Wikijunior:Particles/Print version
| This is the print version of Wikijunior:Particles You won't see this message or any elements not part of the book's content when you print or preview this page. |

Everything is made of matter. Almost everything, that is. There are two criteria that determine whether something is composed of matter:
- Matter must have mass;
- Matter must take up space.
As you know, air takes up space. Therefore, air is matter. In fact, if you were hot enough you could turn into gas too, just like air. That will be explained later.
Here are some other examples of matter:
- You
- Other living things
- Your schoolbag
- Your science textbook
However, not everything is made up of matter. Here are some common exceptions:
- The light that comes out of lamps
- The sound from your radio
But how does this all link to particles? As you will learn later, matter is made up of particles. Since almost everything around us is made of matter, learning about particles is essential in explaining further topics. This book will help you begin your quest by introducing the properties of particles and explaining the types of objects made up from matter. This book is based on the curriculum of chapter 6 of form one in Hong-Kong, but you don't have to be a Hong-Kong-er to read this book!
Using this book
[edit | edit source]Children can use this book in a number of ways.
- To find out more about our world and fulfill their curiosity.
- As a set of notes for studying and future reference.
- To use within the curriculum of a class.
- And pretty much anything else that you can think of.
If you are reading this book as an e-book on page Wikibooks, feel free to edit or modify this book! If you are reading this offline or as a pdf, the url of this book is http://en.wikibooks.org/wiki/Wikijunior:Particles.



As we have found out from the introduction, anything that occupies space and has mass is matter. However, not all matter is the same. Matter, in fact, can appear as three forms: liquid, solid and gas. Water is the only kind of matter whose all three states appear in nature. Therefore, we will use water as a primary example as we go through the three states of matter. Later in the book, we will also explain the three states of matter with the particle model.
Properties of matter
[edit | edit source]
The properties of matter are as follows:
- Solids' shapes cannot be altered, and nor can its volume
- Liquids' shapes can be altered, but not its volume
- Gases' shapes can be altered (so it's compressible), and so can its volume
These properties can be verified with a series of experiments. First, you can try to alter the shape of volume of solids. Try doing that to your pencil – it won't budge. Then try to pour some water from a bottle to a cup. The shape changes. Now, if the bottle is full, but its size reduces, will the liquid spill? The answer is yes. Therefore, liquids have a fixed volume. Finally, try gases. If you transfer some coloured gas from one container to another, whose shapes and sizes are different, there will be no problem.
Change of states
[edit | edit source]
Sometimes matter can change from one state to another. For example, if you blow onto the surface of glass, the water vapour that comes out of your mouth transforms into liquid. If you put the piece of glass into a freezer, a few hours later it will become ice.
There are specific names for the changes of states.
|
Solid |
Melting →→→→→→ |
Liquid |
Boiling →→→→→→ |
Gas |
| ←←←←←← Freezing |
←←←←←← Condensation |
Melting, freezing and boiling happen at fixed temperatures. The temperature of distinction between different states are called the melting point, the freezing point and the boiling point. For water, these points are 0°C, 0°C and 100°C respectively. Actually, the freezing and melting points of any matter are the same.
Condensation does not take place at a fixed temperature. There is a related process called evaporation, which does not take place at a fixed temperature, although a higher temperature does accelerate the rate of evaporation. Evaporation is the process in which a solid changes into a liquid. When your wound is sterillised, it feels cold as the alcohol evaporates, carrying with it heat.
Quiz
[edit | edit source]- X is heated, so that transforms into another state. The initial form is not liquid. What is the current form? What is the process it went through?
- True or false? As the boiling point of Y is 300°C, it cannot change into its gaseous form at 250°C, nor change into liquid form at 350°C.
Answers:
- Liquid; melting
- False. Condensation and evaporation can happen at any temperature.

The particle theory is a theory that scientists have proposed. It suggests the following points:
- Matter is made of particles;
- Particles are extremely small, and current scientific instruments cannot see them;
- Different kinds of particles have different sizes;
- There must be gaps between particles
- Particles are always in motion.
