Structural Biochemistry/Protein function/Heme group/Hemoglobin
Hemoglobin is a protein that is carried by red blood cells. Hemoglobin picks up oxygen in the lungs and delivers oxygen to the tissues to maintain cellular viability. It is composed of four polypetide chains with four prosthetic groups. Two of the units have the same amino acid sequences and are called α-chains, the other two identical amino acid sequences are called β-chains. The α-chains and β-chains combine to form the body of hemoglobin. Usually the α-chains combine with the β-chains rather than α-chains and α-chains while β-chains combine with β-chains. The latter is not expressed before birth. Hemoglobin protein which is found only during fetal development is called gamma (γ). It substitutes for the β-chains. Fetals have γ-chains instead of β-chains because it needs a higher affinity for oxygen, so the fetuses can draw sufficient amount of oxygen from the mother. The fetus accomplishes this task by altering the amino acid sequences of the β-chains. γ-chains altered in the way that it removes the two positive charges on the β-chains in which the 2,3-bisphosphoglycerate (2,3-BPG). This reduces the affinity for 2,3-BPG. Since 2,3-BPG lowers the affinity for oxygen, reduction in that would increase the fetus' ability to bind to an oxygen. The fact that 2,3-BPG helps hemoglobin protein binding oxygen molecule for more oxygen to be delivered to body tissues is known as heterotropic allosteric effect. Bisphosphoglycerate (BPG), pH, and carbon dioxide can have an effect on the hemoglobin. In the cell, when the pH decrease, it lowers the affinity of oxygen in the cell and therefore becomes more efficient oxygen tranporter. When the presence of carbon dioxide increases, it lowers the affinity of oxygen in the cell which is another factor that allows cell to become a better oxygen transporter.
A protein of tetramer, with two α and β units of amino acids or, globin or globular protein and four heme groups that each has an iron atom, and the importance is that Fe2+ is, by which bonding to globin group by proximal histidine and oxygen molecules by bent form of geometry by cooperativeness for the biological function of the protein such that they are circulated through body in red blood cells to be delivered to tissues (to be used for glycolysis and oxidative phosphorylation), and bonding to carbon dioxide for it to be transported to lungs (to be exhaled as waste). Oxyhemoglobin is the form of hemoglobin that the heme group of the protein molecule binds to oxygen; deoxyhemoglobin is that without oxygen.

Binding Sites of Hemoglobin
[edit | edit source]The iron ion of +2 state of heme group is bonded to four nitrogens in one plane of 4 porphyrin ring and a proximal histidine amino acid of imidazole ring of globin molecule under plane of porphyrin ring. The heme group binds oxygen by bending. This does not increase the oxidation state of the iron ion but it decreases in atomic size. This causes the iron ion to move into the plane of porphyrin ring and brings up the proximal histidine amino acid group. Ultimately, it changes the allosteric conformation of globulin molecule. As iron binds oxygen, the iron ion changes its position to stay in the same plane of porphyrin ring. This causes the bond distance of iron and oxygen to increase while the bond distance of iron to histidine of imidizole ring decreases. The decrease of iron to histidine of the imidizole ring causes the plane of the ring to move out of tetramer of hemoglobin. Now, the binding sites of the heme groups are opened up for the bonding of oxygen. Overall, the process is classified as positive cooperativeness. The hemoglobin protein's binding affinity to oxygen can be increased by increasing substrate saturation of oxygen molecule. The bonding of the hemoglobin protein and oxygen changes the conformation of the binding site which results in an increase affinity for other oxygen molecules to bind the protein molecule. The indirectly proportional relation for the binding affinity of hemoglobin protein molecules between oxygen and carbon dioxide is known as Bohr effect, for which, the oxygen binding curve of the protein molecule changes place to the right of the graph, as such, the decreased level of carbon dioxide bonding to hemoglobin increases oxygen bonding affinity to the protein molecule.
2,3 Biphosphoglycerate Function
[edit | edit source]
2,3-BPG binds to hemoglobin in the center of the tetramer to stabilize the T state (E.g. in muscle tissues).
