Structural Biochemistry/Organic Chemistry/Organic Functional Group/Hydroxyl
Basic Information
[edit | edit source]The hydroxy-group is one of many functional groups studied in organic chemistry. The presence of a hydroxy group indicates that the molecule is either an alcohol or a carboxylic acid. The chemical representation of a hydroxy group is –OH, indicating an oxygen atom covalently bonded to a hydrogen atom.
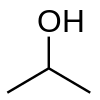
Within the general functional group of alcohol, there are three subgroups: primary, secondary, and tertiary alcohols. An alcohol is classified as a primary, secondary, or tertiary depending on the carbon it is attached to. An alcohol is primary if it is attached to a carbon with only one carbon-carbon bond. By the same token, an alcohol is secondary if the carbon to which it is attached is bound to two other carbons, and tertiary if the carbon’s remaining three bonds are bonded to other carbons. The following examples of primary, secondary, and tertiary alcohols are ethanol, isopropanol (IUPAC nomenclature: propan-2-ol), and tert-butyl alcohol (IUPAC nomenclature: 2-methylpropan-2-ol), respectively.
 |
 |
Physical Properties of Alcohols
[edit | edit source]The bond existing between oxygen and hydrogen is significantly shorter than the bonds formed between carbon and hydrogen. Hydroxy groups are inherently polar. The oxygen in the covalent bond is highly electronegative; thereby, pulling the majority of the electron density shared in the covalent bond towards itself. From this information it becomes clear that the oxygen bears a significant partial negative charge, while the hydrogen bears a partial positive charge of the same magnitude.
The presence of a highly electronegative group allows alcohols to hydrogen bond with other molecules. For example, in ethanol, the negatively charged oxygen on one ethanol molecule is attracted to a positively charged hydrogen molecule on another ethanol molecule. The ability of alcohols to hydrogen bond amongst themselves explains their high melting points. Alcohols also have higher boiling points than the hydrocarbons and ethers which contain a comparable number of carbon atoms.
The solubility of alcohols in water is interesting because alcohols have hydrophobic and hydrophilic centers, making them amphipathic or amphiphilic. The hydrocarbon portion of the alcohol is nonpolar, and thus resists dissolving in water; the polar alcohol group is hydrophilic and thereby promotes dissolution in water. In a small molecule such as propanol, the alcohol dissolves because the polar alcohol group outweighs the very short hydrogen chain. However, as the hydrocarbon chain increases beyond butanol to pentanol and hexanol, alcohols become virtually immiscible in water.
Alcohols are amphoteric species that may exist as both acids and bases. At low pH, or when surrounded by strong acids, they may be protonated to alkyloxonium ions. In contrast, at high pH and when surrounded by strong bases, they may exist as alkoxide ions.
Phenols
[edit | edit source]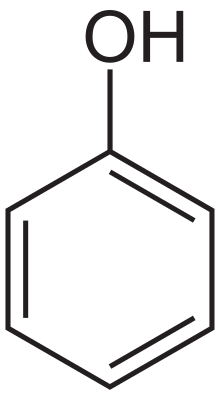
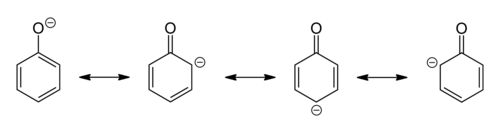
Phenols are hydroxy-arenes. They have some similarities to alcohols in chemical reactivity at oxygen, but they are much more acidic and much less basic. They appear to be enols (hydroxy attached to an alkene) and should be unstable relative to the keto form, but the gain in aromaticity in being in the enol favors the enol form relative to the keto. Examination of the resonance forms indicates that the hydroxyl donates electron density to the ring, hence the hydroxyl group is activating and ortho-para directing.

Nomenclature of Phenols
[edit | edit source]Substituted phenols are named as derivatives of the parent compound phenol with the hydroxyl-bearing carbon being designated as C-1. Some examples are:
| 2-Methyl-4,6-dinitrophenol | Benzene-1,3-diol |
|---|---|
 |

|
Acidity of Phenols
[edit | edit source]The pka of phenols is around 8-10. Phenols are substantially more acidic than alcohols. This is because phenoxide (the corresponding conjugate base) is resonance stabilized.
Synthesis of Phenols: Nucleophilic Aromatic Substitution
[edit | edit source]Direct concerted displacement of leaving groups on aromatic rings is not possible. In appropriately substituted rings, leaving groups may be replaced by hydroxyl involving nucleophilic processes via a 2-step mechanism: nucleophilic aromatic substitution.

