Structural Biochemistry/Catabolism
Overview
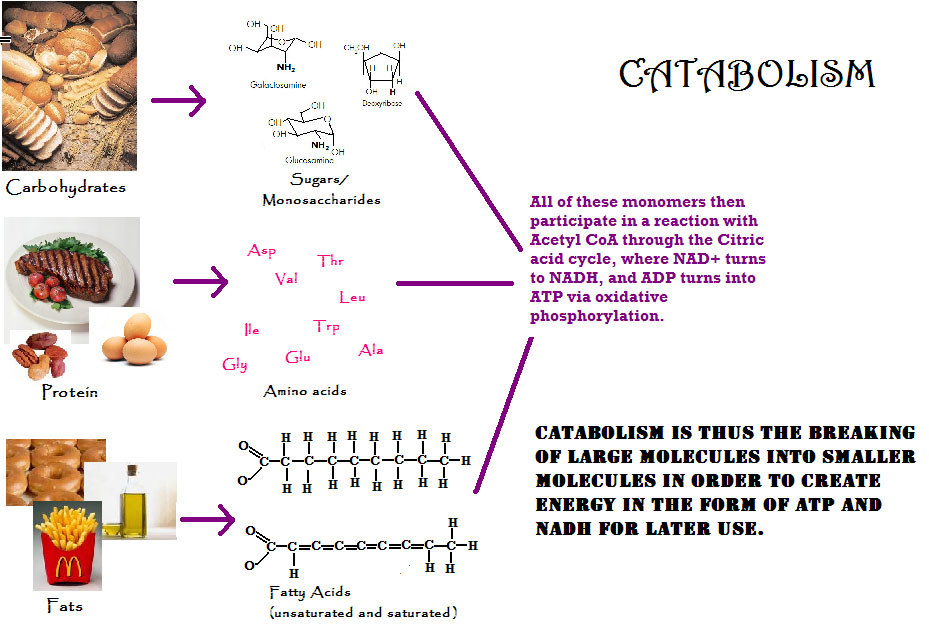
[edit | edit source]Catabolism is the release of energy from a set of metabolic pathways which break down molecules into smaller units, including the breaking down and oxidizing of food molecules. An example would be proteins, nucleic acids, lipids and polysaccharides being broken down into smaller molecules like amino acids, nucleotides, fatty acids, and mono saccharides. By glycolysis, the glucose is broken down into two pyruvates which can be used for later mechanism (Krebs cycle) to produce energy. The oxidation of long-chain fatty acid to acetyl-CoA is a central energy-yielding pathway in many organisms. Its opposite process is anabolism, which combines small molecules into larger molecules. Energy that is released from catabolism will store as ATP within the cell. The cell will then use this source of energy for synthesizing cell components from simple precursors, for the mechanical work of contraction and motion, and for transport of substances across its membrane. Catabolism maintains the chemical energy needed in order to help the cell grow and develop. Some waste products caused by catabolism are carbon dioxide, urea, and lactic acid. Heat is also sometimes released as a by product because these are oxidation processes. Examples of catabolism are the citric acid cycle.
The energy cells contain is liberated through two distinct processes: glycolysis and cellular respiration
GLYCOLYSIS Glycolysis is a series of reactions that break down glucose into two smaller organic molecules.
Glycolytic pathway Glycolysis occurs in the cytoplasm, in the presence or absence of oxygen. The pathway has several steps to convert six-carbon glucose into two molecules of three-carbon pyruvates. The direct generation of ATP from ADP and Pi is known as substrate-level phosphorylation. NAD+ is reduced to NADH twice, so we start with two molecules of NAD+ and finish with two molecule of NADH.
The net reaction for glycolysis is: Glucose + 2 ADP + 2Pi + 2 NAD+ → 2 Pyruvate + 2 ATP + 2 NADH + 2H+ + 2H2O
Most of the chemical energy extracted from the sum is still stored in pyruvate’s bonds. Pyruvate has two potential fates based on the character of the cell’s environment. In aerobic organisms, pyruvate undergoes further oxidation through the mitochondrial electron transport chain. In anaerobic organisms, pyruvate undergoes an oxygen-free process called fermentation. Some cells are obligate aerobes or anaerobes, meaning that they require that designated environment. Others are facultative; they refer one environment over the other but can survive in either.
