Structural Biochemistry/Carbohydrates/Carbohydrate Derivatives
Single units of carbohydrates called monosaccharides may undergo various reactions to form carbohydrate derivatives. Derivatives in case of carbohydrates often means modification of sugar molecules by addition substituents other than hydroxyl group. Amino sugars, acidic sugars, deoxy sugars, sugar alcohols, glycosylamines, and sugar phosphates are examples of carbohydrate derivatives. After modification, a transformed molecule of sugar resembles structure of a sugar and an added substituent; however, it is not considered as sugar anymore because its function and characteristics has changed. For instance, sugar alcohols still have sweet taste, but are not completely absorbed by the human body and, therefore, the impact of sugar alcohols on blood sugar is less and they provide fewer calories per gram. Thus, some sugar alcohols are widely used as sugar replacement in diet and health-oriented foods especially for individuals with diabetes. Most sugar derivatives occur naturally and have important biological functions. For instance, amino sugar heparin occurs in intracellular granules of mast cells that line arterial walls and, when released, inhibits blood clotting. Glycosylamine adenosine is an important part of DNA and RNA structure. Also, due to wide application of sugar derivatives in different areas many of them are commercially synthesized.
Glucose Family
[edit | edit source]



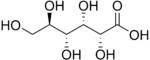
The Glucose Family are monosaccharides with functional groups replacing the hydroxyl group at the C-2 carbon or the C-6 group. Each sugar modification has a prominent effect in metabolism.
β-D-Glucose- This carbohydrate derivative plays a crucial role in metabolism. It is the main source of energy which is starts the process of cellular respiration. It is produced through photosynthesis and is often used in food products. It’s the most common carbohydrate and also circulates through the blood. It is known as blood sugar and can be converted into starch by the body. It is also used in blood tests for diabetes.
β-D-Glucose 6 Phosphate- Plays an important role in glycolysis. It is the final product after glucose is broken down and converted into the energy. The addition of the phosphate group gives the sugar a negative charge which prevents sugars from easily crossing lipid membranes. At elevated levels, it can inhibit brain hexokinase. It can be converted into starch or glycogen where it is stored in the liver and muscles. It plays an important role in blood glucose levels. Low levels can lead to Glucose-6-phosphate dehydrogenase deficiency, but the disease is hereditary. The disease leads to breakdown of red blood cells when exposed to certain environments.
β-D-Glucosamine- This is also an amino sugar that is very important in the formation of lipids and proteins. This can also form chitin in exoskeletons of insects as well as cell walls of plants. One of its common uses is for osteoarthritis. It helps rebuild cartilage and is used in veterinary medicine. Another of its uses is in helping with joint function and connective tissue. It also helps in body regulation and functions with β-D-Glucose 6 Phosphate in the body.
N-Acetyl- β-D-Glucosamine- This sugar is essential for optimal health and function in the body. It aids in cell communication. It also plays a role in how the immune system reacts with HIV and tumors. This also plays a role in osteoarthritis and helps in cartilage formation. It has been shown to play a role in nerve functioning for learning in mammals. This molecule has multiple uses such as limiting cholesterol absorption and decreases insulin secretion. Some receptors have been found in the thyroid to transport iodine proteins. It is found in multiple glands of the body and plays some role in the organ’s function. Muramic Acid-This sugar is a main component of bacteria cell walls and it is a derivative of peptidoglycan. It is used in gas chromatography for laboratory experiments.
N-Acetylmuramic Acid- This is also found in peptidoglycans of bacterial cell walls. It is a the product after N-acetylglucosamine that has been condensed with lactic acid. In bacteria, phosphophoenolpyruvate adds the lactyl group to C3 of N-acetylglucosamine. Beta-lactams compete for binding with transpeptidases, enzymes that catalyze the formation bonds between N-acetylmuramic acid-based peptide chains. Chlamydia lacks N-acetylmuramic acid in its cell walls which is the reason why penicillin does not treat the disease.
β-D-Glucuronate-This molecule is a highly polar molecule. It is used to increase solubility of some drugs. It is incorporated into proteoglycans and then combined with steroid hormones. This molecule forms to help make compounds more soluble for excretion. Bilirubin is one of the main molecules that this molecule makes soluble. This compound is very useful in clearing drugs from the body and making them soluble so that drugs can be processed through the body.
β-D-Gluconate – This is used as an alkalinizing agent for fluid therapy. It can be used in cleaning products because it dissolves mineral deposits. An alkaline solution will aid in dissolving the minerals faster. It can slowly be metabolized into bicarbonate, but the effects are longer lasting. It is an acidic sugar that occurs naturally in plants, honey, and fruits. It can be prepared through fermentation of glucose as well. When combined with calcium, it can form a gel to treat burns that are caused by hydrofluoric acid.