Spaces between particles
[edit | edit source]
There are spaces between particles. Imagine you put pebbles into a measuring cylinder. The reading is 200mL. Then you get 200mL of sand, and mix it in. The result is not 400mL, as there are spaces between the pebbles, and the sand can fill them up.
So, if you mix equal volumes of alcohol and water, the result is not twice the volume of alcohol or water, but slightly less. See the diagram on the right for what the particles look like. Or, if you put sugar into coffee, the coffee level does not rise up the same volume as the sugar. This is because there are spaces between particles.
Particles always moving
[edit | edit source]Particles are always moving, although they do move differently - this will be explained in the states of matter chapter later on. For example, when water is boiled, the 'steam', which are actually tiny water particles, cannot stay in one place. More about this will be discussed in the next chapter, which is about diffusion.
Quiz
[edit | edit source]- Which of the following statements is true?
- Particles in solids are often stationary. (Stationary means not moving.)
- Particles can only be seen with electronic microscopes.
- Heat is not made of particles.
- The size of alcohol and water particles are the same.
- Name the rule that is not used above, and give an example of it.
Answers:
- 3
- There are spaces between particles. When equal amounts of water and alcohol are mixed together, the result is not the same as the sum of the two initial liquids.
As we have found in the last chapter, particles are always moving. However, how exactly would they move? A common phenomenon observed from the movement of particles is diffusion. Diffusion means a kind of particles going from a place of higher concentration to a place of lower concentration. Imagine a herd of lions and a herd of tigers mixed together. If diffusion happens, lions will go to a place with more tigers than lions, and tigers will go to a place with less tigers than lions. At the end, the tigers and lions will be mixed more thoroughly. That is what particles are like during diffusion.
Examples of diffusion
[edit | edit source]
If you dip a paintbrush with a bit of red paint on the tip into a cup of water, the paint will spread all over the cup. The red will spread to places that are more transparent, while the transparency spreads to places that are redder. At the end, the paint and water will be thoroughly mixed so the water is red. (Here we assume that the paint is soluble in water.)
Another example is smell. Imagine a piece of stilton cheese is placed on a table in a corner of a room. You are standing on the opposite corner, a friend of yours is standing in the centre, and another friend is standing right next to the stilton cheese. You must be the last to smell it.
Perfume in a plastic bag
[edit | edit source]This experiment is inspired from an experiment from the following source:
- Chan, W. K.; Luk, W. Y.; Kong, S. W. "Matter as particles". Understanding Integrated Science. Vol. 1B. Aristo Educational Press LTD. p. 131–132.
- Put a drop of perfume into a plastic bag, then tie up its opening.
- Sniff. You can't smell perfume yet.
- Wait a little bit, sniff again. You should be able to smell perfume now.
This experiment proves that there are spaces between the particles of the wall of the plastic bag, and that the perfume diffuses. When the perfume on the bottom of the bag diffuses, it flies to the wall of the bag. As the perfume particles are small enough, they escape through the gaps between the plastic particles and into your nose.
Quiz
[edit | edit source]
If A and B are to meet, do you think A will come to B or B will come to A? Why?
Answer: B will come to A. This is because A is in a place of lower concentration while B is in a place of higher concentration. B will flow to A's place because of diffusion.
Apart from diffusion, there is another interesting phenomenon that can be observed. It is called the Brownian motion, discovered by a scientist called Brown.
Smoke cells and Brownian motion
[edit | edit source]Before we start on Brownian motion, let's look at an interesting experiment. A smoke cell is a small box full of smoke and air. If you have one (or your teacher gives you one), place it under a microscope. Otherwise, there is a short clip here. As you can see, the bits of smoke move at random directions, unassisted. Or do they?
Smoke particles are larger than air particles. That's why we can see the smoke particles better than the air particles. As you will learn later, gas particles always move at high speed, in random directions. The same things is happening to the air particles. When an air particle bombards a smoke particles, the smoke particle moves to the same direction as the air particle that hit it. When another air particle hits the smoke particle, it changes its direction to that of the second air particle, and so on. This is called Brownian motion.
