Structural Biochemistry/Carbohydrates/Polysaccharides


Polysaccharides are complex carbohydrate polymers consisting of more than 2 monosaccharides linked together covalently by glycosidic linkages in a condensation reaction. Being comparatively large macromolecules, polysaccharides are most often insoluble in water. Polysaccharides are extremely important in organisms for the purposes of energy storage and structural integrity.
There are two types of polysaccharides: homo-polysaccharides and hetero-polysaccharides. A homo-polysaccharide is defined to have only one type of monosaccharide repeating in the chain; whereas, a hetero-polysaccharide is composed of two or more types of monosaccharides. In both types of polysaccharide, the monosaccharide can link in a linear fashion or they can branch out into complex formations. It should also be noted that for a polysaccharide to be considered acidic it must contain one or more of the following groups: phosphate, sulfuric, or carboxyl.
Use of Polysaccharides
[edit | edit source]Polysaccharides have several roles. Polysaccharides such as starch, glycogen, and dextrans are all stored in the liver and muscles to be converted to energy for later use. Amylose and Amylopectin are polysaccharides of starch. Amylose has a linear chain structure made up of hundreds of glucose molecules that is linked by a alpha 1,4 glycosidic linkage. Due to the nature of these alpha 1,4 bonds, the macromolecule often assumes a bent shape. The starch molecules form a hollow helix that is suitable for easy energy access and storage. This gives starch a less fibrous quality and a more granule-like shape which is better suited for storage. Unlike the linear structure of Amylose, the Amylopectin starches are branched containing an alpha 1,6 glycosidic linkage about every 30 glucose units. Like amylose it is a homopolymer composed of many glucose units. Glycogen is found in animals, and it is branched like amylopectin. It is formed by mostly alpha 1,4 glycosidic linkages but branching occurs more frequently than in amylopectin as alpha 1,6 glycosidic linkages occur about every ten units. Other polysaccharides have structural functions. For example, cellulose is a major component in the structure of plants. Cellulose is made of repeating beta 1,4-glycosidic bonds. These beta 1,4-glycosidic bonds, unlike the alpha 1,4 glycosidic bonds, force cellulose to form long and sturdy straight chains that can interact with one another through hydrogen bonds to form fibers.
Polysaccharide Branching
[edit | edit source]Unbranched polysaccharides contain only alpha 1,4 linkages. However, there exists branched polysaccharides which are branched by virtue of certain molecules being linked to a molecule via alpha 1,4 and another via alpha 1,6 glycosidic bonds. The rate at which these bonds appear may vary. The plant based amylopectin contains a branch every 30 units while the animal based glycogen contains a branch approximately every 10 units. In digesting these branched polysaccharides, α-amylase is the relevant catalyst. α-amylase, however, only digests α-1,4 glycosidic bonds, leaving disaccharide/polysaccharide fragments containing α-1,6 bonds. These smaller fragments are known as Dextrins.
Glycogen and Starch
[edit | edit source]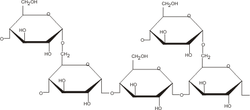
Many organisms store energy in the form of polysaccharides, commonly homopolymers of glucose. Glycogen, the polysaccharide used by animals to store energy, is composed of alpha-1,4-glycosidic bonds with branched alpha-1,6 bonds present at about every tenth monomer. Starch, used by plant cells, is similar in structure but exists in two forms: amylose is the helical form of starch comprised only of alpha-1,4 linkages, and amylopectin has a structure like glycogen except that the branched alpha-1,6 linkages are present on only about one in 30 monomers. These polysaccharides often contain tens of thousands of monomers, and each type is synthesized in the cell and broken down when energy is needed.
