria. When the protein PII is absent, S. elongates NAGK is inactive having low Vmax and high Km for NAG and requiring a low concentration of argigine for inhibition. However, the enzyme A. thaliana NAGK is highly active having a Km four times lower and a Vmax three times greater for NAG than S. elongates NAGK. When PII binds the S. Elongates NAGK, the Vmax for NAG increases up to four times the original amount and decreases up to ten times the original amount for Km. Km is not affected when it binds to A. thaliana NAGK, but the Vmax for NAG increases by five times the original amount. The original amount is the amount with the protein PII absent. The S. elongates PII-NAGK complex has one NAGK hexamer that is sandwiched between two PII trimers. Since the PII proteins are not packed tightly on NAGK, PII only interacts with NAGK on the T-loops and B-loops. The A. thaliana PII-NAGK complex has MgATP bounded to the PII protein with all the NAGK active centers containing bound NAG and ADP.
Asparagine - Asn/ N
[edit | edit source]
Structure Asparagine is polar and uncharged derivative of acidic amino acid aspartic acid or aspartate; as a side chain, it has a carboxamide group, which is neutral at physiological pH and can be changed to carboxylic acid by hydrolysis to form aspartate amino acid. The carboxamide group of the amino acid can form hydrogen bonds.
Features Asparagine is found in abundance in asparagus, and is thus named so. Asparagine is not an essential amino acid, meaning that it is not necessary for humans to ingest it to receive necessary amounts. Asparagine has a high propensity to hydrogen bond, since the amide group can accept two and donate two hydrogen bonds. It is found on the surface as well as buried within proteins. It is a common site for attachment of carbohydrates in glycoproteins. Food sources that contain asparagine is dairy, beef, poultry, and eggs.
Functions Asparagine, along with glutamate, is an important neurotransmitter. Since Aspartic acid and Asparigine have high concentration in the hippocampus and hypothalamus of the brain, which is important in short-term memory and emotions, the two amino acids serves essential role between the brain and the rest of the body. Asparagine is required by the nervous system to maintain equilibrium and is also required for amino acid transformation from one form to the other which is achieved in the liver.
Synthesis Synthesis of asparagine requires oxaloacetate, C4H4O5. The double bonded oxygen attached to carbon-2 is replaced by ammonium group from glutamate via a process called transaminase. The newly formed compound, or aspartate, is converted to asparagine by replacing a negatively charged oxygen end with an ammonium group. The asparagine synthesis converts glutamine to glutamate, and ATP into AMP and pyrophosphate.
Analysis Asparagine can be identified by following methods: UV spectrometry, infrared spectroscopy (IR), nuclear magnetic spectroscopy, (NMR), and mass spectroscopy.
Aspartic acid - Asp/ D
[edit | edit source]
Structure Aspartic acid (C4H7NO4) is also named as a 2-aminobutanedioic acid. Its molecular weight is 133.1 g/mol.
Also known as aspartate, Aspartic acid is an acidic and polar amino acid that has carboxylic acid group, which loses a proton to be carboxylate group for physiological pH and has a negative charge; the carboxylic acid group of the amino acid has a pKa value of 4.1, which is a little basic than the terminal α-carboxyl group. Its pI is 5.41. Proteins are critical to maintain the pH balance in the body. It is the charged amino acids that are involved in the buffering properties of proteins. Aspartic acid is similar to alanine but with one of the β hydrogens replaced with a carboxylic acid group. This carboxylic acid group is what makes aspartate an acidic amino acid. Aspartate has an α-keto homolog, called oxaloacetate. Aspartate and oxaloacetate are interconvertable by a simple transamination reaction. Oxaloacetate is one of the intermediates of the Krebs cycle. The Krebs cycle is the sequence of reactions by which most living cells generate energy during the process of aerobic respiration.
Features Aspartic acid is a non-essential amino acid can be obtained from central metabolic systems.
Functions Aspartic acids are involved in transamination in which oxaloacetate and aspartate is interconvertible. It is also involved in immune system activity by promoting immunoglobulin production and antibody production. Moreover, aspartic acid protects the liver and helps in detoxification of ammonia.
Aspartate, the conjugate base of aspartic acid, also functions as a neurotransmitter. Along with few other amino acids, its primary role is to activate NMDA receptors in brain and; however, its effect is not significant as glutamate's.
Other than its role as an excitatory neurotransmitter, aspartate is proteinogenic amino acids that are used in coding of DNA.
Aspartate plays important roles as acids in enzyme active centers, as well as in maintaining the solubility and ionic character of proteins.
Synthesis Aspartic acid is synthesized from oxaloacetate via transamination. Aspartic acid can be used as an initial reactant in synthesis of other essential amino acids as well: methionine, threonine, isoleucine, and lysine. Aspartic acid needs to be reduced to its semialdehyde form of HOOCCH(NH2)CH2CHO. Asparagine can be also obtained from aspartic acid via transamidation: aspartic acid + glutamine -> asparagine + glutamic acid
Cysteine - Cys/ C
[edit | edit source]
Structure Cysteine, C3H7NO2S with molecular mass of 121.16 g/mol, is an amino acid that is made of the sulfhydryl or thiol group (-SH), which is more nucleophilic than a hydroxyl group. Its alternate name is 2-amino-3-mercaptopropanoic acid. Two cysteine residues can be oxidized to form stable disulfide bonds. Disulfide bonds can help to give a protein secondary and tertiary structure, e.g. protein folding. The unit of two bonded cysteines is known as cystine. Cysteine is considered to be a hydrophilic amino acid based on the fact that the thiol group interacts well with water. It is also a non-essential amino acid, and can be biosynthesized in human bodies.
Functions Nucleophilic thiol groups in cysteine can be easily oxidized; thus, cystein is highly reactive with its neutral pKa and has various functions in biology.
Cysteine is capable of inactivation of insulin in bloodstream. Excessive amount of cysteine reduces one of three disulfide bonds in insulin structure. As a result, insulin loses its functionality. Cysteine's capability of inactivation of insulin can be utilized in medicine and pharmaceutic when a patient experiences hypoglycemia attack due to high level of insulin.
Cysteine promotes iron production in iron deficiency anemia. It also assists in lung diseases by increasing production of red blood cells. Cysteine is a key, active site residue in many important proteins. Cysteine is the key residue in glutathione reductases which has protective effects against UV light, radiation, and free radicals. Additionally, glyceraldehyde-3-phosphate dehydrogenase, a key enzyme in glycolysis, uses cysteine in to achieve its most critical functions.
