Structural Biochemistry/Carbohydrates
Carbohydrates are important macromolecules that consist of carbon, hydrogen, and oxygen. They are organic compounds organized in the form of aldehydes or ketones with multiple hydroxyl groups coming off the carbon chain. Carbohydrates are the most abundant organic compounds in living organisms and account for one of the four major biomolecular classes including proteins, lipids, and nucleic acids. They originate as products from carbon dioxide and water by photosynthesis,
nCO2 + nH2O (+ reducing agents and energy from photon [sunlight]) → ADP + CnH2nOn + nO2
where ADP (Adenosine diphosphate) is a product that can be synthesized to form ATP (Adenosine-5'-triphosphate) - a form of chemical energy used in cells which acts as a fuel of metabolism in plants and animals - through aerobic cellular respiration,
CnH2nOn + nO2 + ADP + Phosphate (+ oxidizing agent and energy from photon [through electrochemical gradient]) → ATP + nCO2 + nH2O
Function of Carbohydrates
[edit | edit source]Carbohydrates play a variety of extensive roles in all forms of life:
The general empirical structure for carbohydrates is (CH2O)n. Monosaccharides, which are simple sugars that serve as fuel molecules as well as fundamental constituents of living organisms, are the simplest carbohydrates, and are required as energy sources. The most commonly known ones are perhaps glucose and fructose.
Carbohydrates exist in a variety of isomer forms. Those that differ in arrangements of atoms are known as constitutional isomers, such as glyceradehyde and dihydroxyacetone. Stereoisomers have the same attachments of the atoms, but different in spatial arrangements, which can be further separated into two types: diastereoisomers and enantiomers. Diastereoisomers are the molecules that are not mirror images of each other and enantiomers exists as nonsuperimposable mirror images. The fact that monosacharides can possess up to three different asymmetric carbon atoms contributes significantly to the wide variety of isomer formations.
* Serve as energy stores, fuels, and metabolic intermediates.
* Formation of the structural framework of RNA and DNA (ribonucleic acid and deoxyribonucleic acid).
* Are structural elements in the cell walls of bacteria (peptidoglycan or murein), plants (cellulose) and animals (chitin).
* They are linked to many proteins and lipids. Such linked carbohydrates are important in cell-cell communication and in interactions between cells and other elements in the cellular environment.
* As "food" for energy supply (starch, glycogen, dextrans) and production of fats.
For carbohydrate derivatives, amino sugar is of amine group in place of hydroxyl group; deoxy sugar is of hydroxyl group replaced by hydrogen, and acidic sugar is of carboxylic acid group.
Microbial Carbohydrates
[edit | edit source]- In a detailed article, written by Hazel M. Holden, Paul D. Cook, and James B. Thoden, recent studies on carbohydrates were revealed and intensively discoveries on their three-dimensional structures were mentioned. Carbohydrates make up 50% of the Earth’s biomass and are known for their functions as energy storage, cell wall structure in plants, and other functions such as fertilization and cell adhesion. Current studies are being directed at new sugars provided by microbes and bacteria that possess chemical importance. Some microbial sugars are the dideoxysugars and trideoxysugars, made by bacteria, fungi, and plants. Some are found in Gram-negative bacteria or erythromycin. Scientists believe that these microbial sugars have unique functions in chemistry and thus, studies have been done looking at their three-dimensional structures. According to recent studies, sugars that are produced by microbes are made from glucose-6-phosphate or fructose-6-phosphate. Deoxysugars are made by modifications from acetylation, amination, epimerization, and methylation reactions. It was discovered that these sugars are extracted from prokaryotic sources, and they themselves carry biological importance in eukaryotic systems.
- The production of unique microbial sugars began with the formation of NDP-4-keto-6-deoxyglucose. This sugar is formed via biosynthesis pathways in which alpha-D-glucose-1-phosphate is attached to an NMP moiety through nuleotidyltransferase, followed by the removal of the C-6’ hydroxyl group as well as the oxidation of C-4’ hydroxyl moiety. With the formation of NDP-4-keto-6-deoxyglucose, important enzymatic reactions take place such as acetylations, isomerizations, dehydrations, epimerizations, etc. These reactions are responsible for the production of unique biosynthesized microbial sugars.
- There are two enzymes that are being investigated for their functions in catalyzing N-acetylations and PLP-dependent aminations. N-acetylransferases are ezymes that catalyze the acetyl transfer from acetyl-CoA to primary amine acceptors. The family members of this particular enzymes are being studied rigorously by scholars and scientists. One of the important family members is LbetaH, responsible in the biosynthesis of lipid A via O-aceyltransferase. Three-dimensional studies have revealed that the structure of Lipid A molecules shows the “left-handed crossover connections and its isoleucine rich hexapeptide motif”. Another important enzyme in this family is PglD, in the CoA complex. This enzyme is a trimer with two domains for each of its subunit. The base of PglD structure was discovered to be His 125. This base is the critical requirement for the catalysis of nucleotide-linked sugars. OdtC is another enzyme that catalyzes the last step in the making of Quip3NAc. This sugar is found in some Gram-negative bacteria . Like PglD, QdtC is a trimer but each of the subunit is engaged in active interactions with other monomers. The significance in the studies of these two enzymes lies in the fact that these two enzymes have very similar structures but surprisingly different biological functions. Detailed structural analysis reveals that enzymes can be very similar structures and possess entirely different activities.n
- Another enzyme that functions in the enzymatic reactions, started by the production of NDP-4-keto-6-deoxyglucose, is PLP-dependent aminotransferases and dehydratases. The name of this enzyme is derived from the fact that for the amino group to be successfully transferred to the sugar, a pyridoxal 5’-phosphate (PLP) is required. Thus, the aminotransferase is PLP-dependent and it is called PLP-dependent aminotransferase. The first sugar to perform this function was ArnB. The sugar catalyzes the amino group transfer of an UDP-linked sugar to produce UDP-4-amino-4-deoxy-L-arabinose. The amination performed by ArnB is discovered to be a “ping-pong mechanism”, with the “ping” being the formation of PMP, and “pong” being its reaction with ketons to form “transaminated products”. In PLP reactions, the -amino group of glutamate acid attacks the C4’ of PLP shifts the position of lysine. As a result, the external aldimine collapses and form -ketoglutarate and PMP. The formation of PMP gave way to a series of reactions between PMP and ketose forms of carbohydrates to produce products via amino group transfer.