2,3-BPG is also extremely important in the role of stabilizing the T state of hemoglobin. The hemoglobin wants to change into a more favorable R state due to the fact that the T state is quite unstable. Pure hemoglobin (without 2,3-BPG) was tested and found to bind oxygen much more vigorously than hemoglobin in blood. A hemoglobin with 2,3-BPG has a lower affinity for oxygen binding in the tissue which allows it to be a better oxygen transporter than a pure hemoglobin, which does not have 2,3-BPG. When 2,3 BPG is present, it transports about 66% of oxygen while the pure hemoglobin only transports about 8%. The reason is that the 2,3-BPG binds inside of the hemoglobin and somehow stabilizes its T state (the state that has less affinity for oxygen). When enough oxygen has bonded to hemoglobin, a transition occurs from the T state to the R state, which releases the 2,3-BPG. 2.3-BPG stays in the hemoglobin until enough oxygen has come to replace it. This keeps the oxygen in its T state until it is ready to transition to the R state (where its affinity for oxygen increases dramatically). This broadens hemoglobin's oxygen saturation curve.
When the oxygen binds to Hemoglobin, the iron in the Heme group moves from the outside to the inside of the plane. In doing so, the Histidine side group also changes its alpha helix when the iron moves. Thus, this triggers the carbonyl terminal of the alpha helix to change position and that favors transition from T state to R state.
2,3-Bisphosphoglycerate (2,3-BPG) or 2,3-diphosphoglycerate (2,3-DPG) binds to deoxyhemoglobin with larger bonding affinity, such that it makes the T state of hemoglobin protein more stable or increases oxygen affinity of the protein; its biological function is to control bonding between hemoglobin and oxygen molecules for oxygen to be released to body tissues.
This allosteric effector binds to a site on the tetramer that is only present on the Tense(T) form of hemoglobin. The site is in the form of a pocket which is bordered by beta subunits. These positively charged subunits, His143, Lys82, and His2, interact with 2,3-BPG holding it in place. When the equilibrium of the tense form is pushed to the relaxed(R) form, the bonds that hold the 2,3-BPG molecule in place are broken and it is released. The hemoglobin stays in the T form during low concentrations of oxygen, so when 2,3-BPG is present, more of the oxygen binding sites must be filled in order for the transition from T to R form to occur.
Cooperativity
[edit | edit source]The cooperativeness of hemoglobin makes it a much more efficient transporter of oxygen than myoglobin. Although myoglobin and hemoglobin both become highly saturated with oxygen at high concentrations (E.g. in the lungs), hemoglobin is characterized by much weaker binding to oxygen at low concentrations compared to myoglobin. The cooperativeness of tetramers work both ways in hemoglobin. As one oxygen molecule binds to one heme group, the oxygen affinity for the other groups increase. Once an oxygen molecule is released, this stimulates the release of the other oxygen molecules. This makes hemoglobin ideal in transporting and releasing oxygen from lungs to tissues where it is needed.
Cooperativeness occurs as a result of a change in the hemoglobin structure. In the deoxy form (where oxygen is absent), hemoglobin exists in the T (tense)-state. Upon oxygenation, the dimers in hemoglobin shift by 15 degrees and the R (relaxed)-state is adopted. The R-state form has a much higher affinity for oxygen.
For specifics sakes, the cooperativeness experienced in hemoglobin among its four separate monomers occurs as a result of a proximal histidine shift when one monomer binds to an oxygen atom. The heme group of hemoglobin is situated in such a way that it is composed of 4 pyrrole coordinating around an iron ion. In addition, there is a proximal histidine group that is also coordinated the iron group constituting the 5th coordination ligand. In the deoxy form, the iron ion is not completely in the plane of the pyrrole rings, in fact it is about 0.4 angstroms below the plane of the ring. This downward shift is due to the proximal histidine ligand on the bottom of the coordination complex. However, when one of the monomers binds to an oxygen molecule, the iron ion gains a sixth coordination ligand, the oxygen molecule itself, and it pulled up 0.4 angstroms to the plane of the pyrrole rings. This shift upwards also pulls the proximal histidine group up as well. It this movement of the histidine group that contributes to the cooperativeness property of hemoglobin. The proximal histidine is located at the interface of the alpha and beta subunits found in hemoglogin (hemoglobin having two identical alpha units and two identical beta units). When the histidine group moves upwards, it forces a conformational change in that interface, which conforms the next monomer to situate itself in a fashion that increases its affinity to another oxygen molecule. As that monomer binds an oxygen molecule, the whole process happens again. It this cascade of events, the iron shifting up upon binding and the histidine moving up as a result, that describes the cooperativeness that hemoglobin has between its four monomers and the transition it makes from the T state to the R state.