-
What will happen to the small black particle on top and the large blue particle when they collide?
-
Their directions will change.
The story of pollen grains
[edit | edit source]
The Brownian motion was discovered by, and named after, a botanist called Robert Brown. A botanist is a person who studies plants. In 1827, he was observing some pollen grains when he saw that the pollen grains were moving mysteriously. Do you know why the pollen grains were moving like that? The movement of the water particles causes that of the pollen grains, enabling them to move in random, constantly changing directions. This is what Robert Brown later figured out.
Quiz
[edit | edit source]- Brownian motion is named after:
- Charlie Brown
- Gordon Brown
- Robert Brown
- John Brown
- Which of the statements below best describes of movements of particles in a smoke cell?
- The smoke particles flow to places of higher concentration
- The air particles flow to places of lower concentration
- The smoke particles dodge the air particles, thereby causing the smoke particles to rotate
- The air particles hit the smoke particles, thereby causing them to move in random directions
Answers:
- 3
- 4
Before we begin on particle models, let us recall a few points from the previous chapters.
- Matter is made of particles.
- Particles are always moving.
- Particles usually move from a place of higher concentration to a place of lower concentration.
Here is one more point that we have not discussed before, but is equally important:
Now that these points are learnt, there are a lot of things we can explain with the particle model. But what exactly is a particle model? A particle model is basically a model which shows particles in action. You cannot find them in real life as they cannot be seen with microscopes. They can be 3-D models or 2-D ones. They can be used to explain a lot of things, such as the three states of matter, density, and thermal expansion and contraction.

Do you still remember that we learnt about the three basic states of matter earlier? We can explain the three states of matter with the particle model. You will find out why matter is compressible, and why its shape is more stable as a solid.
Gas is compressible; however, when compressed, the gas pressure will be different. This chapter will be all about gas and air pressure. Make sure you have read the previous chapter before proceeding to this one.
Density is how much of matter there is in a fixed volume. Reading this chapter does not require much previous knowledge aside from those discussed in the 'Matter' and 'Particle theory' chapters.
As you know, hotter things grow and smaller things shrink. These are called thermal expansion and contraction. But why exactly do they happen? Can they be applied in daily life?
Quiz
[edit | edit source]Which of the following can be easily explained by the particle model, in your opinion?
- The water cycle
- Photosynthesis (the process in which plants make food)
- Comparison of density before and after dissolving
- Magnetic fields
- The movement of particles under different temperatures
- The respiratory system (the system by which you breathe)
Answer:
3 and 5.
This chapter, as mentioned in the previous, is about using the particle model to explain the three states of matter.
To explain the three states
[edit | edit source]To explain the properties of each of the states
[edit | edit source]| State | Model(s) | Flexibility of shape | Compressibility |
|---|---|---|---|
| Solid | 
|
Solids have a fixed shape; their shape cannot be altered. | Solids have a fixed volume; you cannot compress them. |
| Liquid | 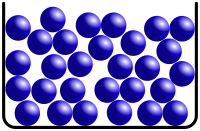
|
Liquids do not have fixed shapes. As you can see, the shape can alter depending on the shape of the container. | Liquids have a fixed volume. You can see from the model that there are the same number of liquid particles and that the size of the particles are the same. |
| Gas |  
|
Gas particles do not have a fixed shape so you can safely bend them and they won't even break. In the model, the shape of the particles changed from the shape of a square and the shape of a triangle. | Gas particles are perfectly compressible, as opposed to the above two. That means you can press it and it will actually become smaller. Then if you put them in a really big container, it will spread all around the container, through diffusion. Note that the triangle here is a lot smaller than the square. |
Quiz
[edit | edit source]- Briefly explain, in your own words, the different properties of the three states of matter and arrangement and movement of their particles.
- Go to the library or search the Internet about 'plasma' the 'fourth' state of matter, and write about 200 words about it.
Answers will vary.
You may have heard about the experiment in which you suspend a glass of water, and the cardboard under it does not fall down. Why does that happen? And what is the secret of the rubber suckers (the real name is suction cup) that hold up your towels? Read this chapter to find out.