When cysteine is taken as a supplement, it is in the form of N-acetyl-L-cysteine (NAC). The body makes this into cysteine and then into glutathione, a powerful antioxidant. Antioxidants fight free radicals which are harmful compounds in the body that cause damage to the cell membranes and DNA. Researchers believe the free radicals play a role in aging as well as the development of a number of health problems, including heart disease and cancer. NAC can also help prevent side effected caused by drug reactions and toxic chemicals. It also helps break down mucus in the body. NAC also benefits in treating some respiratory conditions, such as bronchitis and COPD. COPD is the acronym for chronic obstructive pulmonary disease. Doctors often give NAC to people who have taken an overdose of acetaminophen (Tylenol). The NAC helps to prevent or reduce liver and kidney damage. NAC also helps reduce angina. Angina is chest pain or discomfort when the heart muscle does not get enough blood. Taking NAC will open the blood vessels and improve blood flow to the heart. Studies have also shown that NAC may help relieve symptoms of chronic bronchitis, leading to fewer flare ups. Not all studied gave these results. Some studies did not find any reduction in flare ups. Other studies showed that people with COPD who took NAC lowered the number of flare ups about 40% when used with other therapies. Another study shows that people who took NAC two times a day had fewer flu symptoms than those who took placebo. Some research has shown that intravenous NAC may boost levels of glutathione and help prevent and/or treat lung damage cause by ARDS, acute respiratory distress syndrome. Other results did not coincide with these results. For example, giving NAC to people with ARDS helped reduce the severity of their conditions while not reducing the number of overall deaths compared to placebo. Cysteine is important in keratin structure, which is important in hair and nails formation on skin. Wool obtained from sheeps, and other animals is cysteine containing.
Biosynthesis The precursors of synthesis of cysteine are serine and methionine. Serine has a hydroxide group and methione has a sulfer as their substituents. Methione is initially converted into a homocysteine. With serine, homocysteine becomes cystathione (C7H14N2O4S) with water molecule leaving. Finally, addition of water and departure of ammonia from cystathione result in cysteine and alpha-ketobutyrate as a side-product.
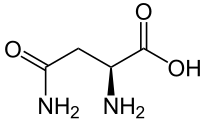
Glutamine - Gln/ Q
[edit | edit source]
Structure
Glutamine, or 2-amino-4-carbamoylbutanoic acid, has a molecular formula of C5H10N2O3 and a molecular mass of 146.16 g/mol. It is a polar and uncharged derivative of acidic amino acid glutamic acid or glutamate; it has a carboxamide group, which is neutral at physiological pH and can be changed to carboxylic acid by hydrolysis to form glutamate amino acid. The carboxamide group of the amino acid can form hydrogen bonds.

Synthesis As previously stated, glutamine is a nonessential amino acid. In the body, glutamine is synthesized from glutamate via the enzyme glutamine synthestase (GS) and through the addition of ATP and ammonia. (See Figure).
Glutamate + ATP + NH3 → Glutamine + ADP + phosphate + H20
The incorporation of ammonia into glutamate is an amidation type reaction and the hydrolysis of ATP to ADP drives the reaction forward. ATP is directly involved in the reaction because it phosphorylates the carboxyl group on the side chain of glutamate and forms an acyl-phosphate intermediate (See Figure: Glutamine Final). The acyl-phosphate intermediate reacts with free ammonia and forms glutamine. Glutamine synthetase (GS) plays a major role because a high-affinity binding-site for ammonia is formed in GS after the formation of the intermediate to prevent hydrolysis of the intermediate. Hydrolysis of the intermediate would not yield glutamine and thus waste a valuable molecule of ATP.
Functions Glutamine is a non-essential amino acid, which means that it will naturally occur in the human body and does not need to be gathered from exogenous sources. It is one of the most abundant amino acid manufactures in the body. Glutamine circulates in the blood and is able to cross the blood-brain barrier directly.
Glutamine has various functions in biochemistry. Its primary role is protein synthesis, but it also helps to maintain neutral pH in the liver by balancing the acid and base levels.
Like glucose, glutamine is capable of fueling cell bodies. It donates nitrogen to cells via anabolic reactions and provides carbons in the citric acid cycle. It is critical in the gastrointestinal system in that it provides energy to the small intestine. Notably, intestine is the only organ in the body that uses glutamine as a primary energy source. The kidney, activated immune cells, and cancer cells also require glutamine, but not as a primary energy source.
Within a cell, glutamine is essential for cell growth and protein translation. Moreover, it serves as a nitrogen donor and assists in maintaining the gradient across the mitochondrial membrane.
Normal cells require glutamine. On the other hand, cancer cells use glutamine in quantities much higher than normal cells. As discussed in the paper "Glutamine addiction: a new therapeutic target in cancer" by David R. Wise and Craig B. Thompson, cancer cells will sometimes exhibit what is called “glutamine addiction”. In this addiction, cancer cells will uptake glutamine from the body in much larger amounts than is necessary for cellular function. In fact, cancer cells will intake more glutamine than the cell can metabolize. Depriving cancer cells of this excess glutamine causes them to die. Such deprivation is the key to potential glutamine-based cancer therapy. Glutamine consumption can exceed the consumption of any other amino acid in the cell by tenfold. In cancer cells, a metabolic shift occurs so that glutamine replaces glucose as the major source of carbon for the cell.
The body can make enough glutamine for its regular needs, but extreme stress, such as heavy exercise or an injury), will make the body require more glutamine. Most glutamine is stored in muscles followed by the lungs, where much of the glutamine is made. Usually the body can make enough glutamine so it is not necessary to take supplements of glutamine. Certain medical conditions, including injuries, surgery, infections, and prolonged sites, can lower glutamine levels, however. In these cases, taking a glutamine supplement may be helpful.
Glutamine is important for removing excess ammonia, which is a common waste product in the body. Glutamine also helps your immune system function and is need for normal brain function and digestion. Glutamine is important in wound healing and recovery form an illness. When the body is stressed, it releases hormone cortisol into the bloodstream. This high concentration of cortisol will lower the body’s stores of glutamine. Other studies have shown that adding glutamine to enteral nutrition it will help reduce the rate of death in trauma and critically ill people. Clinical studies have found that glutamine supplements strengthen the immune system and reduce infections. Glutamine supplements also help in the recovery of severe burns. Another importance of glutamine is to protect the lining of the gastrointestinal tract known as the mucosa. People who have inflammatory bowel disease (IBD) may not have enough glutamine in their body. Two clinical trials found that taking glutamine supplements did not improve symptoms of Crohn’s disease. People with HIV or AIDs often experience severe weight loss, thus those people take glutamine supplements along with other nutrients including vitamin C and E, beta-carotene, selenium, and N-acetylcysteine to increase weight gain and help the intestines better absorb nutrients. Athletes who train for endurance events may reduce the amount of glutamine in their bodies, thus making them more prone to catch a code after an athletic event. Studies show that taking glutamine supplements resulted in fewer infections.
Glutamine and Cancer
It has been shown that some cancer cells have an addiction to glutamine in that there is an increased rate of glutamine uptake. The increase in glutamine uptake is due to glutamine playing roles other than providing nitrogen for protein (amino acid) and nucleotide biosynthesis.