Chemical process by which as active site of enzyme is bonded by substrate, the enzyme can react with substrate with more effect; three forms of which are positive cooperativeness, negative cooperativeness, and non-cooperativeness; for positive cooperativeness, for example, when oxygen binds to hemoglobin, the affinity of the protein for oxygen increases; therefore, binding of oxygen to the protein is more easily done; for negative cooperative, for example, when enzyme binds to ligand, the bonding affinity decreases.
Sigmoid Binding Model
[edit | edit source]From the oxygen binding curve of the hemoglobin, it is said that hemoglobin follows a sigmoid model because it looks like a "S" shaped curve. The curve also suggested that hemoglobin has a lower oxygen binding affinity. This is due to that fact that hemoglobin binds to 2,3 bisphosphoglycerate inside of the red blood cell.
The sigmoid binding model of the curve indicates that hemoglobin follows a special oxygen binding behavior, known as cooperativeness. The curve shows that binding at one site of the protein will increase the likelihood of other binding at other sites. And also the unloading of oxygen at one site will also facilitate the unloading of oxygen at other sites.
The biological of this sigmoid model of oxygen binding leads to efficient oxygen transport. The unloading of oxygen can be seen in the graph where in the lungs (100 torr) the protein is saturated with oxygen and all of the oxygen binding sites are occupied. However when this is moved to the tissues to release the oxygen, the saturation level drops and the total unloading oxygen level is 66%. This situation is favored because the hemoglobin goes through cooperativeness and it increases the tendency for oxygen binding and unbinding. Unlike myoglobin, which binds to tightly to oxygen for its release.
Cooperativity Models
[edit | edit source]In the concerted model, T and R states are the only two forms of hemoglobin that exist. T state is the state where hemoglobin has its quaternary structure in the deoxy form, which is also a tense form. The R state is the state where the hemoglobin has its quaternary structure in completely oxygenated form. This state is relaxed, less constrained, and leaves the oxygen binding sites free. An equilibrium exists between these two states that is shifted by the binding of oxygen, which shifts equilibrium towards R-state. This shift (to R-state) increases the affinity of oxygen of its binding sites. All tetramers of the hemoglobin must be in the same state.
In the sequential model, there is no full conversion from the T-state to R-state. The binding of oxygen changes conformation of the subunits, which subsequently induces changes in other subunits to increase their affinity for oxygen. The subunit to which the a ligand binds changes its conformation without interrupting other subunits to have conformational changing.
In the curve of fractional saturation (fraction of possible binding site that include the binded oxygens) vs. the concentration of oxygen measured by its partial pressure in torr, the T-state binding curve is relatively shallow at low concentration of oxygen when all molecules are in the T state because if a molecule is assumed in the R state, the oxygen affinity increases, which means that new oxygen molecules have more chances to bind to the rest of the three unoccupied sites. The R-state binding curve goes sharply at the beginning but level off when all of the binding sites are occupied by oxygens.
Hemoglobin behavior resembles a mix of these two models. A molecule with only one bound oxygen molecule exists primarily in T-state, but the other subunits have a much higher affinity for oxygen as suggested by the sequential model. Meanwhile, a molecule with three subunits bound exists primarily in the R-state as suggested by the concerted model.
File:Transition.JPG Le Chatelier's Principle can be seen to play a role in the circulation of CO2 in the body. Within the erythrocyte, by decreasing the concentration of HCO3-, it acts a force in which it requires more CO2 to be in the cell so that it can be converted to HCO3-.