Gas pressure
[edit | edit source]Picture this. You have 4 table tennis balls, and you put them in a box of 1m x 1m x 1m. That's pretty big. Now you get a weightlifter to shake it. You will here the sound of the table tennis balls hitting against the wall of the box. Let's assume it's, say, once every ten seconds.
Now you put the balls into a box of 50cm x 50cm x 50cm. This time, you realise that the balls hit against the wall six times every ten seconds! That is because as the volume of the box decreases, there is more chance of the table tennis balls hitting against the wall.
When the table tennis balls hit the wall more, they exert more pressure on it. Pressure is when someone drops a heavy weight on you. Remember that gas particles move freely all the time? When gas particles are put into a container, they hit the walls on the container and exert pressure on them. When you hit put them into a smaller container, they would therefore hit the walls more often as well. The pressure exerted is named 'gas pressure'.
Gas pressure is measured in a unit called 'pascal', or simply 'Pa'. A Bourdon gauge can be used to measure gas pressure. We can also connect a pressure sensor to a datalogger to measure gas pressure.
Air pressure
[edit | edit source]Air is also a kind of gas. Air is made of nitrogen, oxygen and other elements. As air is all around us, it also creates gas pressure. Gas pressure from air is called air pressure or atmospheric pressure. Mind you, you cannot actually feel air particles hitting your skin!
Vacuum
[edit | edit source]
If there is no air, there is no atmospheric pressure. In 1664, the Mayor of Magdeburg combined two hemispheres (halves of spheres), then he pumped the air out of the sphere. He tried to use horses to separate them, but it did not work. Why is that?
This can be explained by the diagram on the left. When the hemispheres are filled with air, the air pressure on the inside equals out the air pressure on the outside. When the inside is hollow, there is no pressure so the air presses the hemispheres together, making it impossible to separate. A place without any air is called a vacuum.

A daily life application is the suction cup where you pump out the air while pressing it onto the wall. As the air is pumped out, the air particles outside press against the sucker, causing air pressure. That's why it takes some effort to take it off!
Quiz
[edit | edit source]Fill in the blanks.
- Gas pressure is caused by gas (1) hitting the wall of its (2).
- (3) pressure is also known as air pressure.
- The power of a (4) can be shown by the (5) hemispheres.
Answers:
- particles
- container
- Atmospheric
- vacuum
- Magdeburg
As you know from kindergarten, some things float while other things sink. You may also know that oil, which is insoluble (cannot be dissolved) in water, floats on top of water. What you might not know is how to find out whether something will sink or float. We will discuss that in this long chapter.
Density
[edit | edit source]First, let's learn a new word: 'mass'. 'Mass' means how much of matter there is. This is not the same as 'volume'. Do you remember that there are always spaces between particles? 'Mass' does not include the spaces between the particles, while 'volume' does. Mass is measured in the units kilogram, gram and milligram.
Imagine you are in an enclosed room, say your bedroom. You are the only one inside the empty, airless room. Therefore, you are the only thing with mass in it. If your room is 1.5m × 2m × 2.5m, and your mass is 30kg, then there are 30kg in every 1.5m × 2m × 2.5m = 7.5 m3. Then, in every average cubic metre, there would be 30 kg ÷ 7.5 m3 = 4 kg.
Remember that in your room, there are (on average) 4 kg in every cubic meter. That is also the density in there: density is the amount of matter in a specific space; the volume is usually represented by an invisible '1'. What are the units of density then? The units of density are always constructed by the unit of the mass, a forward-slash, and then the unit of the volume. By now you may have deduced that the method of finding density is mass over volume. This formula is very important, so we will put it here:
Note: You can also swap them around like you do in maths to find the mass or volume when you have the density.
Floating and sinking
[edit | edit source]
What does all that have to do with floating or sinking? Here's an old riddle: which one is heavier, 1kg of lead or 1kg of feathers? Yes, that's right, they are the same. But what if you put the lead and feathers into a pond? Will they both sink?