The first signs of cancer cells relying on an excess of a given compound to produce energy were discovered by Otto Heinrich Warburg. Warburg noticed that the energy produced in most cancer cells was produced through glycolysis of excess glucose, which is in turn converted into lactic acid during lactic acid fermentation. Such a process is in contrast with energy production in normal cells, in which glycolysis still occurs, but is instead followed by oxidation of pyruvate in mitochondria. As such, Warburg concluded that these cancer cells must have devolved into a more primitive form of metabolism as seen in single-celled eukaryotes. Thus this effect of cancer cells up taking excess glucose for their energy needs has been dubbed the "Warburg Effect". Glutamine was later found to mirror this effect in some tumor cells.
Glutamine has been shown to participate in signaling and uptake of essential amino acids. For instance, it is capable of acting as the substrate of the mitochondria to maintain the integrity of the mitochondrion membrane potential. It also plays integral roles in a variety of anaplerotic reactions.
Glutamine donates nitrogen to cancer cells. Like all cells, cancer cells must synthesize nitrogen compounds to produce nucleotides and other amino acids. Glutamine donates the nitrogen that is necessary for the production of these compounds. Glutamine donates its amide group and is converted into glutamic acid. Glutamatic acid transfers its amine group by transaminases to α-ketoacids which is used to generate the nonessential amino acids. This decomposition provides the nitrogen with several amino acids including alanine, serine, aspartate, and proline. Tyrosine is the only nonessential amino acids not produced from either glucose or glutamine.
- Glutamine is Needed for the Uptake of Essential Amino Acids in Certain Cancer Cells and as a Molecular Signal
- Glutamine is imported through glutamine solute carrier SLC1A5 and quickly exported through the SLC7A5 amino acid transporter in exchange for extracellular essential amino acids. However, when the glutamine importer is impaired, the uptake of essential amino acids is also impaired. Such impairment suggests that glutamine is necessary for essential amino acid uptake. Without essential amino acids, the rapamycin-sensitive (mTORC1) is not activated. mTORC1 plays an essential role in regulation cell growth and protein translation as well as inhibiting macroautophagy. As such, inactivation of mTORC1 inhibits cellular growth and protein translation. Thus, glutamine acts as a signal to mTORC1 and as a resource of essential amino acids in some cancer cells.
- Glutamine Provides Anaplerosis in Cancer Cells
- Anaplerosis is a term used to describe the replenishing of the carbon pool in the mitochondrion. Oxaloacetic acid (OAA) is one of the substrates in mitochondria that eventually lead to synthesis of many essential biological macromolecules like cholesterol. In glioblastoma cells, glutamine metabolism provides the bulk of the OAA cellular pool. Thus, the increased rate of glutamine metabolism into OAA confirms glutamine as a primary substrate in cancer cells that provides the mitochondria with precursor macromolecules to carry out its metabolic functions.
- c-Myc Regulate Glutamine Metabolism in Cancer Cells
- The synthesis of purines and pyrimidine uses glutamine as a source of nitrogen in five enzymatic steps. Three out of the five steps are regulated by c-MYC (Myc), a DNA transcription factor. Oncogenic levels of Myc promote increased glutaminolysis at the transcription level and the metabolism of glutamine into lactic acid. The catabolism of glutamine provides cells with carbons for anaplerosis and NADPH production.
- Myc is a transcription factor that codes for a protein that binds to DNA. In a cancerous cell, Myc is amplified. Myc uptakes glutamine and converts it to glutamic acid and lactic acid. Myc over expression leads to increased catabolism of glutamine, which leads to a larger amount of carbon in the cell, which allows the cell to produce more NADPH. This over-expression of Myc triggers the metabolic switch from glucose to glutamine as the source of carbon for the cell.
- Glutamine-based cancer therapy
- Glutamine addiction in some cancer cells is a target for new cancer therapies. Further research is needed to determine a non-toxic dosage; that is, a dosage that does not inhibit glutamine production indiscriminately and does so only in cancerous cells.
- Since cancer cells are dependent on glutamine, starving these cells of glutamine will cause them to die. Thus, glutamine has become a target for new cancer treatments. New treatments have attempted to deny cancer cells their source of glutamine by reducing the amount of glutamine in the body. However, as glutamine is essential for many other processes in the body, such as synaptic communication in the brain, removing glutamine from the body is not a feasible treatment and is very dangerous. Other treatment methods have attempted to reduce the ability of the cell to uptake glutamine by targeting Myc and other proteins that are responsible for transporting glutamine into the cell. Other treatments have attempted to reprogram the mitochondria so that it will no longer depend on gluatmine. Another treatment involves targetting mTOR’s glutamine response. These treatments show more promise and less harm than removing all glutamine from the body.
- These therapeutic methods target major glutamine activity in cancer cells:
- Glutamine uptake and mTOR activation: L-γ-glutamyl-p-nitroanilide (GPNA) inhibits SLC1A5, a target for Myc. Such inhibition suppresses glutamine uptake in the cell. 2-aminobicyclo-(2,2,1)heptanecarbozylic acid (BCH) also inhibits SLC7A5 and blocks mTOC activation, inducing autophagy.
- Glutamine-dependent anaplerosis and activity in mitochondria: Studies suggest that carbons derived from glutamine enter the citric acid cycle via transaminase. Therefore, Amino-oxyacetic acid (AOA), a transaminase inhibitor, shows potential as a promising cancer therapeutic. Additionally, the regeneration of mitochondrial NAD+ may prevent the entry of glutamine through the citric acid cycle. Metaformin, a biguanide class drug, inhibits this mechanism.
- These therapeutic methods target major glutamine activity in cancer cells:
Usage
- Wound Healing
- Inflammatory Bowel Disease
- HIV/AIDS
- Obesity
- Peritonitis
- Athletes
- Cancer
- etc.
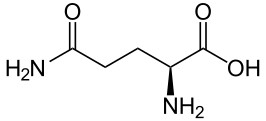
Glutamic acid - Glu/ E
[edit | edit source]
Structure The molecular formula of glutamic acid is C5H9NO4. Its molecular mass is 147.13 g/mol. Also known as glutamate, Gluctamic acid is a polar amino acid that has carboxylic acid group, which loses a proton to become carboxylate group for physiological pH and has a negative charge; the carboxylic acid group of the amino acid has a pKa value of 4.3, which is a little basic than the terminal α-carboxyl group and that of aspartic acid. The pKa of glutamic acid is significantly higher than that of aspartic acid due to the inductive effect o the additional methylene group. In some proteins, due to a vitamin K dependent carboxylase, some glutamic acid will be dicarboxylic acids, referred to as γ carboxyglutamic acid, that form tight binding sites for calcium ion. Glutamic acid and α-ketoglutarate, an intermediate in the Krebs cycle, are interconvertible by transamination. Glutamic acid can therefore enter the Krebs cycle for energy metabolism, and be converted by the enzyme glutamine synthetase into glutamine, which is one of the key players in nitrogen metabolism.