Alcohol-like Reactivity of Phenols
[edit | edit source]Phenols undergo many of the same reactions as alcohols, especially where the nucleophicity of the corresponding phenoxide (alkoxide) is concerned.
Phenol alkylation via Williamson-Type Reaction

Synthesis of Alcohols
[edit | edit source]In industry, methanol is made on a multibillion-pound scale from a pressurized mixture of CO and H2 called synthesis gas.The reaction involves a catalyst consisting of copper, zinc oxide and chromium(III) oxide.
Changing the catalyst to rhodium or ruthenium leads to 1,2-ethanediol (ethylene glycol), an important industrial chemical that is the principal component of automobile antifreeze. Ethanol is prepared in large quantities by fermentation of sugars or by the phosphoric acid-catalyzed hydration of ethene (ethylene).
On a smaller than industrial scale, we can prepare alcohols from a wide variety of starting materials. For example, conversions of haloalkanes into alcohols by Sn2 and Sn1 processes featuring hydroxide and water. These methods are not as widely used as one might think, however, because the required halides are often accessible only from the corresponding alcohols. They also suffer from the usual drawbacks of nucleophilic substitution: Bimolecular elimination can be a major side reaction of hindered systems, and tertiary halides from carbocations that may undergo E1 reactions. Some of these drawbacks are overcome by the use of polar, aprotic solvent.
Conceptually, the easiest way to reduce a carbonyl group would be to add hydrogen, H-H, across the carbon-oxygen double bond directly. Although this can be done, it requires high pressures and special catalysts. A more convenient way is a polar process, in which hydride ion, and a proton, are delivered to the double bond, either simultaneously or sequentially.
Organometallic Reagents
[edit | edit source]Organometallic reagents of magnesium and lithium are used in the synthesis of alcohols. Since the metal in organometallic compounds are extremely electropositive, the carbon can react as if it were carrying a negative charge (giving it nucleophilic behavior). These organometallic compounds can attack the carbonyl group of aldehydes and ketones. An important feature of this reaction is that a new carbon-carbon bond is formed in the process.[1]