Actually, no. While their mass may be the same (1kg), the volume of lead is a lot smaller than that of feathers. As there is the same mass stuffed in a smaller space, it must be denser. The density of water is 1 g/cm3, so if the density of the lead is more than 1g/cm3, it has to sink. On the contrary, the feathers' density is probably less than 1g/cm3, which makes them float.
In other words, whether an object will float or sink in a certain liquid (or gas) depends on their density with relation to the density of the liquid. An example is the diagram on the right.
Unfortunately, we need a bit of math here. The following is a table of the name, mass and volume of some liquids and solids. If all these things are put into a measuring cylinder (or any container for that matter), and nothing dissolves, what will be the order of their altitude, from top to bottom?
| Name | Volume | Mass | Density |
|---|---|---|---|
| Solid W | 0.000 001m3 | 5g | |
| Solid X | 5.4cm3 | 1.8g | |
| Solid Y | 300mm3 | 1.6g | |
| Solid Z | 5cm3 | 0.01kg | |
| Liquid A | 24cm3 | 96g | |
| Liquid B | 24cm3 | 12g | |
| Water | /
|
/
|
Next, we need to know their densities. Remember to change the units. Note that we use division rather than fractions here. We will round off to three significant figures here.
| Name | Volume | Mass | Density |
|---|---|---|---|
| Solid W | 0.000 001m3 = 1cm3 | 5g | 5g ÷ 1cm3 = 5g/cm3 |
| Solid X | 5.4cm3 | 1.8g | 1.8g ÷ 5.4cm3 ≈ 0.33g/cm3 |
| Solid Y | 300mm3 = 0.3cm3 | 1.6g | 1.6g ÷ 0.3cm3 ≈ 5.33g/cm3 |
| Solid Z | 5cm3 | 0.01kg = 10g | 10g ÷ 5cm3 = 2g/cm3 |
| Liquid A | 24cm3 | 96g | 96g ÷ 24cm3 = 4g/cm3 |
| Liquid B | 24cm3 | 12g | 12g ÷ 24cm3 = 0.5g/cm3 |
| Water | /
|
/
|
1g/cm3 |
Then, we arrange them in the correct, ascending order.
5g/cm3, 0.33g/cm3, 5.33g/cm3, 2g/cm3, 4g/cm3, 0.5cm3, 1cm3
0.33g/cm3 < 0.5cm3 < 1cm3 < 2g/cm3 < 4g/cm3 < 5g/cm3 < 5.33g/cm3
Solid X < Liquid B < Water < Solid Z < Liquid A < Solid W < Solid Y

Since Solids W and Y are both on the bottom, they are on the same level—they both sink in liquid A. So, the correct order is as follows:
- Solid X
- Liquid B
- Water
- Solid Z
- Liquid A
- Solid W and Solid Y
You can refer to the diagram on the left, which shows a container with all the solids and liquids in it.
Balloons, ships and submarines
[edit | edit source]Let's get to a topic with no sums: how vehicles work.
Balloons
[edit | edit source]

Once upon a time, when aeroplanes were yet to be invented, the only way to soar through the skies was to ride in a hot-air balloon. A hot air balloon is just like those you ride in amusement parks. They work the same way: using the differences in density to hurl the balloon up.
How exactly? Look at the diagram on the right. Did you know that a hot-air balloon has to be heated before it flies? This is because the particles inside the balloon are just as dense as the particles outside the balloon at the beginning. However, as soon as the particles are heated, they move more vigorously, and as they bump into each other more often, they become less dense. As less dense things float, the balloon will rise.
Ships
[edit | edit source]Many ships are made of steel. Steel is very heavy and dense. Its overall density is approximately 7.85g/cm3. The density of water, however, is much lower, at 1g/cm3. Therefore, it is difficult for steel ships to float if they are made only of steel.
So, how exactly do those ships manage to float? You see, the density of air is very, very low. If there is a large amount of air inside the ship, the total density of the ship will decrease greatly. As long as the density is under 1g/cm3, the ship will float.