H2O + CO2 <---> H+ + HCO3-
This reaction, which is carried out by carbonic anhydrase, also decreases the pH within the erythrocyte. Consequently this encourages the hemoglobin to take on the T-state as the excess hydrogen in the cell allows for salt bridges to form. These salt bridges then induce the cell to form the T-state more often than the R-state.
Allosteric Effectors of Hemoglobin
[edit | edit source]An allosteric effector of hemoglobin is a regulation by a molecule that is structurally unrelated to oxygen and binds to a site completely distinct from the oxygen binding site.
2,3-Bisphosphoglycerate
[edit | edit source]
2,3-BPG is a highly anionic compound found in hemoglobin, making it an efficient oxygen transporter. It lowers the oxygen affinity of hemoglobin by binding in the center of the tetramer, stabilizing hemoglobin's "T" state. 2,3-BPG, with such high negative charge, interacts with 3 positively charged groups on each beta chain in the two alpha-beta dimer. For the transition of "T" state to "R" state to occur, the 2,3-BPG must be broken. For this to occur, more oxygen-binding sites within the hemoglobin tetramer must be occupied. Therefore, the hemoglobin remains in the lower-affinity T state until a much higher oxygen concentration is reached. In pure hemoglobin with no 2,3-BPG, only 8% of the sites would contribute to oxygen transport. The presence of 2,3-BPG in hemoglobin increases the percentage to 66%.

Fetal hemoglobin has a higher affinity for oxygen than does regular hemoglobin. Regular hemoglobin is made of two alpha/beta dimers, while fetal hemoglobin is made of two alpha/gamma dimers. The gamma subunits have a lower affinity for binding 2,3-BPG. Thus, with less 2,3-BPG, fetal hemoglobin has a higher affinity for oxygen. This is advantageous for the fetus, as oxygen must be carried longer distances (from the mother) than in regular situations.
The Bohr Effect (Hydrogen Ions and Carbon Dioxide)
[edit | edit source]Hydrogen
[edit | edit source]A hemoglobin traveling from a region of high pH to a region of lower pH has a tendency to release more oxygen. This is because as pH decrease, the oxygen affinity of hemoglobin decreases. The "T" state of the hemoglobin is stabilized by 3 amino acids (alpha2 Lys40, beta1 His146, beta1 Asp94) that form 2 salt bridges. The residue at the C terminus of the His146 forms salt bridge with the lysine residue in the alpha subunit of the other alpha-beta dimer. The salt bridge between the His146 and the Asp94 is formed only when pH drops, protonating the side chains of His146.
Carbon Dioxide
[edit | edit source]Carbon dioxide also stimulates oxygen release in the hemoglobin. Carbonic anhydrase takes carbon dioxide diffused from the tissue into the red blood cell and water to yield carbonic acid (H2CO3), which is a strong acid (pKa 3.5). Once this compound is formed, it dissociates into HCO3- and H+, which increases the acidity of the environment and lowers the pH level. This drop in pH level will again stabilize the T-state of the hemoglobin. In the hemoglobin, there are three key amino acid residues responsible for the bind of oxygen to the active site: lysine (Lys), histidine (His), and aspartate (Asp). The three amino acids are linked by two salt bridges. One of the salt bridge, between histidine and aspartate, does not form until there is an proton added to histidine. Under conditions of low pH, the histidine gets protonated to allow then the formation of the salt bridge and thus, a conformational change that stabilizes the T-state, lower its affinity for oxygen. In addition, carbon dioxide reacts with the amino-terminals of hemoglobin, resulting in the formation of negatively charged carbamate groups which further stabilize the T state by supporting the salt bridge interactions. This is convenient on a physiological sense. Since tissues tend to be low in oxygen and high in carbon dioxide concentration, the low pH environment will lower hemoglobin's affinity for oxygen and cause the red blood cell carriers to release the oxygen at the tissues.