The first step of this reaction involves the nucleophilic carbon attacking the carbonyl carbon of a ketone or aldehyde to form the carbon-carbon bond. Simultaneously, two electrons from the carbon-oxygen double bond (carbonyl) are pushed by the electrons from the attacking carbon to form a metal alkoxide with the metal from the organometallic compound. Upon aqueous work-up, an alcohol is formed by hydrolyzing the metal alkoxide.[1]
Different types of alcohols can be formed by changing the compound reacting with the organometallic compound. For example, a reaction between an organometallic compound and formaldehyde will result in a primary alcohol. Reaction with any other type of aldehyde will produce secondary alcohols; reaction with ketones will yield tertiary alcohols.[1]
Hydroxylation
[edit | edit source]An important function of metabolic degradation is the introduction of hydroxy groups into unfunctionalized parts of molecules in our body (hydroxylation). A set of proteins, known as the cytochrome proteins, are vital biomolecules that are present in almost all living cells, which assist with the process of hydroxylation. An example of a cytochrome protein is cytochrome P-450. Cytochrome P-450 uses O2 to add hydroxy groups to alkyl substituents. The importance of hydroxylation can be signified by the excretion of drugs to prevent toxic accumulation. Hydroxylation helps increase the water solubility of compounds through addition of hydroxy groups to specific regions of the compounds. Thus in the liver, hydroxylation helps to accelerate the excretion of medicine that we ingest by making it more soluble. If it weren't for this process, ingested drugs would stay in our bodies much longer and have a much greater chance of toxic accumulation. [1]
Hydroxylation can also be selective in the addition of hydroxy groups to a molecule. Protein's undergoing hydroxylation can control the order in which hydroxy groups are added, as well as, the positions to which they are added. This is a key function of the process of hydroxylation, as it allows molecules to undergo oxidation at specific regions of the molecule. Also it allows molecules to bind or react with other compounds in specific orientations. [1]
Nomenclature
[edit | edit source]The system used to name organic molecules – IUPAC – is relatively simple to use for alcohols, though it can get more complicated if the molecule containing the alcohol group contains substituents containing other functional groups, such as alkenes, alkynes or halogens. The simplest case is when the molecule contains only hydrocarbons and the hydroxy group is the sole functional group, making it purely an alcohol.
In general, the presence of functional groups is indicated by a characteristic suffix that is distinct for each functional group and a number designating the location of the functional group on the molecule. For alcohols, the characteristic suffix used is –ol.
Case 1: Hydrocarbons and alcohols In this case, the longest chain of the hydrocarbons is counted – this becomes the primary hydrocarbon chain of the molecule. Then the longest carbon chain is numbered with the goal of putting as small a number as possible on the hydroxyl group. The longest chain is named as it is for alkanes, but the suffix for alcohols is –ol. Thus, for a propane chain which contains an alcohol group on the second carbon is named propan-2-ol or 2-propanol. Because the alcohol is the only functional group, it does not matter which of the two names is used. It is assumed that the “2” corresponds with the location of the hydroxy group. However, this rule does not always hold true when other functional groups are present.
Case 2: This is more complex because there is a hierarchy of functional groups in organic chemistry. Each functional group when isolated wants to be labeled as the smallest number. However, some functional groups are of “higher importance” and thus are numbered first. Also, if the alcohol is no longer the most “important” functional group, the molecule does not the suffix for the alcohol. This may present a slight problem initially, because the –ol was the only hint that the molecule possessed an –OH group. However, the presence of the –OH group is designated by using the word “hydroxy” after the number of the carbon on which the –OH group is bound to. An example is: 1-chloro-2-hydroxybenzene.
If there are multiple –OH groups present, the nomenclature is modified only slightly. The suffix used in the name is the number of alcohol groups followed by –ol. For example, a compound containing two –OH groups has a suffix of “diol”; for a compound with three –OH groups, the suffix is a “triol.” The numbers of the carbons containg the alcohol groups are stated in the same manner as if there were only one; giving the hydroxy group the lowest number possible. For example, a diol would be named as 3,3 pentandiol. This name indicates that the third carbon in the five-carbon chain is bonded to two –OH groups.
Unique Infrared Spectroscopy of Alcohols and Phenols
[edit | edit source]Infrared Spectroscopy is a technique used by chemists to elucidate structural information about an organic compound. Organic molecules absorb electromagnetic radiation in the infrared region and promote specific vibrational states. These vibrations are called fundamental absorptions, and they arise from the excitation from the ground state to the lowest-energy excited state. These modes of vibration are then picked up by a machine which interprets the radiation in the form of a spectrum. Since every type of covalent bond has a different natural frequency of vibration, two different molecules will never share the same infrared spectrum. The absorption of certain bonds have very distinct vibrational frequencies. A small range of absorption can be allocated to a specific type of bond. For instance, an absorption around 1715 is normally due to the carbonyl group.
Alcohols and Phenols have unique Infrared spectrum and usually show strong and broad streching bands between 3400 and 3300 cm−1. In solution, it is sometimes possible to observe a free stretching band at around 3600−1 with a sharp but weaker peak.
The broad peak that is well known as the alcohol or phenol peak is created by the stretching vibrations between the O-H. Intermolecular hydrogen bonding usually weakens the O-H bond, and shifts the band to a lower frequency. Phenols are known to have a broader O-H band than alcohols. However, it is hard to determine the difference by looking at the O-H band due to their similar shapes. The bending vibrations between the C-O-H usually yield weak and broad peaks around 1440–1220 cm−1. However, these peaks are difficult to see due to interference by similar bands from strong CH3 bending peaks. Strong C-O bond stretches are observed from 1260 – 1000 cm−1. Since the C-O bond is adjacent to the C-C bond, the shape and size of the peak can be used to determine whether an alcohol is primary, secondary, or tertiary. It can also be used to distinguish between phenols and alcohols. In addition, alcohols that are present in amides, ethers, and aldehydes can also be distinguished in IR spectrum. Infared spectrum is a useful analytical tool that is used to identify which function groups are present or which ones are not.
NMR of alcohol groups
[edit | edit source]Nuclear magnetic resonance (NMR) is a technique used to identify organic compounds. Since the atomic nucleus has a spinning charge due to the positively charged protons, it can generate a magnetic field. Because of this, the protons will have two orientations, a β and α spin state that have different energies in the presence of an external magnetic field. When the compound is exposed to electromagnetic radiation, resonance (when the nuclei is flipped back and forth between orientations) will occur and when the amount of radiation matched the difference in energy of the two spin states, an NMR signal is recorded and the NMR detector records peaks.
One of the most common forms of NMR is the proton NMR (H+ NMR). The NMR spectrum is read by the chemical shift of the different groups. In a proton NMR a compound with an -OH group will have a chemical shift (peak) at around 0.5-5.0 ppm. For example, ethanol will have a peak at around 4.0 ppm indicating the hydroxide groups while its two other peaks at around 1.0 ppm and around 3.0 ppm will indicate the position of the C-H protons in accordance to their position in the compound.
References
[edit | edit source]1. Schore, Neil E. (2011). Organic Chemistry Structure and Function 6th edition. W. H. Freeman.
2. Hammond, Christina N., Mohrig, Jerry R., Schatz, Paul F.. (2010). Techniques in Organic Chemistry 3rd Edition. W.H. Freeman.
3. Atkins, Peter, Jones, Loretta. (2008). Chemical Principles: The Quest for Insight 4th Edition. W.H. Freeman.