Amino Sugars
[edit | edit source]


Amino sugars are generally known as monosaccharide carbohydrate sugars that have replaced an -NH2 amine group with the 2'-carbon hydroxyl substituent. The most abundant amino sugar is one of the oldest and most abundant organic compounds on Earth. More than 60 amino sugars are known, many of them having been isolated and identified only recently as components of antibiotics. Examples of amino sugars include:
Galactosamine, for example, is one of eight essential amino acids that function in cell to cell interaction. Though research on galactosamine has just begun, research has shown that it may help those with joint inflammations. Lacking in galactosamine may even be one of the factors related to heart disease. Also, it may also function as a toxin leading to liver failure. In addition, galactosamine may also function as one of the sugars composing the follicle stimulating hormone (FSH) and the luteinizing hormone (LH), both of which are needed in the reproductive processes of the human body. Sources of galactosamine include bovine (which includes both cattle and oxen), red algae and shark meat.
Glucosamine, contrary to galactosamine is a very popular and relatively well-known amino sugar that our bodies use to produce glycoconjugates like glycosylated lipids and proteins. Glucosamine has a structural role in composing the hard exoskeleton of chitins, such as a variety of arachnids, crustaceans, and insects. Our bodies can obtain glucosamine from external sources such as the fermentation of different grains, like wheat and rice and barley, and like galactosamine, from bovine and shark. As far as its effect on bodily functions, glucosamine's precursor, glucosaminoglycans are major segments involved in joint cartilage, and thus can help in the treatment of osteoporosis, or osteoarthritis.
Sialic Acid is a very important sugar amine necessary to our bodies' mental and physical well being. Without sialic acid, our bodies would deteriorate in both aspects. For example, children found lacking in this particular sugar amine have been known to be adversely affected in their development, growth and in the pigmentation of their hair and skin. In a study done by Bing Wang et al., improved sialic acid concentrations in infants proved to improve their synaptogenesis and their neurological development. However, although sialic acid may serve to help us, it may also be the culprit that allows different viruses to enter. Sialic acid is normally found on the surfaces of cell membranes, it is not only responsible for fluid uptake to its negatively charged regions, but it also has the ability to recognize and bind to proteins on certain viruses, such as Hemagglutinin which is found on the cell surface of the Influenza virus. Thus, once the two bind together, it opens a door for the virus to enter into the cell and spread to other parts of the body, causing injurious repercussions.
'N-Acetyl-d-glucosamine' is the main component of the polysaccharide in chitin, the substance that makes up the tough outer skeleton of arthropods and insects.
Deoxy Sugars
[edit | edit source]


Deoxy sugars are carbohydrate derivatives that are lacking an -OH hydroxyl group at the 2'-Carbon of the sugar cyclic ring, hence "deoxy," de- being a prefix meaning "to remove," and "oxy" representing "oxygen."
Deoxyribose is the most commonly known deoxy sugar because it is the exact sugar used in the backbone of our DNA double helices. Scientists speculate that the deoxyribose sugar derivative was used as opposed to ribose because of its more stable structure. Unlike ribose, it doesn't contain a hydroxyl group at its 2'-Carbon, which would otherwise make it susceptible to hydrogen bonding with other species or molecules, thus disrupting the kind of stable matrix that our DNA needs to have. In the DNA backbone, deoxyribose sugars are bound to phosphate groups via phosphodiester linkages, and are each covalently attached to one of the four DNA nitrogenous bases. They therefore play very important roles in in the flexibility of the DNA backbone and in ensuring that the polar nitrogenous bases are facing into the double helix rather than outwards where they would be unstably exposed to hydrophobic media.
Fucose, although less talked about in our science books, is also an essential sugar derivative needed to maintain our bodies' overall well-being and proper development. Fucose is known to be excreted by nursing mothers, and in recent studies has shown to have a significant role in the fetal development of newborns, as well as to the advancement of their immune systems. Fucose and its derivatives also ensure the proper transmission of nerve signals or cell to cell communication, improve the brain's long-term memory and even play an important role in inhibiting the spread of tumor and cancerous cells.
Rhamnose is not as well known, and for a while scientists had trouble studying this sugar and understanding what its purpose was in the body. In fact, it used to be thought of as an inert deoxy sugar. It is also unusual in the sense that unlike most other sugars, its natural occurring form is its L configuration (i.e. L-Rhamnose as opposed to D-Rhamnose). Interestingly enough, Rhamnose sugar can be extracted from poison sumac. Generally, rhamnose is found mostly in the cells of plants and bacteria rather than animals. Rhamnose may participate in the body in cell proliferation, collagen synthesis and in the degradation of free radicals.