Function Glutamic acid is highly involed in metabolism. In citric acid cycle, transamination of alpha-ketoglutarate with alanine or aspartate each gives off glutamate and pyruvate or oxalatate respectively. Pyruvate and oxalatate formed fram transamination play critical roles in cellular metabolism.
Glutamic acid is a non-essential amino acid. It plays an important role in DNA synthesis. It also assists in wound and ulcer healing. Glutamic acid takes places in the excitatory neurotransmitter and the metabolism of sugars and fats. It aids potassium move through the blood-brain barrier. Glutamic acid is a source of fuel for the brain. It is capable to attach to amine group to form glutamine. The process of forming glutamine will detoxifies ammonia that the body contains.
Glutamic acid can be used in correcting personality disorders and treating childhood behavioral disorders. It also takes places in treating epilepsy, mental retardation, muscular dystrophy, ulcers, and hypoglycemic coma.
Other minor uses include flavor enhancer, GABA precursor, nutrients, and fertilizers for plants
Synthesis A biosynthesis of glutamic acid involves various schemes. The most common scheme is the conversion of glutamine to glutamic acid by adding water molecules with glutaminase as a helper enzyme. The side product is an ammonia group. Addition of water to a N-Acetylglutamic acid also produce glutamic acid and acetate. Ketoglutaric acid is another common precursor in synthesis of glutamic acid. Addition of NADPH ad ammonia or alpha amino acid produces glutamic acid. Such enzymes involved are glutamate dehydrogenase and transaminase. Other methods include 1-pyrroline-5-carboxylate + NAD+ + HOH and N-formimino-L-glutamate + FH4.
Glutamic acid is easily converted into proline. First, the γ carboxyl group is reduced to the aldehyde, yielding glutamate semialdehyde. The aldehyde then reacts with the α-amino group, eliminating water as it forms the Schiff base. In a second reduction step, the Schiff base is reduced, yielding proline.
Glycine - Gly/ G
[edit | edit source]
Structure Glycine's molecular formula and mass are C2H5NO2 and 75.07 g/mol. Being the smallest amino acid out of all 20 amino acids, glycine only has a hydrogen atom as its substituent. For this reason, it has the ability to fit into tight spaces of molecules where no other amino acid could possibly fit therefore glycine is evolutionarily conserved. Most proteins contain small amount of glycine, however collagen is one of the exception that contains 35% glycine. Thus, if glycine were cleaved from an amino acid chain composing a whole protein, it would either alter the function of that protein, or denature it entirely. It is also the only achiral amino acid since its R group is simply a H atom. In particular it does not favor the helix formation.
Functions Glycione is non-essential amino acids meaning the human can manufacture it in their body. It serves an important role in maintaining central nervous and digestive systems. Glycine prevents the breakdown of muscle by increase creatine, which is a compound that helps build muscle mass. Glycine also keeps the skin firm and flexible. Without glycine, the skin can be damage from the UV rays, oxidation and free radical.
Glycine regulates blood sugar levels and helps provide glucose for the body.
Glycine serves as an inhibitory neurotransmitter in the central nervous system, especially in the spinal cord. When glycine binds to receptors, it activates chloride ion channels to open. As chloride ions enter the channels, the membrane becomes hyperpolarized, causing an inhibitory postsynaptic potential (IPSP).
Some disorders that can be treating using glycine is used for treating schizophrenia, stroke, benign prostatic hyperplasia (BPH), and some rare inherited metabolic disorders. It is also used to protect kidneys from the harmful side effects of certain drugs used after organ transplantation as well as the liver from harmful effects of alcohol. Other uses include cancer prevention and memory enhancement.
Some people apply glycine directly to the skin to treat leg ulcers and heal other wounds. The body uses glycine to make proteins. Glycine is also involved in the transmission of chemical signals in the brain, so there is interest in trying it for schizophrenia and improving memory. Some researchers think glycine may have a role in cancer prevention because it seems to interfere with the blood supply needed by certain tumors.
Biosynthesis Glycine is a derivative form of serine and 3-phosphoglycerate. The conversion of serine requires a specific enzyme called serine hydroxymethyltransferase and co-factor pyridoxal phosphate. The process can be simplied as the following reaction: serine + tetrahydrofoate -> glycine + N5, or N10-methylene tetrahydrofolate + water.
The reaction continues to carry out in the liver. Glycine synthase is used as enzyme in the conversion of N5, or N10-methylene tetrahydrofolate. In this reaction, carbon dioxides, ammonium, NADH, and protons transform the tetrahydrofolate molecule into glycine.
Degradation of glycine has three pathways. The most common pathway is the opposite of the previous reaction: conversion of glycine into a tetrahydrofolate molecule. Another pathway is the conversion of serine into pyruvate and serine dehydratase. The last pathway involves converting glycine to gloxylate by D-amino acid oxidase. This pathway leaves glycoxylate oxidized to oxalate.
Histidine - His/ H
[edit | edit source]
Structure Histidine, C6H9N3O2, is also called 2-amino-3-(1H-imidazol-4-yl)propanoic acid. Its molecular mass is 155.15 g/mol. It is a basic, polar amino acid with an imidazole group, which is an aromatic ring that can be of positive charge and hydrophilic. The imidazole group of the amino acid has a pKa value of 6, which can be either uncharged or positively charged at neutral pH. This amino acid is often present in active sites of enzymes wherein the imidazole group acts as a buffer (proton acceptor or donor) for chemical reactions. Histidine is a precursor of histamine, a compound released by the immune system cells during an allergic reaction.
Features At a physiological pH of around 7, the Henderson-Hasselbalch equation can be used to give a ratio of deprotonation/protonation of the imidazole side chain (pKa = 6). As it turns out, the histidine side chain is approximately 10% protonated at a neutral pH. That is not a negligible amount and it gives the histidine residue a certain amount of buffering capacity. The basic nitrogen activates imidazole sites as a nucleophile.
Functions Histidine is found in high concentrations in hemoglobin. As a result, it aids in treatment of anemia and maintaining optimal blood pH. Also, histidine is the precursor of histamine, which is involved in local immune responses.
Histidine is an essential amino acid, which means that the body cannot manufacture it. Histidine plays important roles in stimulating the inflammatory response of skin and mucous membranes. It also stimulates the secretion of the digestive enzymes gastrin and acts as the source and control for histamine levels. Histidine is required for growth and for the repair of tissues, as well as the maintenance of the myelin sheaths that act as protector for nerve cells. Histidine is also required to manufacture both red and white blood cells. With histidine in the body, it helps protect the body from damage caused by radiation and in removing heavy metals from the body. Histidine is also in the stomach. It is helpful in producing gastric juices, and people with a shortage of gastric juices or suffering from indigestion, may also benefit from this nutrient. It is thought that histidine may be beneficial to people suffering from arthritis and nerve deafness. This is not conclusively proven. Histidine is also used for sexual arousal, functioning and enjoyment. Histidinemia is an inborn error of the metabolism of histidine due to a deficiency of the enzyme histidase, where high levels of histidine are found in the blood and urine, and may manifest in speech disorders and mental retardation. There are no reported side effects with histidine, but too high levels of histidine may lead to stress and mental disorders such as anxiety and people with schizophrenia have been found to have high levels of histidine. People suffering from schizophrenia or bipolar (manic) depression should not take a histidine supplement without the approval of their medical professional.