Glycogen metabolism is an intricate process involving many enzymes and cofactors resulting in the regular release and storage of glucose. This metabolic process is in turn broken down to glycogen degradation and synthesis. Glycogen synthesis is carried out by the enzyme glycogen synthase in which the activated form of glucose, UDP-glucose (uridine diphosphate), is formed by way of the reaction between UTP and glucose-1 phosphate. From this synthesis two outer phosphoryl groups are released from UTP producing the pyrophosphate compound. Pyrophosphate becomes an important aspect in this portion of the synthesis as the reaction to produce UDP-glucose is readily reversible. What allows the reaction to be driven forward is the hydrolysis of the pyrophosphate to orthophosphate in an irreversible reaction thus allowing the production of UDP-glucose to continue unhindered. The UDP-glucose is then attached to the non-reducing ends of glycogen. How this is accomplished is through an alpha-1,4-glycosidic linkage at the C-4 terminal with the terminal hydroxyl group ready to bind on glycogen. At this point the enzyme glycogen synthase plays the important role of catalyzing the attachment of UDP. Since an oligomer of at least four monomers is required for glycogen synthase to extend a chain, the process uses a primer that is itself provided by another enzyme, glycogenin. After several units of UDP have been attached to the glycogen by way of alpha-1,4 linkages, branching begins to take place by breaking an alpha-1,4 link and forming a alpha-1,6-link.A number of other enzymes, including insulin, play important roles in glycogen's synthesis. The breakdown of glycogen is completed through an entirely different biochemical pathway. Epinephrine and glucagon are signaling molecules whose binding to certain 7TM receptors activate the degradation, which is carried out in the cells by glycogen phosphorylase. This enzyme breaks up the polysaccharide chain by replacing the glycosidic bond with a phosphate group. As with its synthesis, glycogen's degradation requires numerous enzymes besides those mentioned here.
Starch is a good storage of carbohydrates because it is an intermediate compared to ATP and lipids in terms of energy. In plants, starch storage folds to allow more space inside cells. It is also insoluble in water, making it so that it can stay inside the plant without dissolving into the system. Starch can also be used as a back up source of energy when plants cannot obtain carbon dioxide, light, or nutrients from the surrounding soil.
Cellulose
[edit | edit source]Cellulose is the major polysaccharide found in plants responsible for structural role. It is one of the most naturally abundant organic compounds found on the planet. Cellulose is an unbranched polymer of glucose residues put together via beta-1,4 linkages, which allow the molecule to form long and straight chains. This straight chain conformation is ideal for the formation of strong fibers.
Although mammals cannot digest cellulose, it and other plant forms are necessary soluble fibers that mammals can eat. Pectin, for example, slows down the movement of food molecules in the digestive tract, which thereby allows for more necessary nutrients to be absorbed by the body instead of being quickly passed through as waste. Likewise, insoluble fibers like cellulose expedite the digestive movement of food molecules, which is imperative in the quick removal of harmful toxins.
Humans can't digest cellulose because we lack cellulases that would allow us to cleave the beta 1,4 linkages. However, some animals do eat and obtain energy from cellulose. One example of that is termites. These animals digest cellulose in a stepwise manner, using a combination of their own cellulases (produced in the foregut) and those of a microbial community resident in the distal parts of their digestive tract. This is a great example of a symbiotic relationship.
Cellulose is insoluble in water and aqueous solutions. It forms crystals and hydrogen bonds with amino acids. This quality of using intra and intermolecular hydrogen bonds to make crystals renders cellulose excessively insoluble in water and aqueous solutions. However, individual strands of cellulose aren't very hydrophobic as compared to other polysaccharides. It is the property of forming crystals that makes cellulose so insoluble.
Use of Cellulose
[edit | edit source]Cellulose has many uses, for example, as a gelling agent because of cellulose's properties of holding on to water. It is also used as an anticake agent, stabilizer, thickener and dispersing agent. Water cannot enter crystalline cellulose but dry cellulose absorbs water and it becomes flexible. Cellulose can give improved volume and texture particularly as a fat replacer in sauces and dressings but its insolubility means that all products will be cloudy.
