Structural Biochemistry/Organic Chemistry/Carbohydrates
Background Information
[edit | edit source]Carbohydrates consist of numerous functions that are important to living organisms. They are also known as saccharides, or sugar if they exist in small quantities; these names are used interchangeably to describe the same thing. The simplest carbohydrates are the monosaccharides, also known as simple sugars. Disaccharides are double sugars, consisting of two monosaccharides joined by a covalent bond. Carbohydrates also include polysaccharides, which are polymers composed of many sugar building blocks. The name "carbohydrate" is derived from 'hydrates of carbon', and they arise from photosynthesis, where they exist as products.
Carbohydrates are the most abundant aldehyde compounds found in living organisms. They provide storage, transport starch and glycogen that provide energy to bodies, and contain structural components such as cellulose in plants and chitin in animals. Additionally, they contribute to the immune system, fertilization, pathogenesis, blood clotting, and development.
Structure of Carbohydrates
[edit | edit source]
The common chemical formula for carbohydrates is Cn(H2O)n, where the ratios are usually 1 Carbon: 2 Hydrogens: 1 Oxygen. Trioses, pentoses, and hexoses are found most commonly among monosaccharides.Their structure is composed of the functional groups, aldehyde and ketone, which are attached with various amount of hydroxylgroups. The hydroxyl groups are usually attached to the carbons not a part of the aldehyde or ketone functional groups, to form aldoses and ketosesStructural Biochemistry/Carbohydrates/Ketoses, respectively. The most elementary carbohydrates comprise polyhydroxyaldehydes (an aldehyde moiety) or polyhydroxyketones (a ketone moiety). There are four general classes of carbohydrates: monosaccharides, disaccharides, oligosaccharides, and polysaccharides.
Classification and Nomenclature
[edit | edit source]The most important carbohydrate is glucose. In general, monosaccharides have one carbonyl group (aldehyde, ketone, or acid), and the remaining carbons each bear one hydroxyl group. Monosaccharides can be linked together via ether and/or acetal bonds to form very large polymers called polysaccharides. A disaccharide consists of 2 linked monosaccharides and so on. Almost all saccharides in nature have at least one chiral carbon and they occur in nature as a single enantiomer. Glucose has 4 chiral carbons and has 15 other stereoisomers for a total of 16 possible stereoisomers of this gross structural formula.
The suffix –ose is often used in describing and naming carbohydrates. For example:
- A carbohydrate with 6 carbons is called a hexose
- A carbohydrate with 5 carbons is called a pentose
- A carbohydrate with an aldehyde as its carbonyl unit is called an aldose
- A carbohydrate with a ketone as its carbonyl unit is called a ketose
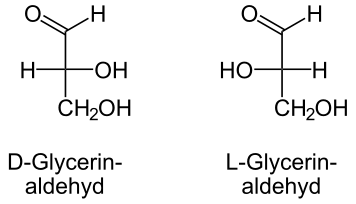
Looking at glyceraldehyde:
Enantiomers behave identically whether its a D or L conformation they both have the same boiling point, melting point and solubility. A different conformation seen in carbohydrates are diastereomers. Diasteriomers have the same chemical formula but different connectivity. A monosaccharide that has diastereomer conformation have different chemical and physical property. the D/l configuration applies to the highest numbered stereocenter (in most cases the highest stereocenter is the second to last hydroxyl group in a sugar molecule). The D/L configuration, like chiral molecules, means that the molecules will gradually rotate under polarization direction of linearly polarized light as it passes through, even under solution. D/L configuration also can be used in nomenclature to distinguish carbohydrate stereoisomers (meaning they have the same physical and chemical properties as well as the same formula, but rotate differently in polarized light) with the same formula. Naturally-occurring glyceralde has an R-configured chiral carbon. This is called D-glyceraldehyde. The opposite enantiomer is called L-glyceraldehyde. The D and the L refer to the configuration of the highest numbered chiral carbon when viewed in Fischer configuration. D-has the hydroxyl on the right side and the L has the hydroxyl on the left side. Almost all naturally occurring carbohydrates are of D-configuration.
Monosaccharides
[edit | edit source]The most basic carbohydrate is the monosaccharides (e.g. glucose, fructose, and galactose with the structural formula C6H12O6), and it consists of 3 carbons or more within the molecule. Monosaccharides may appear to be linear molecules, but when they come in contact with aqueous solutions, they tend to form 5-carbon ring structures, which makes the molecules more stable. Monosaccharides are classified by their placement of the carbonyl group and its chirality. If the carbonyl group is a ketone, the monosaccharide is referred to as a ketose. However, if the carbonyl is an aldehyde, the monosaccharide is called a aldose. There are more possible configurations of the aldose form than ketose due to the presence of more chiral carbons found in aldoses. Carbons that have a hydroxyl group (-OH), disregarding both the first and last carbons are asymmetric. The asymmetric carbons lead to the two possible forms (R and S)which corresponds to the D- and L- configurations. Combining two monosaccharides together would result in a disaccharide, linked via a glycosidic bond; and condensation reaction is the process that fuses two monosaccharides together. Such reactions form a disaccharide by removing a hydroxyl group from one monosaccharideMonosaccharides and a proton from the other.
Ring Structure of Monosaccharides After hemiacetals and hemiketals form, the carbohydrate will form a ring structure. For example, in Glucose the Hydroxyl group from the Carbon 6 will attack the carbonyl Carbon from Carbon 1, with a hemiacetal intermediate. The Carbon that will then change from a carbonyl carbon to a carbon with a hydroxy will then be called an anomeric carbon. An anomeric carbon is the hemiacetal or hemiketal that is bounded by an alcohol group and an ester bond. Anomers are another form of isomers that differ in the hemiacetals or hemiketals.The most common structure of a glucose ring is the alpha conformation in contrast with the sterically hindered Beta conformation. The alpha conformation is structure in which the hydroxyl in Carbon 1 is facing the opposite of the plane as carbon 6, while the beta conformation is facing in the same plane as the Carbon 6 thus causing steric hinderance. For this reason of steric hinderance the alpha conformation is much more stable than beta.
Monosaccharide Streoisomer