Metabolism Histidine can be converted into histamine by histidine decarboxylase. The carboxyl group leaves histidine.
Food sources Food that contain histidine are dairy, meat, poultry, fish, rice, wheat, and rye.
Isoleucine - Ile/ I
[edit | edit source]
Structure Isoleucine, HOOCCH(NH2)CH(CH3)CH2CH3, is also known as a 2-amino-3-methylpentanoic acid and has a molar mass of 131.17 g/mol. Isoleucine is a nonpolar, aliphatic or hydrophobic amino acid that has two chiral centers for α-carbon atom and the R group. Isoleucine, because it contains two stereocenters, is a diastereomer. If it weren't for the selectivity of living things for one particular stereoisomer, there would be 4 possible stereoisomers because of the 2 chiral centers. However, only one version persists in living organisms: the 2S, 3S version. The structure stabilizes water-soluble proteins by hydrophobic effect.
Features Isoleucine cannot be distinguished by MS from leucine because of the simple fact that they have the same molecular weight. Instead, these two residues would usually have to be isolated and characterized by HPLC or TLC against known standards.
Isoleucine is also degraded into succinyl CoA and acetal CoA and consumed by TCA cycle.
Functions Isoleucine is an essential amino acid, meaning the human body cannot manufacture it. It is needed for the formation of Hemoglobin and to regulate blood sugar and energy levels. Isoleucine serves important roles in muscle strength and endurance and is a source of energy for muscle tissues.
Isoleucine promotes muscle recovery after an intense workout. Isoleucine is necessary for the formation of hemoglobin as well as assisting with regulation of blood sugar levels as well as energy levels. It is also involved in the formation of blood clots.
Symptoms of people with a deficiency of isoleucine may result in headaches, dizziness, fatigue, depression, confusion as well as irritability. Symptoms of deficiency may mimic the symptoms of hypoglycemia. This nutrient has also been found to be deficient in people with mental and physical disorders, but more research is required on this. Consuming higher amounts of isoleucine is not associated with any health risks for most people but those with kidney or liver disease should not consume high intakes of amino acids without medical advise. People who take in higher amounts of isoleucine report elevated urination. People involved with strenuous athletic activity under extreme pressure and high altitude may benefit from supplementation of this nutrient.
Food sources of isoleucine Food containing isoleucine are almonds, cashews, chicken, eggs, fish, lentils, liver, meat, etc.
Biosynthesis Synthesis of iseoleucine involves multiple steps. Isoleucine can be derived from pyruvate and ketoglutarate. Catalytic enzymes required are the followings:
1. Acetolactate synthase 2. Acetohydroxy acid isomemoreductase 3. Dihydroxyacid dehydratase 4. Valine aminotransferase
Industrially, isoleucine can be synthesized from 2-bromobutane and diethylmalonate.
Leucine - Leu/ L
[edit | edit source]
Structure Leucine's molecular formula and mass are C6H13NO2 and 131.17 g/mol respectively. Leucine, also known as a 2-amino-4-methylpentanoic acid, has aliphatic R group. It is one of the three amino acids with branched hydrocarbon side chains (generally buried in folded proteins) and result as a nonpolar or hydrophobic amino acid. The hydrophobic effect counts for stabilization of water-soluble proteins.
Features Leucine cannot be distinguished by MS from isoleucine for the simple fact that they have the same molecular weight. Instead, these two residues would usually have to be isolated and characterized by HPLC or TLC.
Functions Leucine has all functions of the amino acid Isoleucine as their similarity in branched hydrocarbon side chain. Leucine facilitates skin healing and bone healing by modulating the release of natural pain-reducers, Enkephalins. It is also a precursor of cholesterol and increases the synthesis of muscle tissues by slowing down their degradation process. Leucine is an essential amino acid. It is essential in promoting growth in infant and regulating nitrogen concentration in adults. Leucine is generally used as a flavor enhancer.
Deficiency and Excess Deficiency of this particular amino acids can result in Hypoinsulinemia, Depression, Chronic fatigue syndrome, Kwashiorkor (or starvation), etc. Excess of Leucine leads to Ketosis.
Biosynthesis As an essential amino acid, leucine cannot be synthesized in human bodies, and must be obtained from outside sources. Starting from pyruvic acid, the conversion includes valine, ketovalerate, isopropylmalate, and ketoisocaproate via reduction. Enzymes required are: 1. acetolactate synthase, acetohydroxy acid isomeroreductase, dihydroxyacid dehydratase, isopropylmalate synthase and isomerase, and leucine aminotransferase.
Lysine - Lys/ K
[edit | edit source]
Structure Lysine is an essential amino acid. This means that is is necessary for human health but the body cannot produce it so you have to get the amino acid from food or supplements. Lysine are the building blocks of protein. Lysine has a positively charged amine group chain. The ε-amino group has a significant high pKa value of about 10.8, which is more basic than the terminal α-amino group. This basic amino group is highly reactive and participates in the reactions at the active center of enzymes. Although the terminal ε-amino group is charged under physiological condition, the hydrocarbon side chain with three methylene group is still hydrophobic.
Features Lysine is a naturally occurring essential amino acid in human body. It promotes optimal growth of infants and nitrogen equilibrium in adults.
Functions Lysine can be a treatment of Herpes Simplex and virus-associated Chronic Fatigue Syndrome as it inhibits viral growth. It facilitates the formation of collagen, which is the main component of fascia, bone, ligament, tendons, cartilage and skin. It also helps in absorption of calcium, which is critical in bone growth of infants.
Lysine is important for proper growth, and it plays an essential role in the production of carnitine, a nutrient responsible for converting fatty acids into energy and helping to lower cholesterol. Lysine helps the body absorb calcium, and it plays an important role in the formation of collagen, a substance important for bones and connective tissues including skin, tendon, and cartilage.
Herpes Simplex Virus (HSV) Consuming lysine on a regular basis may help prevent outbreaks of cold sores and genital herpes. Lysine has antiviral effects by blocking the activity or arginine, which promotes HSV replication. It has been studied that lysine at the beginning of a herpes outbreak did not reduce symptoms. Studies show that lysine with L-arginine makes bone building cells more active and enhances production of collagen. No studies have examined whether lysine helps prevent osteoporosis in humans.