Dideoxy and Trideoxy Sugars
Dideoxy sugars and trideoxy sugars are sugars where two or three of the hydroxyl groups, respectively, are replaced with hydrogen atoms. These sugars are most commonly made by plants, fungi and bacteria. Over one hundred different sugars of these types have been isolated from prokaryotic sources alone.
Di and trideoxy sugars are found attached to the lipopolysaccharides that are embedded in the outer membrane of Gram-negative bacteria, as well as in other areas of these bacteria’s S-layer. They are also found on the S-layer of Gram-positive bacteria, which is the outer peptidoglycan membrane which is thicker in Gram-positive bacteria than in Gram-negative bacteria. In medicine, these rare sugars are used in antibiotic, antifungal, anthelmintic and antitumor agents, and are therefore quite valuable to science.
'Their Synthesis'
The enzymes concerned with their production are largely unknown, mainly because these unusual dideoxy and trideoxy sugars are not commercially available, so not much study has gone on concerning them thus far. However, the overall method of their synthesis has been observed. In the biosynthesis of these dideoxysugars and trideoxysugars, one of the most important intermediates to form is NDP-4-keto-6-deoxyglucose (where NDP stands for nucleotidyl diphosphate). This molecule serves as the branching point for hundreds of different di and trideoxy sugars. Its synthesis undergoes two basic steps (illustrated in Steps 1 and 2).
- In Step 1, α-D-glucose-1-phosphate is attached to an NMP moiety using a nucleotidyltransferase type enzyme. Which particular enzyme depends on which nucleotide is included in the reaction, as each nucleotide has a unique shape.
- In Step 2, the carbon number 6 is reduced, removing the hydroxyl to give a CH3 instead of the original CH2OH. Also, the carbon number 4 hydroxyl group is oxidized, leaving one double-bonded oxygen in its place. This step is catalyzed by another enzyme given the name NDP-glucose-4,6-dehydratase.
- Step 3 shows seven enzymatic reaction types commonly utilized to form the plethora of possible di and trideoxy sugars (see illustration). These are the reactions where many of the enzymes involved still remain unknown. Their discovery and determinations of their structures and more detailed function is the next step for biochemists in the field of dideoxy and trideoxysugars.
Acidic Sugars
[edit | edit source]

Acidic Sugars- According to Brønsted and Lowry, an acid is defined as any molecule that has the ability to donate one of its hydrogens (protons). It is this losing of a proton that makes a molecule acidic. As far as sugars go, a sugar's simplest structure is called a monosaccharide (if you think of it in SAT terms: atom is to element as monosaccharide is to sugar), and what makes a sugar acidic is the oxidation of one of its -OH groups after a hydrogen (proton) has been "lost" turning the carbon into a carbonyl carbon (carboxyl group) making an acid out of the molecule.
Some Of The Most Commonly Found Acidic Sugars
[edit | edit source]Glucuronic Acid: Glucuronic acid derives from Alpha D-glucose by means of oxidation. Mechanism can be found here [1](image drawn by author using a chemistry drawing program from https://scifinder.cas.org). The most common function of glucuronic acid is in the metabolism of foreign compounds (i.e. medication or poisons) in an organism. This molecule is extremely important to the chemists who make and design drugs as it is pertinent to know if and how a medication will be metabolized in the body. Glucuronic acid in Greek is called "sweet urine" which is a sugar that exists in urine. The acid functions to bind together toxins, such as drugs, hormones and steroids, found in the liver, helping them flush out from the body. Glucuronic acid is often used as a detoxicating agent which can help with drug overdosing and minimizing drug interaction within the body. Recent studies also show how Glucuronic Acid prevents prostate cancer in men by detoxing chemicals within the body. Ascorbic Acid: Ascorbic acid is most commonly known by the name Vitamin C (the L-enantiomer of ascorbic acid). Because of its ease in being able to oxidize, vitamin C has been used for such things as a preservative. It is also known as a cure for scurvy (a lack of vitamin C in the body). This was most common back in the day when sailors or pirates would go out on long trips on the ocean, their supply of fruit would expire and they would have no source of vitamin C and thus get scurvy. Since human bodies cannot produce vitamin C on their own, we must be able to get our vitamin C from the food and fruits that we eat. Ascorbic Acid also has antioxidant properties which can help protect nucleic acids, proteins, lipids, and other cell organelles from free radicals such as hydroxyl radicals (reactive oxygen molecules) that could else wise be damaging and tumor-inducing. The oxidized version of ascorbic acid is relatively safe, unreactive, and can be metabolized without any problems. However, an excess amount of ascorbate, which is an oxidized ascorbic acid molecule, could potentially promote and initiate instead of limiting free radical reactions within a living system.