- Within the family of PLP-dependent aminotransferase, PseC has been studied because its structures are believed by researchers to serve significant biological roles. They believe that PseC can provide a first look at an aminotransferase with a nucleotide-linked sugar as a substrate. From recent studies, PseC is revealed to be involved in the biosynthesis of pseudaminic acid and it also catalyzes the amino group transfer of the C-4’ position of carbohydrates. Another important enzyme is ColD, ColD can transform a sugar via dehydration to remove the 3’ hydroxyl group. GDP-perosamine synthases is also fascinating in that it uses the same substrate as ColD but instead of performing dehydration to remove a hydroxyl group, GDP-perosamine synthase catalyzes the amination at the C4’ position.
- The production of unique microbial sugars began with the formation of NDP-4-keto-6-deoxyglucose. This sugar is formed via biosynthesis pathways in which alpha-D-glucose-1-phosphate is attached to an NMP moiety through nuleotidyltransferase, followed by the removal of the C-6’ hydroxyl group as well as the oxidation of C-4’ hydroxyl moiety. With the formation of NDP-4-keto-6-deoxyglucose, important enzymatic reactions take place such as acetylations, isomerizations, dehydrations, epimerizations, etc. These reactions are responsible for the production of unique biosynthesized microbial sugars.
- Overall, many enzymes were studied by scientists because of their importance in enzymatic reactions that yield the biosynthesis of sugars in bacteria and microbes. Because of the wide varieties found in enzymes, scientists seek to study and explain the formation of unique microbial sugars.
Below is an image of Salmonella typhimurium from which ArnB is found. ArnB is known to be the first sugar that modifies the aminotransferase structure.

Categorization of Carbohydrates: 3 types
[edit | edit source]1. Mono saccharides
2. Disaccharides
3. Polysaccharides
Monosaccharides
[edit | edit source]Monosaccharides (C-H2O)n, n > 3, are the simplest form of carbohydrates. They may be subcategorized as aldoses or ketoses, if the molecule contains an aldehyde or ketone functional groups respectively. Aldoses have a carbonyl group in the form of an aldehyde on the end of the carbon chain and ketoses have a ketone group somewhere along the sugar backbone. The simplest ketose is dihydroxyacetone (has no chiral centers) while the simplest aldose is glyceraldehyde, which can be found as either the L or D enantiomer. Monosaccharides may be further classified based on the number of carbon atoms as triose (n=3), tetrose (n=4), pentose (n=5), hexose (n=6), heptose (n=7), etc. For example, glucose is known as a aldohexose (a six carbon aldose) and ribose is considered an aldopentose (a five carbon aldose). Sugars have the ability to form cyclic compounds, typically five or six member heterocyclic rings. In addition, sugars can be reducing or non-reducing depending on whether a sugar has a carbonyl moiety in its linear form. This means that the sugar in cyclic form is either in hemiacetal or hemiketal form. Monosaccharides aid in the development of nucleic acids. They are often represented by a Fischer Projection. L and D represent the absolute configuration of the asymmetric carbon farthest away from the ketone or aldehyde group on the monosaccharide. Biological carbohydrate is D sugar mostly. Note that the D and L designations are not correlated with the D and L designations for specific rotation (dextro and levorotary molecules). In addition, each carbon atom bonded to a hydroxyl group except for the first and last ones is a stereocenter of R or S, potentially forming many isomers for the same chemical formula of saccharide. In cyclic form, sugars can be described using Haworth projections.
Monosaccharides are classified as α or β anomers in their cyclic forms where the carbon atom of the carbonyl oxygen is called the anomeric carbon atom. Because the carbonyl carbon is sp2 hybridized in the open chain form, when the sugar cyclizes, the carbonyl carbon forms a new chiral center; the α anomer of glucose has the hydroxyl group attached to anomeric carbon in a trans position relative to heteroatom oxygen. The β anomer occurs when the hydroxyl group is on the same side or in cis position as the heteroatom oxygen. Both anomers are in equilibrium and are constantly changing between the ring and straight-chain conformations.
Anomeric carbon is defined as hemiacetal or hemiketal carbon for which results in different forms of stereoisomer for the saccharide. The anomeric position is easily oxidized and can form glycosidic linkages readily.