Osteoporosis Lysine helps the body absorb calcium and thus decreases the amount of calcium that is lost in urine. Calcium is essential for strong bones so some researchers assumed lysine may help prevent bone loss associated with osteoporosis.
Deficiency and Excess Deficiency of lysine is seen in Herpes, Chronic Fatigue Syndrome, AIDS, Anemia, hair loss, and weight loss, etc. Having excessive lysine can result in high concentration of ammonia in the blood. Most people get enough lysine in their diet, although athletes, vegans who do not eat beans, as well as burn patients may need more. Not enough lysine can cause fatigue, nausea, dizziness, loss of appetite, agitation, bloodshot eyes, slow growth, anemia, and reproductive disorders. For vegans, legumes such as beans, peas, and lentils are the best sources of lysine.
Food Sources Foods rich in lysine are meat, cheese, fish, nuts, eggs, soybeans, spirulina, and fenugreek seed. Brewer's yeast, beans, and other legumes, and dairy products also contain lysine, Many nuts contain lysine.
Methionine - Met/ M
[edit | edit source]
Structure Methionine is one of the two amino acids with side chain containing sulfur. It contains a largely aliphatic side chain that includes a thioether (-S-) group. Unlike Cysteine, the chemical linkage of the sulfur in methionine is thiol ether. This sulfur does not participate in covalent bonding like that of cysteine. The high inclination of the sulfur oxidation in methionine is one of the causes of smoking-induced emphysema in the human lung tissue.
Features Methionine is a naturally occurring essential amino acid, which plays a critical role in supplying free methyl groups and sulfur in metabolism. It is also one of only two amino acids coded for by a single codon.
Functions Methionine helps the breakdown of fat and reduces blood cholesterol levels. It is an antioxidant that neutralizes free radicals and removes waste in the liver. Synthesis of DNA and RNA requires the presence of Methionine. It is also a precursor of several critical amino acids, hormones, and neurotransmitters in human body. Its AUG codon also serves as a "start" signal for ribosomal translation of messenger RNA or mRNA; this means that every peptide chain began with an methionine residual at its N-terminal. It may however be removed later on by cleavage.
Deficiency and Excess Methionine deficiency can be seen in chemical exposure and vegetarians. Severe liver disease can result from having excessive methionine.
Phenylalanine - Phe/ F
[edit | edit source]
Structure The amino acid phenylalanine is a derivative of alanine wherein a phenyl group takes the place of one of the hydrogens on the CH3 group. Phenylalanine has stronger hydrophobic properties when compared to the other aromatic amino acids, i.e. tyrosine and tryptophan. Tyrosine and tryptophan are less hydrophobic than phenylalanine due to their hydroxyl and indol substituents. Phenylalanine is often found buried in the proteins due to its hydrophobicity. Neighboring phenyl rings (on adjacent amino acids) can stabilize each other by pi stacking.
Features Individual amino acids as well as peptides are occasionally analyzed by UV light. Phenylalanine, along with the few other aromatic amino acids, fluoresces when UV light is applied. UV light can be a useful technique for verifying the presence of Tyr, Phe, and Trp. It can also quantify those amino acids if a sensitive enough assay is developed.
Functions Phenylalanine is a precursor of the amino acid tyrosine, which gives rise to neurotransmitters, such as dopamine, norepinephrine and epinephrine. It can be used to manage certain types of depression as a powerful anti-depressant and can also enhance memory, thought, and mood. This amino acid also plays a role in decreasing blood pressure in hypertension. The D form of phenylalanine can be used to reduce pain in arthritis which is a rare instance among amino acids. Phenylalanine is a naturally occurring amino acid that promotes growth in infants and regulates nitrogen concentration in adults.
Deficiency and Excess Deficiency of Phenylalanine can be seen in depression, AIDS, obesity, Parkinson's Disease, etc. Some people have a autosomal recessive genetic disorder called phenylketonuria, or PKU. This disorder is due to the lack of an enzyme that breaks down phenylalanine amino acids, which leads to a large accumulation of this amino acid, and in large quantities, phenylalanine is toxic, particularly to the brain. This leads to the possibility of mental retardation from this disorder. As a result, babies were blood tested early for signs of PKU, and if they have it then they must follow a strict diet that reduces the amount of natural phenylalanine in the food.
Proline - Pro/ P
[edit | edit source]
Structure Proline is one of the twenty DNA-encoded amino acids. It is unique among the 20 protein-forming amino acids because the α-amino group is secondary rather than primary as other amino acid. The distinctive cyclic structure of proline side chain locks its φ backbone dihedral angle at approximately -75°, giving proline an exceptional conformational rigidity compared to other amino acids. Hence, proline loses less conformational Entropy upon folding, which may account for its higher prevalence in the proteins. Proline, strictly speaking, can also be referred to as an imino acid. It greatly influences protein architecture because of its ring structure that makes it more conformationally restricted than the other amino acids.
Functions Proline behaves as a structural disruptor in the middle of regular secondary structure elements. However, proline is commonly found as the first residue of an alpha helix and in the edge strands of beta sheets. Proline is most commonly found in turns, which may account for the curious fact that proline is usually solvent-exposed although it has a completely aliphatic side chain. Because proline lacks of hydrogen on the amide group, it cannot act as a hydrogen bond donor, only as a hydrogen bond acceptor. Proline is important in healing, cartilage building, and in flexible joints and muscle support. It also helps reduce the sagging, wrinkling, and aging of skin resulting from exposure to the sun. Proline by breaking down protein and helps create healthy cells. It is essential both to skin health, and for the creation of healthy connective tissues and also muscular tissue maintenance.
Deficiency and Excess Proline deficiency is generally caused by people who perform prolonged exercises. Vitamin C deficiency will also cause proline to be lost in the urine because of collagen breakdown. Generally, people's body with proline deficiency tends to metabolize muscle cells instead of carbohydrates first if glucose levels are low. Proline is needed to maintain proper collagen creation and stabilize muscular tissue as well. The lack of proline could lead to symptoms such as fatigue, weight loss, dehydration, dizziness, and nausea.
Serine - Ser/ S
[edit | edit source]
Structure This amino acid's R group is a hydroxyl group attached to a CH2 group. The hydroxyl group is polar giving serine polar/hydrophilic properties. It has a pH of 5.68. pKa = 2.21, 9.15.
Features Serine is a non-essential amino acid which means it can be synthesized by the human body. For instance, serine can be synthesized from glycine. Serine is a precursor of glycine and cysteine.
Biosynthesis The biosynthesis of serine begins with the oxidation of 3-phosphoglycerate (an intermediate in glycolosis) to 3-phosphohydroxypyruvate which is then transaminated to 30phosphoserine. This last intermediate is then hydrolyzed to serine.