Submarines
[edit | edit source]Submarines work in a way that is similar to the ship, except the overall density of the ship can be altered at will so that the submarine sinks or floats as desired. Look at the diagram below. A tank called a ballast tank takes up a significant part of the submarine's area. Note that it does not have to be position like that in the diagram; it can be anywhere the shipbuilder wants it to be. The ballast tank fills up with sea water when the submarine needs to be submerged. When it needs to surface again, the sea water is pumped out of the ballast tank with the air in another tank.
Quiz
[edit | edit source]- Explain why a hot-air balloon cannot rise up if there is no fire.
- Evaluate the following.
| Matter | Mass | Volume | Density |
|---|---|---|---|
| Solid P | 15 | 3 | a |
| Solid R | 100 | b | 4 |
| Liquid H | c | 15 | 6 |
Answers:
- It is because the fire makes the air particles inside the balloon vibrate more vigorously, thus decreasing the density of air inside the balloon, so that it floats.
- a) 5; b) 25; c) 90
Have you ever noticed that there are tiny gaps in rails and flyovers? They are designed that way to prevent the matter from bending or breaking in summer when it expands. Matter always expands, or becomes bigger, when it is heated. This is called thermal expansion. Matter also contracts, or becomes smaller, when it is cooled. This is called thermal contraction. Thermal expansion and contraction greatly affects our daily life and can be explained with the particle model.
Explanation
[edit | edit source]Do you remember, from the last chapter, that as heat made the particles move more vigorously, the volume of the air inside the balloon expanded and density of the air decreased? This is due to thermal expansion. When particles move more vigorously, they take up more space because they need more space for movement. See the diagram below.
The bimetallic strip
[edit | edit source]
A bimetallic strip is made of two separate strips of metal stuck together. These two strips expand to a different extent when heated. So, when the strip is heated, one strip will be longer than the other. Therefore, the bimetallic strip will bend. An example is iron and brass.

Bimetallic strips are used in daily life for heat detection, such as that in fire alarms and thermostats. A thermostat is a small gadget used in various household appliances to automatically switch off the appliance when it is overheating. In such applications, the bimetallic strip bends in order to complete or break a circuit. A circuit means the path on which electricity flows from its source and back. In thermostats, the circuit is broken so that the appliance it is used in can no longer operate. In fire alarms this is the opposite: the circuit is completed so that the fire bell can ring and everyone can escape.
Quiz
[edit | edit source]- Try to draw a diagram that illustrates how a fire alarm works.
- True or false?
- When some home appliances overheat, the bimetallic strip inside them will contract.
- Thermal expansion is limited to liquid and solid only.
- Liquids are not as dense as solids.
- Answers to "True or false?"
- False
- False. Thermal expansion applies to all matter.
- True. As particles become hotter and move more, the spaces between them become greater so they are less dense. Water is an exception though; liquid water is denser than ice.
OK, you've finished the whole book now. In order to supplement (add to) the exercises at the end of each chapter, there is a big quiz you have to finish before getting to the end of this book so that you know how well you did. Older kids may want to use this to prepare for tests or exams. If your computer has flash player 8 or above, you should use the interactive version at this website. Are you ready? On your marks, get set, go!
Part 1: True or false?
[edit | edit source]Each question carries one mark only.
Part 2: Multiple-choice questions
[edit | edit source]Each question carries two marks.
|
1. Which of the following is not matter? | |||||
|
| ||||
|
2. Name the process in which water changes into gas at a fixed temperature. | |||||
|
| ||||
|
3. Which of the following is incorrect according to the particle theory? | |||||
|
| ||||
|
4. Which of the following best describes the movement of pollen grains when hit by water particles? | |||||
|
| ||||
|
5. Hi! If you put me in a box whose capacity is the same as my volume, I will fit nicely inside, but you cannot squash me. I am insoluble in water, and my density is a lot less than water. What could I be? | |||||
|
| ||||
|
6. Hi! We are very good brothers. Once we come together, our total volume decreases. If some other particles come here, we welcome them happily, and let them spread evenly around us. What could we be? | |||||
|
| ||||
|
7. In which state of matter can particles slide over one another but not move freely? | |||||
|
| ||||
|
8. Which of the following statements about air particles in a hot-air balloon is not true? | |||||
|
| ||||
| 9. Given that the particles are gaseous and are enclosed in the boundaries of the diagram, what will the red particles do and why? |
| ||||
|
| ||||
|
10. Which of the following inventions is related to thermal expansion? | |||||
|
| ||||
How did you do?