Hemiacetals/Hemiaketals, Intramolecular Cyclization and Function
Hemiacetals and hemiaketals are formed under acidic conditions when an aldehyde or ketone reacts with an alcohol, respectively. The acidic condition protonates the carbonyl carbon on the aldehyde or ketone, followed by a nucleophilic attack of the hydroxyl group on the alcohol on the carbonyl carbon (electrophile). In monosaccharides, their physical structure can exist either as an open chain or a cyclized ring. The ring structure is more energetically stable and is more common in the case of glucose, fructose and ribose. There are two possible ring formations for these sugars, known as pyranose and furanose formations. Pyranose rings are formed from aldoses (aldehyde sugars) and furanose rings are formed from ketoses (ketone sugars). Pyranoses are 6 membered rings that resemble the structure of a pyran, whereas furanoses are 5 membered rings that resemble a furan. In hexoses, for example, the C-5 hydroxyl group acts as the alcohol (nucleophile) that attacks the C-1 aldehyde carbonyl carbon (electrophile). This results in an intramolecular hemiacetal at the C-1 position. To form a intramolecular hemiaketal, the C-5 hydroxyl group of a ketohexose attacks the C-2 keto group to form the 5 membered ring, furanose.

It is possible for pentoses and hexoses to cyclize. In fact, many 5- or 6-carbon sugars, such as glucose, ribose, and fructose, exist predominantly as cyclic molecules. The carbonyl group in ketoses and aldoses reacts with an alcohol group within the same sugar molecule. This intramolecular reaction produces either an intramolecular hemiacetal or hemiketal. If the resulting cyclic sugar is a 5-membered ring, it is a furanose. If the resulting cyclic sugar is a 6-membered ring, it is a pyranose. Only the 5-membered and the 6-membered ring can be formed because they are the only types of ring formations that are energetically favored. It is important to note that when sugar undergoes a cyclic transformation, an anomeric carbon is formed in the process. An anomeric carbon is the hemiacetal or hemiketal carbon where there can be two different configuration in cyclic sugar. It can either be α or β depending on the relative position of the -CH2OH group and the -OH group on the anomeric carbon. α refers to the two groups being on opposite sides, and β refers to the two group being on the same side.
The cyclic sugar has conformational isomers. When carbon 2 is out of the plane it is called C-2 endo. Also it has chair and boat conformations. Due to steric hindrance in boat form, chair form is more favorable.
Overall the sugar has many stereochemistry such as enantiomers (D and L), Diasteromers, Anomers (alpha and beta) and molecules with same stereochemical configuration but differ in 3D conformation (chair and boat).

Structure of monosaccharides
[edit | edit source]Structure of monosaccharides can be drawn either as
1. The Fisher Projection, which is in straight chain perspective,
The Fischer projection of D-glucose is shown above.

2. The Haworth Projection, which displays molecules as cyclic or planar with substitutes above or below the ring.
The Haworth projection of α-D-glucopyranose is shown above.
3. In heterocyclic compounds, five-membered and six-membered rings are favored over others due less steric strain of the cyclic structure. Thus, furanoses (pentoses) and pyranoses (hexoses) rings are the predominant cyclic structures in monosaccharides. The rings of monosaccarides resemble the molecules furan and pyran, thus the names furanoses and pyranoses.
Example (pyran is left ring and furan is right ring):

Of straight-chain monosaccharide, aldehyde or ketone group react with other hydroxyl group, for which oxygen is bonded between the two carbon atom, to form a hemiacetal or hemiketal, which forms a heterocyclic ring; the five-atom furanose and six-atom pyranose forms are in equilibrium with their straight-chain structures.
The pyranose ring can take on different conformations. Either of the chair or boat form. The chair form allows for substituents to be in two positions, either axial or equatorial. Substituents in the axial position create steric hindrance, thus larger substituents normally are in the equatorial position, due to less crowding. The boat form is disfavored due to steric hindrances. The furanose ring can take on the form of a envelope. It has four nearly coplanar atoms with a puckered fifth atom.
Basic formation of a ring structure from the Fischer Projection
1. Rotate the substituents from last carbon in a way so that the hydroxyl group is connected by a vertical line.
2. All the substituents on the right hand side of the spine, connected to their carbons by a horizontal line, will be located at the bottom of the ring.
3. All the substituents on the left hand side of the spine, connect to their carbons by a horizontal line, will be located on top of the ring.
4. Connect the anomeric carbon to the hydroxyl group connected to the last carbon in a clockwise fashion.
5. The ring structure is normally drawn as a pyranose (six member ring) or furanose (five member ring), in a Haworth Projection (side plane view).
Types of Monosaccharides
[edit | edit source]Trioses (n=3 where n refers to the number of carbons that make up the skeleton of the sugar) are the smallest forms of monosaccharides: Dihydroxyacetone (a ketose), Glyceraldehyde (an aldose)
- Examples of monosaccharides:
- Glucose (the most common sugar)-Glucose is Glucose is a polyhydroxyaldehyde. Emil Fischer, a carbohydrate chemist received the Nobel Prize for his proof of the structure of glucose.
- Fructose (sweetens fruit)- Fructose is a polyhydroxyketone.
- Galactose (sugar in milk)-Galactose is the C4 epimer of glucose. Like glucose, galactose mutarotates when it dissolves in water.
- Ribose (component of RNA)-The C2 epimer of arabinose, is most stable in its furanose form. Each ribose unit carries a heterocyclic base that provides part of the information needed to specify a particular amino acid in protein synthesis.
- Examples of monosaccharides:
***Deoxyribose (component of DNA)-The principal difference between RNA and DNA is the presence of o-2-deoxyribose as the sugar in DNA instead of the o-ribose found.
in RNA.