Fermentation In the glycolysis reaction equation, NAD+ is a necessary reagent and must be present for glycolysis to occur. At the end of glycolysis, the coenzyme is present only in its reduced form, NADH. One way to regenerate NAD+ is through oxidation in the electron transport chain, but anaerobic organisms would not participate in that. So another method is used: fermentation. Fermentation reduces pyruvate to either ethanol or lactic acid.
Alcohol Fermentation This process occurs in yeast and some bacteria. Pyruvate is first decarboxylated to acetaldehyde, which is then reduced by NADH to ethanol, thereby regenerating the NAD+ Pyruvate (3C) → CO2 + Acetaldehyde (2C) Acetaldehyde + NADH + H+ → Ethanol (2C) + NAD+
Lactic acid Fermentation This process occurs in some fungi and bacteria, as well as in mammalian muscles when oxygen demand exceeds supply. Basically, many glucose molecules are put through glycolysis, yielding twice as many molecules of cellular respiration, so it builds up. Concurrently, NADH builds up, depleting cells’ supply of NAD+. To keep muscles working, pyruvate is reduced to lactic acid, and NADH is oxidized back to NAD+. Lactic acid decreases the local pH, which we fell as the burn and fatigue effects of strenuous exercise. Once oxygen supply catches up to demand, the lactic acid may be converted back to pyruvate in the process known as the Cori cycle. The amount of oxygen necessary to do this is known as the oxygen debt. Pyruvate (3C) + NADH + H+ → Lactic Acid + NAD+
CELLULAR RESPIRATION
It is the most efficient means of glucose catabolism, generating approximately 36 to 38 ATP per molecule of glucose. Respiration is aptly named; it is an aerobic process using an electron transport chain, with oxygen being the final electron acceptor.
There are three key phases: pyruvate decarboxylation, the citric acid cycle, and the electron transport chain. A productive way to keep track of these reactions will be the follow the carbon.
Pyruvate decarboxylation The first step in aerobic respiration is pyruvate decarboxylation. This step itself does not require oxygen, but it only occurs once the cell commits to aerobic respiration—and the commitment is made only in the presence of oxygen. Pyruvate is transported from the cytoplasm into the mitochondrial matrix, where it is decarboxylated. The remaining acetyl (2C) group is bound to a coenzyme A molecule to form acetyl-CoA. Once NAD+ is reduced to NADH per pyruvate; in other words, two NAD+ molecules are reduced per molecule of glucose. Acetyl-CoA is a key intermediate in the utilization of fat, protein, and other carbohydrate energy reserves.
2 Pryuvate (3C) + 2 CoA + 2 NAD+ → 2 NADH + 2 Acetyl-CoA (2C) + 2 CO2 (1C)
Citric Acid Cycle The citric acid cycle starts with the combination of acetyl-CoA (2C) and oxaloactate (4C) to generate citrate (6C). Through a series of eight reactions, two CO2 molecules are released, and oxaloacetate is regenerated. The citric acid cycle does not directly generate much energy. Each turn of the cycle generates one ATP via substrate-level phosphorylation and a GTP intermediate, for a total of two pyruvates per glucose molecule. The value of the citric acid cycle is its ability to generate high-energy electrons that are carried by NADH and FADH2. For each molecule of acetyl-CoA that enters the cycle, three NADH and one FADH2 are produced by two to account for the fact that the cycle turns twice per molecule of glucose. These enzymes then transport the electrons to the electron transport chain on the inner mitochondrial membrane, where more ATP is produced via oxidative phosphorylation. At the end of the citric acid cycle, oxaloacetate is regenerated in anticipation of the next round.
The overall reaction is: 2 Acetyl-CoA + 6 NAD+ + 2 FAD + 2 GDP + 2 Pi + 4 H2O → 4 CO2 + 6 NADH + 2 FADH2 + 2 ATP + 4 H+ + 2 CoA Structural Biochemistry/Lipids/Fatty Acids