Carbon Dioxide Transport from Tissues to Lungs
[edit | edit source]Hemoglobin's loss of oxygen
[edit | edit source]Hemoglobin is an efficient oxygen transporter around the body. How does it release oxygen to the tissue? Hemoglobin releases oxygen where it is a necessity. Examples include working muscles and tissues. When tissue is metabolizing, it releases carbon dioxide and hydrogen ions. Hemoglobin reacts these conditions. These are called the carbon dioxide effect and the pH effect.
Christian Bohr discovered that hemoglobin is found to have a lower oxygen saturation in lower pH. The release of protons signifies a change in pH. The reason is that protons protonate a histidine on the end of one of the beta chains found on the hemoglobin. Consequently, this makes the histidine charged and creates a salt bridge (ion-ion interaction) with aspartate (negatively charged) on the same polypeptide chain. That salt bridge stabilizes the T state of hemoglobin, which favors the release of oxygen.
Carbon dioxide released by cells are mixed with the blood serum to make carbonic acid. Carbonic acid is a relatively strong acid, so it dissociates into bicarbonate and a proton (which can be used above). The carbon dioxide itself, however, can also participate in oxygen release. When the carbon dioxide meets the terminal amino group of hemoglobin's peptides, it can react to form carbamates, which are negatively charged. This reaction also produces an additional acidic proton. These negatively charged groups can also participate in salt bridges that further stabilize the T state of the hemoglobin to further facilitate the release of oxygen. This effect was seen when someone noticed that oxygen saturation was lowest when in an acidic and carbon dioxide-rich environment.
Mutations in Hemoglobin
[edit | edit source]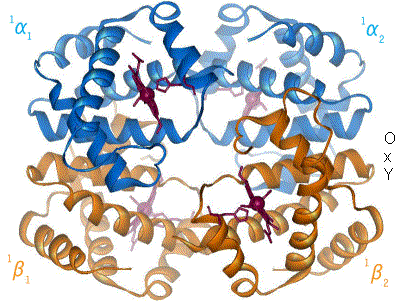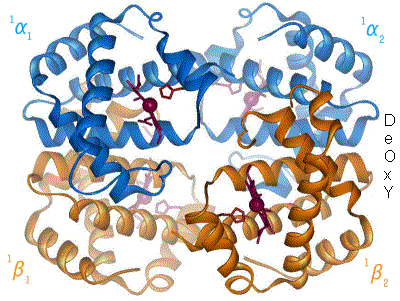
The deficiency or mutations of hemoglobin can be a result of abnormality of structure of protein molecules, which is related to anemia and sickle cell disease - a condition that malformed red blood cells are resulted from structural abnormality of hemoglobin molecules restrict passage of blood vessels for the supply of blood flow to body tissues.
Sickle cell anaemia is a disease caused by one amino acid substitution. In this case, valine was substituted for glutamate in position 6 of the beta chain of hemoglobin. The mutated form is known as Hemoglobin S (HbS). The elimination of the negative charges of glutamate and the substitution of glutamate with hydrophobic valine causes hydrophobic interactions between different hemoglobins. From electron micrographs, studies have shown that hemoglobin in sickle red blood cells form large fibrous aggregates. The fibrous aggregates form across the red blood cells distorting the shape and increasing their potential to clog small capillaries. This detrimental effect leads to a high risk of stoke and bacterial infection from poor blood circulation. Not only does the sufferer attain insufficient amounts of oxygen, but the aggregated hemoglobins make it harder for blood to flow through small vessels which can cause blood clots. It is interesting to note that the areas with high population of people carrying sickle cell traits is correspond to the areas with high prevalence of malaria. Sickle cell traits means that a person have one normal gene and one mutated gene, disease does not take place in these people. The reason is that a person carrying sickle cell traits are resistant to malaria. The malaria causing parasite can not reproduce effectively in people with sickle cell traits. It should be noted however that the sickle cell gene is codominant. This means that in a person carrying one normal gene and one mutated gene, there will be both regular healthy blood cells and mutated blood cells. However, the function of the normal blood cells makes up for the abnormal cells and no symptoms are felt.