-
D-ribose
-
D-fructose
-
Fructofuranose
-
Glucopyranose
Disaccharides
[edit | edit source]Disaccharides, or chemical formula of Cn(H2O)n-1, n > 5, are namely two monosaccharides linked by condensation or dehydration synthesis. As a result, a glycosidic bond is formed when the -OH of one sugar molecule joins with that of another sugar molecule. The glycosidic bond can be broken by hydrolysis, which is the addition of the water molecule, to form two monosaccharides.
- Examples of Disaccharides:
- Lactose (sugar in milk) - 1 galactose + 1 glucose
- Examples of Disaccharides:
Lactose consists of galactose bonded to glucose by a β-1,4-glycosidic linkage. Lactase is the enzyme in humans that hydrolyzes lactose back to its two monosaccharides.
- Maltose (malt sugar) - 1 glucose + 1 glucose
Maltose is made from two glucose molecules bonded by a α-1,4-glycosidic linkage. Maltase is the enzyme that hydrolyzes maltose.
- Cellobiose - 1 glucose + 1 glucose
Cellobiose is also made from two glucose molecules. Unlike maltose it is bonded by a β-1,4-glycosidic linkage, making it a non-digestable sugar due to the equatorial linkage. Cellobiose is the disaccharide of cellulose and a reducing sugar because it has hemiacetal.
- Sucrose (table sugar)- 1 glucose + 1 fructose
Sucrose is formed from a glucose and a fructose. They form a glycosidic linkage between the α glucose and β fructose. Since they are linked at their anomeric carbons, sucrose is not a reducing sugar, because neither glucose or fructose can be changed to a aldehyde or ketone.
Glycosidic bond
[edit | edit source]In general, a glycosidic bond is the covalent bond between two monosaccharides to form a disaccharide. It is formed between the hemiacetal group of anomeric carbon of a carbohydrate in acid by a condensation reaction to a hydroxyl group of an organic compound. The condensation reaction causes the formation of one additional water molecule. This results in the formation of an acetal group on the anomeric carbon, which stabilizes the bond. Glycosidic bonds are notably stable, but they can be broken chemically by strong aqueous acids. There are two types of glycosidic bonds, α and β glycosidic bonds. A substance with a glycosidic bond is called a glycoside.
For saccharides; if they have anomeric carbons, they can form glycosidic bonds to form cyclic structures and not linear forms. The chemical bonds is accounted as O-glycosidic bonds (type II linkage), for which, the anomeric carbon is bonded by oxygen to other group. O-glycosidic bond creates a covalent linkage between a serine or threonine hydroxyl side chain amide and sugar. N-glycosidic bond, type I linkage, is a type of carbohydrate-protein covalent linkage between an asparagine side chain amide and a sugar. N-glycosidic bond involves the bonding of the anomeric carbon of a sugar and the nitrogen atom of an amine group.
In the formation of a glycosidic bond, the hemiacetal group of carbohydrates (which contains the anomeric carbon) forms the glycosidic bond in the presence of an acid. Fischer glycosidation is a result from this condensation, which releases a water from the previous reaction.
Though Glycosidic bonds are relatively stable under most conditions, they are easily broken under many aqueous acidic conditions. There are enzymes called glycoside hydrolases that also break the bonds, but they are only able to break either the alpha or beta conformation, not both. The following image shows an example of a reaction that generates glycosidic bonds. http://en.wikipedia.org/wiki/File:Ethyl-glucoside.png
The stereochemistry of the bond is based on the position of the stereocenter furthest away from the C1. The alpha conformation of the glycosidic bond creates an axial conformation, whereas the beta conformation develops an equatorial conformation.
Oligosaccharides
[edit | edit source]Oligosacchardies are polysaccharides with a smaller number of sugar molecules; disaccharides bonded to simpler polysaccharides, for which, n=3 or 4 simple sugar units. Since almost all the screted and membrane-associated proteins of eukaryotic cells are glycosylated, Oligosaccharides are covalently attached to proteins by either N-glycosidic or O-glycosidic bonds.
There are two main types of Oligosaccharides:
- N-linked Oligosaccharide: In N-linked oligosaccharides, GlcNAc is variably β-linked to the amide N of an Asn residue in the sequence Asn-X-Thr or Asn-X-Ser, where X is amino acid except possibly Pro or Asp. There is enormous diversity among the oligosaccharides of N-linked glycoproteins. In some glycoproteins, processing if brief, leaving "high-mannose" oligosaccharides. In some other glycoproteins, extensive processing generates large oligosaccharides containing several kinds of sugar residues. N linked oligosaccharides have a common pentasaccharide core which contains 3 mannose and 2 N acetylglucosamine residue.
Proteins containing N-linked oligosaccharides typically are glycosylated and then processed as elucidated, in large part:
1. An oligosaccharide containing 9 mannose residues, 3 glucose residues, and 2 GlcNAc residued is attached to the Asn of a growing polypeptide chain that is synthesized by a ribosome associated with the endoplasmic reticulum.
2. Some of the sugars are removed during the process, which begins in the lumen of the endoplasmic reticulum and continues in the Golgi apparatus.
3. Additional monosaccharide residues, including GlcNAc, galactose, fucose, and sialic acid are added by the action of specific glycosyl-transferases in the Golgi apparatus.
- O-linked Oligosaccharide: The most common O-glycosidic attachment involves the disaccharide core β-galactosyl-(1->3)-α-N-acetylgalactosamine linked to the OH group of either Ser or Thr. O-linked Oligosaccharides are synthesized in the Golgi apparatus by the serial addition of monnosaccharide units to a completed polypeptide chain. Synthesis starts with the transfer of GalNAc to a Ser and Thr residue on the polypeptide. N-linked oligosaccharides are transferred to an Asn in a specific amino acid sequence, but O-glycosulated Ser and Thr residues are not members of any common sequence. Instead, the locations of glycosylation sites are specified only by the secondary or tertiary structure of the polypeptide. O-glycosylation continues with stepwise addition of sugars by the corresponding glycosyltransferases.