Function Serine is a non-essential amino acid which means it can be synthesized by the human body. For instance, serine can be synthesized from glycine. Serine is also a precursor of glycine and cysteine. Serine is found in phospholipids, active sites of trypsin and chymotrypsin. It can synthesize pyrimidines and proteins, cysteine and tryptophan. It is also involved in fat and fatty acid formation, muscle synthesis. Serine can be deaminated by the catalyst serine dehydratase, yielding to pyruvate and ammonium. The deamination of threonine follows a similar process.
Threonine - Thr/ T
[edit | edit source]
Structure Threonine is a polar, uncharged amino acid. Its side chain contains a secondary alcohol and a methyl group; hence it can be characterized as a hydrophilic amino acid. Threonine incorporates two chiral centers, just like isoleucine. If it weren't for the selectivity of living things for one particular stereoisomer, there would be 4 possible stereoisomers because of the 2 chiral centers. However, only one version persists in living organisms: the 2S, 3R version.
Features Threonine is an essential amino acid, which means it cannot be synthesized by the human body. Humans must ingest it in the form of threonine-containing foods.
Functions Threonine aids the formation of elastin and collagen. In the immune system, threonine aids in the formation of antibodies. It also promotes growth and function thymus glands and absorption of nutrients. In addition, threonine is the precursor to isoleucine. Threonine can be deaminated by the catalyst threonine dehydratase, yielding to α-ketobutyrate and ammonium. The deamination of Serine follows a similar process.
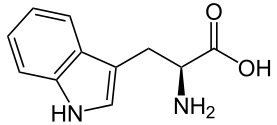
Tryptophan - Trp/ W
[edit | edit source]
Structure Tryptophan is an amino acid of aromatic group of an indole group bonded to a methylene group as the side chain, which is of two aromatic rings of nitrogen and hydrogen group and is hydrophilic. One of the side chains is 5-membered while the other is 6, and 2 carbons are shared by both aromatic rings.
Features Individual amino acids as well as peptides are occasionally analyzed by UV light. Tryptophan, along with the few other aromatic amino acids, fluoresces when UV light is applied. UV analysis can be a useful technique for verifying the presence of Tyr, Phe, and Trp. It can also quantify those amino acids if a sensitive enough assay is developed.
Functions Tryptophan is the precursor for various proteins, serotonin and niacin. It also promotes the formation of peptides and proteins. It is an essential amino acid, meaning it cannot be produced by the human body. It is usually present in peptides, enzymes, and structural proteins.
Deficiency and Excess Excess tryptophan has been linked with eosinophilia-myalgia syndrome (EMS). A deficiency of tryptophan is known as Pellagra which causes a deficiency of niacin. However, with vitamin supplements, this disease is no long as prominent. Symptoms of the disease include dementia and schizophrenia. Hartnup Disease is a genetic autosomal recessive disease in which a person cannot effectively digest this amino acid in their digestive tract. Although the disease of experiences symptoms similar to those of pellagra, however being slightly less severe. Patients suffering from the disease are generally seen with red rashes that are further aggravated by UV light from the sun. Further mental retardation could occur if not treated correctly with vitamin supplementation.
Tyrosine - Tyr/ Y
[edit | edit source]
Tyrosine Tyrosine is a nonpolar aromatic amino acid that contains a hydroxyl group attached to an aromatic ring. The hydroxyl group is particularly important because these residues are utilized in the phosphorylation of other proteins. Tyrosine is a non essential amino acid meaning it can be synthesized in the body. It is synthesized using phenylalanine in the body.
Features Individual amino acids as well as peptides are occasionally analyzed by UV light. Tyrosine, along with the few other aromatic amino acids, fluoresces when UV light is applied. UV light can be a useful technique for verifying the presence of Tyr, Phe, and Trp. It can also quantify those amino acids if a sensitive enough assay is developed.
Functions Tyrosine plays crucial roles in the human body: It helps deal with stress by becoming an adaptanogen helps minimize effects of the stress syndrome, in drug detoxification such as for cocaine, coffee and nicotine addictions. It reduces withdrawals and abuse. It assists in treating Vitiligo, pigmentation of skin, Phenylketonuria, the condition where phenylalanine is not metabolized. In addition, it is effective for depression treatment.
Tyrosine is also important in the production of epinephrine, norepinephrine, serotonin, dopamine, melanin, and enkephalins, which has pain-relieveing effects in the body. It also affects the function of hormones by regulating thryoid, pituatary and adrenal glands. For example, one need only look at the thyroid hormone thyroxine to see that it is synthesized from tyrosine. Tyrosine is known to dislodge molecules that may be harmful to cells, therefore it has protective qualities.
Deficiency and Excess Deficiency of tyrosine can result in low blood pressure, depression, and low body temperature. Tyrosine is a major amino acid responsible for skin, hair, and eye pigments. A loss of tyrosine amino acid in the body may lead to failure to form melanin pigments, resulting partial or full albinism. Interestingly enough, Tyrosine is produced mainly from phenylalanine in which a loss of one would lead to the increase of the other amino acid present in the organism's body.
Valine - Val/ V
[edit | edit source]
Structure Valine is an amino acid with an aliphatic, isopropyl side chain and is therefore a hydrophobic amino acid. Valine differs from threonine in that the OH group of threonine is replaced by a CH3 group. This is a nonpolar amino acid. It is an essential amino acid; therefore it cannot be produced by the human body. Being hydrophobic, this amino acid is often found in the interior of proteins.
Features In animals, valine must be ingested. In plants, it is created by using pyruvic acid, converting it to leucine followed by the reductive amination with glutamate. Valine is found in the following foods: soy flour, fish, cheese, meat and vegetables.
Functions Valine is essential in muscle growth and development, muscle metabolism, and maintenance of nitrogen balance in the human body. It can be used as an energy source in place of glucose. It can also be used as a treatment for brain damage caused by alcohol.
Deficiency and Excess Deficiency of valine affects myelin sheets of nerves. Maple Syrup Urine Disease is caused because leucine, valine and isoleucine cannot be metabolized.
Ionization of amino acids
[edit | edit source]The 20 standard amino acids have two acid-base gorups: the alpha-amino and the alpha-carboxyl groups attached to the Cα atom. Those amino acids with an ionizable side-chain (Asp,Glu,Arg,Lys,His,Cys,Tyr) have an additonal acid-base group. At low pH (i.e. high hydrogen ion concentration) both the amino group and the carboxyl group are fully protonated so that the amino acid is in the cationic form H3N+CH2COOH. As the amino acid in solution is titrated with increasing amounts of a strong base (e.g. NaOH), it loses two protons, first from the carboxyl group which has the lower pK value (pK=2.3). The pH at which Gly has no net charge is termed its isoelectric point, pI. The α-carboxyl gorups of all the 20 standard amino aicds have pK values in the range 1.8-2.9, while their α-amino groups have pK values in the range 8.8-10.8. The side-chains of the acidic amino acids Asp and Glu have pK values of 3.9 and 4.1, respectively, whereas those of the basic amino acids Arg and Lys, have pK values of 12.5 and 10.8, respectively. Only the side-chain of His, with a pK value of 6.0, is ionized within the physiological pH range (6-8). It should be borne in mind that when the amino aicd are linked together in proteins, only the side-chain groups and the terminal α-amino and α-carboxyl gorups are free to ionize.