Artificial Sweeteners
[edit | edit source]Artificial sweeteners provide the same, if not more, sweetness of the sugar without the high calories or subsequent tooth decay that accompanies sugar consumption.
Aspartame
[edit | edit source]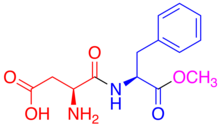
This sugar substitute, known on the market as NutraSweet (for baking purposes) or Equal, was discovered in 1965, but not approved by the FDA until 1981. It is 180 times as sweet as sucrose and contains 4 calories per gram, which is considered negligible. It is unstable in heat and decomposes in liquid during prolonged storage. However, although the aspartame may decompose in liquid, it is not hazardous for consumption. The break-down of aspartame only affects the quality of the beverage. Aspartame is a dipeptide consisting of two amino acids in an ester bond with methanol. Aspartic acid is at the N-terminus and phenylalanine is at the C-terminus where it bonds with methanol. Aspartame is metabolized in the gastrointestinal tract, where the peptide and ester bonds are broken, separating the amino acids and the methanol. The effects of the increase in methanol concentration from the breakdown of aspartame in the body was studied by Soffritti, et al. in Italy. This group studied rats and the effect of high dosages of aspartame in linkage to lymphomas and leukemia. It was found that lymphomas and leukemia increased in the female rats at dosages around the acceptable daily intake (ADI). And the levels increased in male rats only at the highest dosage which was 100 times the ADI. They also noticed that increasing the methanol intake through water increased the leukemia, as did the addition of methyl-ter-butyl ether (MTBE) which metabolizes to produce methanol. Methanol in the body turns into formaldehyde which can turn into formic acid. The study also showed that increasing the amount of formaldehyde increased the leukemia and lymphomas. While this study seems to indicate that aspartame consumption is a risky health hazard, there is much criticism for the study. For one, the group did not allow another group to examine their samples which goes against the customary procedure of verification. In addition, the FDA has criticized this lab in the past for conducting unreliable work.
While the validity of aspartame being a health hazard is being questioned, it is known for a fact that aspartame is quite hazardous to individuals with Phenylketonuria (PKU). Those with PKU cannot metabolize phenylalanine which causes a toxic amount of phenylalanine to accumulate in the body if substances that contain phenylalanine, like aspartame, are consumed.
Saccharin
[edit | edit source]
Also known as Sweet-N-Low, saccharin is one of the oldest artificial sweeteners. It is 300 times as sweet as sucrose. Saccharin was discovered at Johns Hopkins University in 1879 in the course of research on coal-tar derivatives. Its name, which comes from the Latin word for sugar, saccharin, has no structural relationship to any sugar. Saccharin itself isn't very soluble in water. However, the proton bonded to nitrogen is fairly acidic and saccharin is normally marketed as its water-soluble sodium or calcium salt. Its earliest applications were in as a replacement for sugar in the diet of diabetics before insulin became widely available. The enhanced sweetness permits less to be used and therefore reducing the cost of production. Using less carbohydrate-based sweetener also reduces the number of calories.
Sucralose
[edit | edit source]
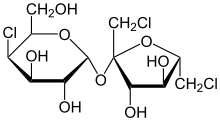
More popularly known as Splenda, sucralose is 600 times sweeter than sucrose. It is very similar to the disaccharide structure of sucrose. Three of the hydroxyl groups are simply replaced by chloride atoms. Although this change does make the molecule more likely to react with nucleophiles and could present as a danger due to the addition of chlorine, it is not a cause for concern. The levels of chlorine are no different than in comparison to consumption of table salt which is made of sodium chloride, and is harmless in moderation. Sucralose is considered calorie-free because the body does not to recognize the molecule as sugar, so it does not get broken down for energy.
References & External links
[edit | edit source]Carey, Francis A., and Neil T. Allison. "23." Organic Chemistry. 8th ed. New York: McGraw-Hill, 2011. 1049. Print. Carey, Francis A., and Robert M. Giuliano. "23.12 Amino Sugars." Organic Chemistry. 8th ed. New York: McGraw-Hill, 2011. N. pag. Print.
http://www.drugs.com/dict/d-glucose-6-phosphate.html
http://www.revolutionhealth.com/healthy-living/vitamin-index/n-acetyl-d-glucosamine-ns
http://www.glyconutrient.biz/N-AcetylGlucosamine.htm
http://themedicalbiochemistrypage.org/non-glucose-sugar-metabolism.html
Meister, K. “Sugar Substitutes and Your Health.” Comprehensive Reviews in Food Science and Food Safety, 2006.
Soffritti, M, et. al. "Aspartame Induces Lymphomas and Leukaemias in Rats" Eur. J. Oncol., vol.10, 2005.