Endoplasmic Reticulum (3) = N-linked Golgi complex (8)= O-linked and N-linked
- Sequencing of Oligosaccharides: To determine the structures of oligosaccharide and their points of attachment, Berg et al.,[1] states that the first step is to detach the oligosaccharides from the protein and then the oligosaccharides will be isolated and analyzed. Berg et al. state that mass spectrometric techniques can provide the mass of an oligosaccharide fragment. An example given by Berg et al. is the release of N-linked oligosaccharide from proteins by an enzyme such as a peptide N-glycosidase F. The N-glycosidase F cleaves the N-glycosidic bonds linking the oligosaccharide to the protein.


Polysaccharides
[edit | edit source]Polysaccharides (also called Glycans), the chemical formula is written as Cn(H2O)n-1, for which 200 < n < 2500, or (C6H10O5)n, 40<n<3000, are polymers or multiple units of monosaccharides. They are constructed by glycosidic bonds and make ideal storage products due to their structure and solubility. Oligosacchardies are polysaccharides with smaller number of sugar molecules; disaccharides bonded to simpler polysaccharides, for which, n=3 or 4 simple sugar units. Oligosaccharides are covalently attached to proteins by either N-glycosidic or O-glycosidic bonds. Polysaccharides can be further classified into homo-polysaccharides and hetero-polysaccharides based on the composition of individual monosaccharides. In addition, branched and unbranched polysaccharides are formed by glycosidic bonds on different Carbons of the monosaccharides.
Homo-polysaccharides are composed of one type of monosaccharids. They can be branched or unbranched. Hetero-polysaccharides can be composed of two or more types of monosaccharides in straight or branched chains. Example of Homopolysaccharides: 1) some are storage of energy (starch, glycogen, and dextrans). 2) Others have structural roles (cellulose, chitin).
Examples of polysaccharides:
- Cellulose
Cellulose [(C6H10O5)n] is a homo-polysaccharide that serves a significant structural role in plant cell walls. Cotton fibers and the wood of trees and bushes, for instance, are largely composed of cellulose. Glucose residues in cellulose are linked by β-1,4-glycosidic bonds in a linear fashion. Intermolecular and intramolecular hydrogen bonds allow the more hydrophobic part of cellulose to stack, leading to complete insolubility of cellulose in normal aqueous solution.
- Starch
Starch is a polymer of glucose, and is constructed by glycopyranose units that are bonded by α-glycosidic bonds. Starch is a homo-polysaccharide that serves as a major source of energy storage in plants and also nourishment to most animals, including humans. Amylose and amylopectin are examples of starch carbohydrates. Amylose are linear chains linked with thousands of glucose residues by α-glycosidic bonds, whereas Amylopectin are branched chains of polysaccharides. The linear binding of amylose are α-1,4-glycosidic linkages. The branching chains of Amylopectin are α-1,6-linkages. The branching in Amylopectin is less than in glycogen. These branchings occur about once for every thirty α-1,4-linkages. The polymer of glucose is not soluble in water.
- Glycogen
Glycogen, also known as "animal starch", is the storage of glucose as a source of energy to animal cells. Its structure is similar to that of the amylopectin and has even more branches. Glycogen is made up of chains of glucopyranose units connected by α-1,4-glycosidic bonds and the branching of the polymer is due to α-1,6-glycosidic bonds. These branchings occur once for every ten 1,4-linkages. Glycogen is primarily produced in the liver and muscles cells.
- Chitin
Chitin is a homopolysaccharide made from repeating units of a derivative of glucose, N-Acetylglucosamine. These units are connected linearly through β-1,4-linkages. Chitin is a very important structural component making up the cell walls of fungi, the exoskeletons of arthropods and insects, and other such components. Its structure and linkages are similar to that of cellulose, except that the hydroxyl group on the 2' carbon of glucose is replaced by an acetylamine group.
Structural Differences
The linkage by α-1,4-glycosidic bonds in starch and glycogen give them a very different structure than cellulose(linked by β-1,4-glycosidic bonds). The α-1,4-glycosidic linkage results in a hollow helical structure that is more suitable for energy storage, whereas the β-1-4-glycosidic bonds allows cellulose to form linear chains that are stabilized by hydrogen-bonding with adjacent chains to form tensile fibers.
-
Cellulose
-
Starch
-
Glycogen
-
Chitin
Types of Sugars
[edit | edit source]Sugars may also be classified as either reducing or nonreducing sugars.

Reducing Sugars
[edit | edit source]
Reducing sugars have access to their open chain form. Reducing sugars are basically sugars with an aldehyde group (in their open form) or a hemiacetal group (in their ring form)at the anomeric carbon that is ready to oxidize. In other words, reducing sugars allow for chain formation and elongation. The presence of reducing sugars can be tested using Fehling's solution, a solution of Cu2+ ions that readily oxidizes free aldehydes or ketones. A similar test can be done with Benedict's reagent, which also contains the critical Cu2+ ion. A characteristic of a reducing sugars is the β-glycosidic bond, where the β anomeric carbon is involved in the linkage. Examples of reducing sugars are glucose, maltose, and lactose. Reducing sugars can form aldehyde or ketone groups under basic conditions as the carboxyl group is reduced to carbonyl group of aldehyde or ketone group and occurs when the anomeric carbon is not bonded to hemiacetal or hemiketal hydroxyl group.