Modified Monsaccharide

Disaccharides and Polysaccharides
[edit | edit source]Monosaccharides not only form disaccharides, but polysaccharides as well. It is called an oligosaccharides if monosaccharides are linked by O-glycosidc bonds. The only difference between disaccharides and polysaccharides is that there are more monosaccharides combined together into a long chain, whereas disaccharides only consist of 2 combined monosaccharides. Polysaccharides are frequently long chains of glucose monomers bonded together. There are two types of polysaccharides: Homo-polysaccharides and Hetero-polysaccharides. Homo-polysaccharides are chains of one type of monosaccharides, while Hetero-polysaccharides consist of multiple types of monosaccharides. Both types of polysaccharides can exist in either branched or unbranched forms. Some crucial homo-polysaccharides, like starch, glycogen and dextrans, play a role in energy storage, while other homo-polysaccharides like cellulose and chitin have more structure-based roles. The diversity of these branched and unbranched carbohydrates is due to the number of hydroxyl groups present in the sugar. Any one of these hydroxyl groups can act as the alcohol in the formation of the glycosidic linkage. One sugar acts as the alcohol while the other has the anomeric carbon that ca for the O-glycosidic bond. This reaction can occur 1,4 or 1,6 depending on the alpha (pointing down) and beta (pointing up) orientations of the hydroxyl groups. These orientations organize the sugars into different structures. Alpha and Beta sheets form with sugars arranged with the least amount of steric hindrance.
Monosaccharides and disaccharides seem to be stable compounds, but that is not necessarily true, since they are hemiacetals with reactive carbonyls in their structure. Hemiacetals, with the general formula R1R'1C(OH)OR2 where R2 is not a hydrogen, and is formed by the reaction of carbonyl compounds with alcohols, and the carbonyl group is fairly reactive. Because the carbonyl groups are very reactive, they can oxidize to products in a short period of time. However, some carbohydrates are acetals and, as such, prevent carbohydrates from oxidizing. This occurs because the anomeric carbon is fixed in a glycosidic linkage. Because of this resistance against oxidation, acetals are known as the non-reducing sugars, like sucrose, while the hemiacetals are known as the reducing sugars, such as glucose, maltose, and lactose.
Two or more monosaccharides are linked by a glycosidic bond. The links can be alpha or beta depending on the position of the bond formed. Monosaccharides can also form bonds with amino acids to form glycoproteins. A sugar linked to an asparagine is N-linked, or linked from the sugar to the nitrogen of ASN. A sugar attached to Serine or Threonine is O-linked, or linked from the carbon on the sugar to the oxygen in those two amino acids. An Asparagine residue can only accept an oligosaccharides if the residue is a part of an Asn-X-Ser or Asn-x-Thr sequence, where x can be anytype of amino acid. Therefore, potential site can be detected within amino acid sequences.
Glucogen Metabolism Glucose metabolism and various forms of it in the process is described by the process below. Glucose-containing compounds are digested and taken up by the body in the intestines, including starch, glycogen, disaccharides and as monosaccharide. Glucose is stored in mainly the liver and muscles as glycogen. It is distributed and utilized in tissues as free glucose.

Hemiacetal and Hemiketal
[edit | edit source]An aldehyde or a ketone can react with an alcohol to yield a hemiacetal or a hemiketal.Hemiacetals and hemiketals are compounds that are derived from aldehydes and ketones respectively. The Greek word hèmi means half. These compounds are formed by formal addition of an alcohol to the carbonyl group. When the alcohol group is replaced by a second alkoxy group, an acetal or a ketal, respectively, is formed. For example, the acetal formation ends with having two ethers on the target carbon, whereas the hemiacetal and hemiketal has both an alcohol and ether group.
Example of Hemiacetal and Hemiketal Formation


In Carbohydrates
[edit | edit source]The same way aldehydes and ketones react with alcohols to form hemiacetals and hemiketals, respectively, carbohydrates react intermolecularly to form rings. When forming a ring 5 or 6 membered ring is most favorable and will only be formed. The Carbon 1 will be attacked by either the Carbon 5 or Carbon 6 hydroxyl group to form a 5 or 6 membered (respectively)carbohydrate ring.
The carbohydrates are a major source of metabolic energy, both for plants and for animals that depend on plants for food. Aside from the sugars and starches that meet this vital nutritional role, carbohydrates also serve as a structural material (cellulose), a component of the energy transport compound ATP, recognition sites on cell surfaces, and one of three essential components of DNA and RNA. Carbohydrates are called saccharides or, if they are relatively small, sugars.
References
[edit | edit source]Berg, Biochemistry, 6th Edition
http://themedicalbiochemistrypage.org/carbohydrates.html
Viadiu, Hector. "Carbohydrates." Chem 114A. UCSD, La Jolla. 19 Nov. 2012. Lecture.