Most papers are made of cellulose. The most important role of cellulose is that it is the major constituent of paper and cardboard and of textiles made from cotton, linen, and other plant fibers.
Cellulose can also be converted into cellophane, a thin transparent film, and into rayon, an important fiber that has been used for textiles since the beginning of the 20th century. Both cellophane and rayon are identical to cellulose in chemical structure. They are known as "regenerated cellulose fibers" and are usually made from viscose, a viscous solution made from celluloseare . A more recent and environmentally friendly method to produce rayon is the Lyocell process.
In the laboratory, cellulose is used as the stationary phase for TLC (thin layer chromatography). It is the raw material in the manufacture of nitrocellulose (cellulose nitrate) which was historically used in smokeless gunpowder and as the base material for celluloid used for photographic and movie films until the mid 1930s.
About a third of the world's production of purified cellulose is used as the base material for a number of water-soluble derivatives with pre-designed and wide-ranging properties dependent on groups involved and the degree of derivatization. Cellulose is also used to make hydrophilic and highly absorbent sponges. Derivatizing cellulose interferes with the orderly crystal-forming hydrogen bonding, described above, so that even hydrophobic derivatives may increase the apparent solubility in water. Methyl is thermogelling, which is made by methylating about 30% of the hydroxyl groups, forming gels above a critical temperature due to hydrophobic interactions between high-substituted regions and consequentially stabilized intermolecular hydrogen bonding. Such gels break down when cooling, in a manner similar to that causing the solubility minimum for non-polar gases; hydrophobic saccharides becoming less soluble as the temperature increases. This property is useful in forming films as barriers to water loss and for holding on to small gas bubbles.
Cellulose fibers are also used to make filter bed of inert material in liquid filtration.
Cellulase, an enzyme found in organisms that can digest cellulose, has recently garnered the attention of the scientific community and especially of alternative energy researchers who hope to produce biofuel (such as ethanol) by enzymatically breaking down the cellulose found in plants. How cellulase operates is not yet fully understood, but as more knowledge about the mechanism is uncovered researchers will be able to improve its efficiency (known cellulases are currently too slow to be used in industry) and apply its use to producing "green" energy sources. In this way, the most abundant source of bioenergy on Earth, cellulose, can become a part of the world's accessible energy supply. Some types of cellulase already find uses in industry, for example in food production and the textile industry.
Chitin
[edit | edit source]Chitin is a linear homopolysaccharide (long chain polymer) comprising N-acetyl-glucosamine, derivatives of glucose. These subunits form beta-glycosidic linkages similar to those formed by glucose molecules in cellulose. In fact, the only chemical difference from cellulose is the replacement of a hydroxyl group at C-2 with an acetylated amino group. Chitin can thus be described as cellulose, but simply with a different group at the second carbon. This increases hydrogen bonding, resulting in stronger molecules.
Chitin is the exoskeleton of many arthropods, and is the main component of cell walls in fungi, radulas of mollusks etc. Like cellulose, it is indigestible by vertebrate animals. Chitin has also been used as surgical thread, making it very valuable.
Glycosaminoglycan
[edit | edit source]Glycosaminoglycans (known as GAGs or mucopolysaccharides) are long unbranched polysaccharides made of repeating disaccharide units. Glycosaminoglycans are made from protein cores that are made in the endoplasmic reticulum that are posttranslationally modified by the Golgi. Here GAG disaccharides are added to protein cores to create proteoglycans.
GAGs are essential to life; they form important components of connective tissues and are found in collagen and elastin due to GAG chains that are covalently bonded to other proteins, forming proteoglycans. Because of this, water sticks to GAGs and, since water is not compressable, allows resistance to pressure. It is also used in smoke detectors due to its polarity.
Links
[edit | edit source]References
[edit | edit source]1. Berg, Jeremy M. 2007. Biochemistry. Sixth Ed. New York: W.H. Freeman. 310-323.