Pyridoxal 5’-Phosphate-Mediated Decarboxylation of an �-Amino Acid
[edit | edit source]Step 1: The amino acid reacts with enzyme-bound pyridoxal 5�-phosphate (PLP). An imine linkage (CoeN) between the amino acid and PLP forms, and the enzyme is displaced.
Step 2: When the pyridine ring is protonated on nitrogen, it becomes a stronger electron-withdrawing group, and decarboxylation is facilitated by charge neutralization.
Step 3: Proton transfer to the � carbon and abstraction of a proton from the pyridine nitrogen brings about rearomatization of the pyridine ring.
Step 4: Reaction of the PLP-bound imine with the enzyme liberates the amine and restores the enzyme-bound coenzyme.
References
[edit | edit source]Berg, Jeremy, Tymoczko J., Stryer, L.(2012). Protein Composition and Structure.Biochemistry(7th Edition). W.H. Freeman and Company. ISBN1-4292-2936-5
Berg, Jeremy M., ed. (2002), Biochemistry (6th ed.) New York City, NY: W.H. Freeman and Company,
Hames, Daivd, Hooper,Nigel. Biochemistry, 3rd edition. Taylor and Francis Group. New York. 2005.
Wise R, David; Thompson B, Craig “Glutamine addiction: a new therapeutic target in cancer” Trends in Biochemical Sciences 35 (2010) 427—433. Retrieved 2010-10-16.
http://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/Hydrogen_bonds
http://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/Hydrophobic_interaction
http://en.wikibooks.org/wiki/Structural_Biochemistry/Organic_Chemistry/Organic_Functional_Group/Carboxyl
http://en.wikibooks.org/wiki/Structural_Biochemistry/pH
http://en.wikipedia.org/wiki/Alpha_carbon
http://en.wikibooks.org/wiki/Structural_Biochemistry/Enzyme
http://en.wikibooks.org/wiki/Structural_Biochemistry/Nucleic_Acid/DNA
http://en.wikibooks.org/wiki/Structural_Biochemistry/Nucleic_Acid/RNA
http://en.wikibooks.org/wiki/Structural_Biochemistry/Organic_Chemistry/Important_Organic_Reactions_in_Biochemistry/Peptide_Bonding
http://en.wikipedia.org/wiki/Ramachandran_plot
http://en.wikipedia.org/wiki/List_of_standard_amino_acids
http://en.wikipedia.org/wiki/Glycine
http://en.wikipedia.org/wiki/Alanine
http://en.wikipedia.org/wiki/Valine
http://en.wikipedia.org/wiki/Leucine
http://en.wikipedia.org/wiki/Isoleucine
http://en.wikipedia.org/wiki/Methionine
http://en.wikipedia.org/wiki/Serine
http://en.wikipedia.org/wiki/Cysteine
http://en.wikipedia.org/wiki/Threonine
http://en.wikipedia.org/wiki/Proline
http://en.wikipedia.org/wiki/Phenylalanin
http://en.wikipedia.org/wiki/Tyrosine
http://en.wikipedia.org/wiki/Tryptophan
http://en.wikipedia.org/wiki/Histidine
http://en.wikipedia.org/wiki/Lysine
http://en.wikipedia.org/wiki/Arginine
http://en.wikipedia.org/wiki/Aspartate
http://en.wikipedia.org/wiki/Glutamate
http://en.wikipedia.org/wiki/Asparagine
http://en.wikipedia.org/wiki/Glutamine
http://en.wikibooks.org/wiki/Structural_Biochemistry/Protein_function/Antibodies
http://en.wikipedia.org/wiki/Ramachandran_plot
http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6VS6-4V16RKY-1&_user=4429&_coverDate=12/31/2008&_rdoc=1&_fmt=high&_orig=search&_origin=search&_sort=d&_docanchor=&view=c&_acct=C000059602&_version=1&_urlVersion=0&_userid=4429&md5=4f09c690eac3ec0f0c4b8f31dc907058&searchtype=a
http://www.ncbi.nlm.nih.gov/pubmed/20570523
Wise DR. Thompson CB., "Glutamine Addiction: a new therapeutic target in cancer". Trends Biochem Sci. 2010 Aug; 35(8):427-33.
http://en.wikibooks.org/wiki/Structural_Biochemistry/Nucleic_Acid/Nitrogenous_Bases/Purines
http://en.wikibooks.org/wiki/Structural_Biochemistry/Nucleic_Acid/Nitrogenous_Bases/Pyrimidines
http://en.wikibooks.org/wiki/Structural_Biochemistry/Proteins/Amino_Acids#Asparagine_-_Asn.2F_N
http://en.wikibooks.org/wiki/Structural_Biochemistry/Hemoglobin
http://en.wikibooks.org/wiki/Structural_Biochemistry/Proteins/Purification/High_Pressure_Liquid_chromatography
http://en.wikibooks.org/wiki/Structural_Biochemistry/Chromatography/Thin_Layer
http://en.wikibooks.org/wiki/Structural_Biochemistry/Lipids/Cholesterol
http://en.wikibooks.org/wiki/Structural_Biochemistry/Nucleic_Acid/RNA/Messenger_RNA_(mRNA)
http://en.wikibooks.org/wiki/Structural_Biochemistry/Entropy
"Chemistry of Health", US Department of Health and Services, NIH Publications,Reprinted 2006
http://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?cid=5950 http://www.biology.arizona.edu/biochemistry/problem_sets/aa/alanine.html http://www.mayoclinic.com/health/l-arginine/NS_patient-arginine http://www.webmd.com/heart/arginine-heart-benefits-and-side-effects http://www.anyvitamins.com/asparagine-info.htm#Asparagine is required for http://www.biology.arizona.edu/biochemistry/problem_sets/aa/asparagine.html http://www.biology.arizona.edu/biochemistry/problem_sets/aa/aspartate.html http://www.umm.edu/altmed/articles/cysteine-000298.htm http://www.umm.edu/altmed/articles/glutamine-000307.htm http://www.biology.arizona.edu/biochemistry/problem_sets/aa/glutamate.html http://www.webmd.com/vitamins-supplements/ingredientmono-1072-GLYCINE.aspx?activeIngredientId=1072&activeIngredientName=GLYCINE http://www.anyvitamins.com/histidine-info.htm http://www.anyvitamins.com/isoleucine-info.htm http://www.umm.edu/altmed/articles/lysine-000312.htm
- ↑ Berg, Jeremy, Tymoczko J., Stryer, L.(2012). Protein Composition and Structure.Biochemistry(7nd Edition). W.H. Freeman and Company. ISBN1-4292-2936-5