It has only been mentioned that aldoses can form reducing sugars. What about ketoses? Mechanistically, when ketoses isomerize to their ring form, acetals are formed. So no, ketoses do not form reducing sugars. However, ketoses can tautomerize to aldoses, where a hemiacetal can then be formed upon ring closure.[2]
Nonreducing sugars
[edit | edit source]
Nonreducing sugars do not have access to free aldehyde or ketone such as glycosides. These type of sugar is basically an acetal (in its ring form) at the anomeric carbon. Since the anomeric carbon is fixed in a glycosidic linkage, the sugar chain cannot form or be elongated. A non-reducing sugar can also be identified by an α-glycosidic bond in which an α anomeric carbon is involved in the linkage. An example of a nonreducing sugar is sucrose.
Glycoproteins
[edit | edit source]- Structure
Glycoproteins, namely organic compounds of a mixture of carbohydrates and proteins bonded by covalent bonds, are formed in the process of glycosylation. Carbohydrate groups may be covalently bonded to a protein to form a glycoprotein, which is an important part of the cell membrane in such processes as cell adhesion and the binding of sperm to eggs in fertilization. In glycoproteins, the carbohydrate weight percentage is much less than the weight of carbohydrates found in proteoglycans. The sugar group of glycoproteins help protein folding and increase protein stability. Sugars within glycoproteins are found attached either to the amide nitrogen atom side chain of asparagine by N-linked glycosidic bond or to the oxygen atom in the side chain of serine or threonine by O-linked glycosidic bond. The bond formed between carbohydrate and protein is by bonding to one of four amino acids of asparagine, hydroxylysine, serine, or threonine. Thus protein glycosylation sites can be predicted because an asparagine can only be glycosylated when found in the form: Asn-X-Ser, Asn-X-Thr, or Asn-X-Cys sequence, where X can be any amino acid but Proline. But not all potential sites are glycosylated. Glycosylated sites have other factors such as the cell type in which the protein is expressed and protein structure. In N-linked oligosaccharides, it has a common pentasaccharide core with three mannose and two N-acetyglucosamine residues. To form a great variety of oligosaccharide pattern, additional sugars are attached to this core. Integral membrane proteins of glycoproteins are important for interactions between one cell to the other.
- Glycosylation
Glycosylation of the extracellular part of proteins takes place in the Endoplasmic Reticulum and in the Golgi complex. The ER and the Golgi complex are organelles involved in major roles in protein trafficking/packaging. Golgi is the major sorting center of the cell. The Golgi complex alter and add onto the carbohydrate units of glycoproteins because it is the major packaging/sorting center of the cell. Protein proceed from Golgi to lysosomes, secretory granules, or plasma membrane. Ribosomes on the cytoplasmic face of the rough ER synthesize the protein that is taken into the lumen of the ER. N-linked glycosylation starts in the ER and continues into the Golgi complex. It begins with the addition of an oligosaccharide precursor made up of a chain of 14 sugar molecules. However, the O-linked glycosylation site is exclusively in the Golgi complex. Also unlike N-linked glycosylation, O-linked glycosylation has the sugar molecules added one at a time, each by a different glycotransferase enzyme. One example includes the addition of N-acetylgalactosamide (GalNAc), in the cis-Golgi area, by N-acetylgalactosamide transferase. (It should be noted that the Golgi complex is split into three areas the cis, trans, and medial). In the trans-Golgi, a galactose residue is attached to N-acetylgalacosamide by a galactosamide transferase specific to the this region of the Golgi complex. An oligosaccharide precursor that is to be attached to the amide side chain of an asparagine residue in a protein is first attached to dolichol phosphate. Dolichol phosphate is a lipid molecule found in the ER lumen and is made of about twenty isoprene units. The terminal phosphate group of dolical phosphate is the site of attachment of the oligosaccharide. With the help oligosaccharide-protein transferase, the oligosaccharide is transferred from dolichol phosphate to the asparagine molecule. Proteins from the lumen of the ER and the ER membrane are then transferred to the Golgi complex, where the carbohydrate part of the glycoprotein is altered. Since the Golgi has three areas, each with its own set of enzymes, modifications to the precursor oligosaccharide allows for a range of oligosaccaride structures to form. After the Golgi complex, proteins proceed to either lysosomes, secretory granules, or the plasma membrane, depending on the signals embedded within the amino acid sequences and the three-dimensional structures.
- Examples
Erythropoietin(EPO) is a glycoprotein hormone that stimulate the production of red blood cells. The presence of three Asn residues and one Ser residues allow oligosaccharides to link the protein at the three N-linked glycosylation and one O-linked glycosylation sites. It is secreted by the kidney. A mature EPO contains about 40% carbohydrate and by going through glycosylation it will enhance its stability of protein. When EPO does not go through glycosylation its protein is rapidly being removed from the blood and therefore contains only 10% carbohydrate.

