Structural Biochemistry/Exergonic reaction
General information
[edit | edit source]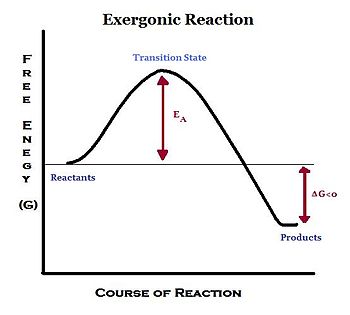
An exergonic reaction refers to a reaction where energy is released. Because the reactants lose energy (G decreases), Gibbs free energy (ΔG) is negative under constant temperature and pressure. These reactions usually do not require energy to proceed, and therefore occur spontaneously. In a chemical reaction, breaking and forming bonds between atoms is a form of energy. Since chemical reactions mainly consist of forming and/or breaking chemical bonds, exergonic reactions release energy by breaking less stable chemical bonds and forming more stable bonds. The example of exergonic reactions occur in our body is cellular respiration: C6H12O6 (glucose) + 6 O2 -> 6 CO2 + 6 H2O this reaction release energy which is used for cell activities.
However, some exergonic reactions do not occur spontaneously and require a small input of energy to start the reaction. This input of energy is called activation energy. Once the activation energy requirement is fulfilled by an outside source, the reaction proceeds to break bonds and form new bonds and energy is released as the reaction takes place. This results in a net gain in energy in the surrounding system, and a net loss in energy from the reaction system; hence, the change in Gibbs free energy (ΔG)] is negative (ΔG < 0). A negative ΔG denotes that the reaction is spontaneous and thermodynamically favorable.
Here is an endergonic reactions of ATP give energy. First Breaking down the ATP formed ADP and Pi is an exergonic reaction, where Delta G is less than 0. However, by combining the reaction glucose+Pi ---> glucose 6-phosphate, a thermodynamically unfavorable reaction. The cell can drive an endergonic reaction.
Conclusion
[edit | edit source]Exergonic reaction is
- Free energy of final state < free energy of initial state
- Free energy is free during the reaction
- The energy can be used to do biological work
- Able to work in spontaneous reaction
- doesn’t have to occur in great rate which is where the enzyme comes in
- -ΔG (negative number)
Reference
[edit | edit source]http://academic.brooklyn.cuny.edu/biology/bio4fv/page/exergon.htm