[edit | edit source]If you got over 80% of the answers, congratulations! You are now familiar with the science of particles. If you got under 50%, you'd better read through the book again to ensure that your understanding of particles is good. Before you put down this book, we will look through the main points again. If you forgot about some of the things, you can always look at the summary again. This will save you a lot of time.
- Properties of matter
- Matter must have mass
- Matter must take up space
- Different kinds of matter
- Solids' shapes cannot be altered, and nor can its volume
- Liquids' shapes can be altered, but not its volume
- Gases' shapes can be altered, and so can its volume
- Change of states
- Solid → Liquid: Melting
- Happens at a fixed temperature
- Liquid → Solid: Freezing
- Happens at a fixed temperature
- Gas → Liquid: Condensation
- Does not happen at a fixed temperature
- Liquid → Gas: Boiling
- Happens at a fixed temperature
- Evaporation does not happen at a fixed temperature
- Solid → Liquid: Melting
- Matter is made of particles;
- Particles are infinitesimal, and current scientific instruments cannot see them;
- Different kinds of particles have different sizes;
- There must be gaps between particles
- Particles are always in motion.
- A common phenomenon observed in particle movement
- Particles always go to a place of lower density
- When particles collide, they move in random directions
- Observed in smoke cells
- Discovered by Robert Brown
- In 1827
- While observing pollen grains
- A model that mimics the movement of particles
- Can explain:
- The three states of matter
- Gas pressure
- Density
- Thermal expansion and contraction
- Heat makes particles move more vigorously
- Solid
- Particles cannot move freely
- Particles only vibrate in fixed positions
- Particles take on a fixed shape
- Liquid
- Particles do not take on a fixed shape; their shape depends on that of their container
- Particles have a fixed volume
- Particles can slide over one another
- Particles do not move as freely as those in gas
- Gas
- Particles move freely in all directions
- The shape and volume depend on that of the container
- Refers to the pressure exerted on a surface by gas
- Measured
- in Pascals, or 'Pa'
- With a Bourdon gauge or pressure sensor
- Appears in air, in which case it's called air or atmospheric pressure
- Vacuum
- Absence of air
- Magdeburg hemispheres
- Two hollow hemispheres form an empty sphere
- The hemispheres cannot be separated
- The air pressure on the outside greatly exceeds that of the inside
- If an object is submerged in a liquid or gas, it:
- Floats if its density is less than that of the liquid or gas
- Sinks if its density is more than that of the liquid or gas
- Density of water = 1
- Hot-air balloons
- There is an engine in the balloon
- As the engine is heated, the particles inside the balloon move more and are therefore further apart
- As density of the air inside the balloon deceases, it floats.
- Ships
- The overall density of ships are under that of the water because there is a lot of air inside
- Submarines
- Whether it is submerged or surfaced depends on the ballast tank
- When the ballast tank is filled with seawater, it sinks
- When the water is pumped out, it surfaces
- Particles move more vigorously when heated, and are further apart as a result
- This makes the object larger, or expands
- When they are cooled, the particles move less vigorously and are closer together
- The object is made smaller, or contracts
- Bimetallic strips
- Made of two metal strips composed of different metals
- The metals do not expand to the same extent
- The strip bends when heated as a result
- Used in
- Thermostats
- The connection is cut off when an appliance is too hot
- The bimetallic strip in the appliance expands and therefore no longer completes the circuit
- Fire alarm
- The circuit is closed when the fire alarm is too hot
- This makes the alarm ring because the built-in bimetallic strip completes the circuit
- Thermostats
If you have contributed to this book, by commenting on it, correcting typos, adding BookCats, expanding content, reverting vandalism, etc., then add your name here with *{{user}}.