The zona pellucida is a glycoprotein membrane, where it appears at multilaminar primary oocytes around the plasma membrane. The zona pellucida structures must initiate the acrosome reaction, in order to binds with the spermatozoa. Therefore, scientists found four zona pellucidas that are responsible binding the spermatozoa and the acrosome reaction within the mouse. The most important zona glycoprotein is the ZP3, because ZP3 is responsible for sperm binding. The sperm protein is adhering with the plasma membrane of the oocyte. In addition, the ZP3 is involved with the acrosomal reaction; this lead to the releasing the spermatozoon of the acrosomal vesicle. The ZP2 is responsible of mediating the subsequent of the sperm binding. Hence, the ZP2 and ZP3 cross linked, due to ZP1. The ZP4 is the protein that human encodes the genes. For humans, it takes five days after fertilization that the zona hatching was performed by the blastocyst. On the other hand, the zona pellucida is being replaced by the layer of trophoblastic cells, when zona pellucida is decomposes and degenerate. Overall, the zona pellucida is has a great importance on the egg death and began the fertilization.
- Sequence Oligossacrides
Oligosaccharides can be sequenced by enzymatic analysis and mass spectroscopy. It is hard to know the structure of sugars so remove sugar from glycoprotein. You will use enzyme and mass spectroscopy to find out the order of these sugars that are attached.
- Glycosylation Errors
Carbohydrate attachment to proteins is important for processing, stability, and targeting these proteins. Improper glycosylation of proteins can lead to inheritable human diseases called congenital disorders of glycosylation. An example involves I-cell disease. I-cell disease is a lysosomal storage disease. A carbohydrate marker is used for directing degradative enzymes. The lysosomes of people with I-cell disease have large inclusions of undigested glycosaminoglycans. These inclusions are present because the lysosomes of I-cell patients lack the enzyme to degrade them. However, these enzymes are present in high volumes elsewhere in the body, thus indicating incorrectly delivered enzymes in I-cell patients.
Carbohydrate-binding proteins
[edit | edit source]
It has been shown that carbohydrate-protein complexes function in cell-cell recognition processes as well as adhesion of cells to neighboring cells and the extracellular matrix. The diverse carbohydrate structures displayed on cell surfaces are well suited to serve as interaction sites between cells and their environments. A glycoprotein is formed when a carbohydrate group attaches to a protein through a covalent bond. These glycosidic bonds link carbohydrates to the amino and hydroxy side chains of asparagine and serine or threonine, respectively. An N-linkage is the bond between a carbohydrate and the nitrogen in the asparagine side chain, and an O-linkage is the bond between a carbohydrate and the oxygen of serine or threonine. An asparagine residue can accept an oligosaccharide only if the residue is part of an Asn-X-Ser or Asn-X-Thr sequence, in which X can be any amino acid, except proline. Thus, potential glycosylation sites can be detected in a proteins primary structure. Not all potential sites are glycosylated, however. Glocosylated sites depend on protein structure within the region and the cell type in which the protein is expressed. All N-linked oligosaccharides have in common a pentasaccharide core consisting of three mannose and two N-acetylglucosamine residues.
Glycoproteins play several roles in terms of the medical world. Modified carbohydrates have the ability to interfere with the interactions between carbohydrates and proteins. This leads to the inhibition of the cell–cell recognition and adhesion that is a major factor contributing to cancerous growth. Thus, these the ligands of the carbohydrate-binding proteins could potentially evolve into new forms of cancer treatment. There has been research on the development of protein serum-based cancer diagnostics.[1] Similarly, the glycoprotein hormone erythropoietin, from the kidneys, has improved the treatment for anemia by stimulating an increase in red blood cells. Also, proper glycosylation of membrane proteins by stabilizing potassium channels prevents degradation of human tissue and muscular dystrophy.
Lectins
[edit | edit source]Specific carbohydrate-binding proteins in plants and animals are lectins, which are the partners that bind carbohydrate structures and facilitate cell-cell interaction. Typical lectins contain 2+ binding sites for carbohydrates and exhibit weak interactions between the protein and carbohydrate. These multiple weak interactions sum together to form a strong linkage. Linkage of lectin is like Velcro, weak interaction but strong composite! Lectins are found in animals, plants and microorganism. Lectins also play an important role in cell recognition since modified carbohydrates and oligosaccharides have the ability to interfere with carbohydrate–protein interactions and therefore, inhibit the cell–cell recognition and adhesion processes. Lectins are exquisitely specific: they can recognize individual monosaccharides in particular linkages to other sugars in an oligosaccharide (this property also makes lectins useful laboratory tools for isolating glycoproteins and oligosaccharides). Carbohydrates on the surface of one cell bind to the binding sites of lectins on the surface of another cell.
- Importance of Carbohydrates in Cell Communication
Carbohydrates have found to be capable of containing an abundance of information as a result of the various composition and structures that are possible. These diverse compounds result from the fact that monosaccharaides contain many OH groups available for linkage. This further allows for extensive branching. Additionally, the substituent attached to the anomeric carbon can assume either an α or β configuration. The presence of these various carbohydrates on cell surfaces allows for effective cell-to-cell communication.
- C-Type Lectins

C-Type lectins are those that require a calcium ion. The calcium ion helps bind the protein and carbohydrate by interacting with the OH groups found on the carbohydrate. Calcium can also form a linkage between the carbohydrate and glutamates in the lectin. Binding is further strengthened through hydrogen bonds that form between the lectin side chains and the OH groups of the carbohydrate. Carbohydrate recognition and binding is made possible by a homologous domain consisting of 120 amino acids. These amino acids determine the specificity of carbohydrate binding.
- Examples of Lectins
Embryos are attached to the endometrium of the uterus through L-Selectin. This activates a signal to allow for implantation. E. coli are able to reside in the gastrointestinal tract by lectins that recognize carbohydrates in the intestines.
Malectin
[edit | edit source]Malectin is a carbohydrate-binding protein present in the endoplasmic reticulum of a cell. It plays important roles in the early stages of N-Glycosylation.
Selectin
[edit | edit source]Selectins are transmembrane glycoproteins that form a single chain. They mediate the attachment between leukocytes (circulating white blood cells) and the surfaces of endothecial cells (the cells that line blood vessels. Selections are members of the C-type lectins. The selectins recognize and bind specific oligosaccharides on cell-surface glycoproteins. Reciprocal selectin-oligosaccharide interactions between the two cells types allow the endothelial cells to "capture" circulating leukocytes, which then crawl past the endothelial cells on their way to eliminate the infection or help repair damaged tissues. Selectins bind specific immune cells to sites of injury and inflammation. There are various types of selectins. For example, the L-type selectin refers to lymph-node vessels, the E-type selectin refers to endothelium, and the P-type is for platelets.
Viruses
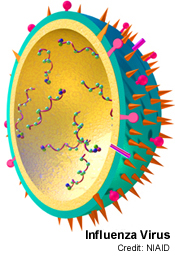
[edit | edit source]Adhesion to carbohydrates on the cell's surface grants some viruses access to the cell. Viruses can bind to certain residues on glycoproteins and once within the cell, release proteins which will break certain bonds within the glycoprotein to grant the virus free access. The influenza virus binds to sialic acid residues which is present on cell surface glycoprotein. The viral protein that's binds to the silica acid residues is called hemagglutinin. After the virus penetrates the cell membrane, another viral protein, neuraminidase (sialidase), cleaves the glycosidic bonds to the sialic acid residues, freeing the virus to infect the cell. Inhibitors of this enzyme such as Tamiflu and Relenza are important anit-influenza agents.
Diversity of Carbohydrates
[edit | edit source]Carbohydrates are very diverse due to these reasons:
1. Different monosaccharides can be joined to one another through any of several OH group.
2. The C-1 linkage can have either an alpha or a beta configuration, and extensive branching is possible.
So many different oligosaccharides can be formed from four sugars compared to oligopeptides.
Diversity as a result of carbohydrate chemical properties
Chemical structure.
Monosaccharides are naturally complex building blocks, owing to their high number of stereogenic centers. C6H12O6 (a hexose) has 16 stereoisomers. Two of these are the enantiomers D- and L-galactose (D-Gal and L-Gal). In humans, only D-Gal is present, whereas in plants both D and L forms can be found. Free monosaccharides are polyhydroxyaldehydes or polyhydroxyketones and can exist in an open form or undergo an intramolecular reaction to yield cyclic hemiacetals. The attacking OH determines the ring size, normally of six members (pyranosic ring) or, less commonly, five members (furanosic ring). As a result of cyclization, a new chiral carbon is formed and generates two possible anomers (α and β), in equilibrium with the open form. These hemiacetals can react to form acetals, fixing the stereochemistry of the anomeric carbon and generating diverse carbohydrate derivatives. For example, D-Gal is present in certain microorganisms in both pyranosic and furanosic forms, even in the same glycoconjugate.
Chemical connectivity: glycosidic bonds and branching.
Because of the different OH groups available for intermolecular acetal formation, many glycosidic linkages can potentially be formed between the anomeric carbon of one monosaccharide and any OH group in another. This reaction determines the type of linkage (α- or β-bond), which is essential for enzymatic recognition. This availability of diverse branching points is a key factor that influences carbohydrate compactness, flexibility and also physical and biochemical properties. A clear example of its relevance is the difference between starch—which is composed of amylose, a linear homopolymer of α(1,4)-glucose residues deposited in semicrystalline granules in plants—and glycogen. Glycogen is also a homopolymer of α(1,4)-glucose residues, but as there is a α(1,6) branching point present every ten units, it exists in animal cells as a noncrystalline polysaccharide.
Noncarbohydrate modifications.
Glycan structures can be modified by diverse substitutions, including sulfation, acetylation or phosphorylation.
References
[edit | edit source]- ↑ Berg J. M., Tymoczko J L., Stryer L. " Biochemistry" Sixth Edition, (c) 2007, 2002 by W. H. Freeman and Company, New York
- ↑ http://www.chem.ucalgary.ca/courses/350/Carey5th/Ch25/ch25-2-5.html
1. Berg, Jeremy M. 2007. Biochemistry. Sixth Ed. New York: W.H. Freeman. 303-325.
2. "The selectin family of carbohydrate-binding proteins: Structure and importance of carbohydrate ligands for cell adhesion". Richard D. Cummings, David F. Smith. BioEssays.Volume 14, Issue 12, Pages 849-856, 1992 Wiley Periodicals, Inc.
3. "Carbohydrate-binding proteins are becoming extremely useful in curing various illnesses", April 2004, News-Medical.Net [2]
4. Nangia-Makker, P.; Conklin, J.; Hogan, V. and Raz, A. "Carbohydrate-binding proteins in cancer, and their ligands as therapeutic agents", Trends in Molecular Medicine. Volume 8, Issue 4, 1 April 2002, Pages 187-192
5. Richards, M.R. & Lowary, T.L. Chemistry and biology of galactofuranose-containing polysaccharides. ChemBioChem 10, 1920–1938 (2009).
6. Wade, L. G. "23 Carbohydrates and Nucleic Acids." Organic Chemistry. 7th ed. Upper Saddle River, NJ: Pearson Education, 2009. 1097-141. Print.
7. Holden, Hazel M., Paul D. Cook, and James B. Thoden. “Biosynthetic enzymes of unusual microbial sugars”. Current Opinion in Structural Biology. (2010): 543-550. ScienceDirect.