Structural Biochemistry/Enthalpy
History of Development
[edit | edit source]The enthalpy of reaction at specific values of T and P is defined as the heat exchanged between the system and the surroundings as the reactants are transformed into products at conditions of constant T and P. The heat flowing into the system is given a positive sign. Also, the standard enthalpy of formation refers to one mole of the specified reaction at a pressure of 1 bar. The term "enthalpy" and its accurate definition did not come about until the late 1840s. Before then, scientists and engineers knew from experience that a fluid called heat transfers from a hotter object to a cooler object when they are brought into contact. The result is that the cold object becomes warmer, and the hot object becomes cooler as heat transfers between them. Enthalpy is also the heat energy that absorbed or released as the reactants become the products at a constant pressure. When enthalpy is negative, the heat energy is released. However, when enthalpy is positive, the reactants absorb the heat from their surroundings.
A reasonable view is that there is something transferred from the hot object to the cold one. This “something” was referred as heat. Thus, it was convenient to say that heat always flows from higher temperature to lower one. This conclusion established the concept of temperature gradient, which may be viewed as the driving force for the transfer of energy as heat. Later, through conducting more precise measurements and experiments, scientists concluded that the rate of heat transfer from one object to another is proportional to the temperature difference between the two objects. Thus, the heat transfer is negligible when the temperature differences between the two objects are negligible.
A Thermodynamic View of Heat
[edit | edit source]Heat, or energy transferred between a hot and a cold object, is never regarded as being stored with an object from the thermodynamic point of view. Form this view point, heat is defined to exist only as energy is in transit from one object to another; or in the thermodynamic terminology, from the system to the surroundings. When heat is added or subtracted from a system or its surroundings, it is thought that energy is not stored in or being pulled away from the point of contact. Rather, heat is thought to act as kinetic and potential energy. The addition and subtraction processes are considered to be the transfer from potential energy to kinetic energy, and vice versa.
Relationship between Heat and Enthalpy
[edit | edit source]In spite of the transient nature of heat, the definition of heat is always viewed as a unique property of the object that is being measured. As mentioned above, temperature changes were used as the primary units to measure heat until the late 1840s. In fact, the definition of the unit of heat was based on the temperature change of a unit mass of water, as seen in the British thermal unit. The British thermal unit is commonly known as the Btu.
The British thermal unit is defined as 1/180th quantity of heat which when transferred to one kilogram mass of water raised its temperature from 0 to 100 degree Celsius at standard atmospheric pressure. This primitive definition of heat failed to provide an accurate measurement. The main reason accounts for this inaccuracy lie in the reference object, which in this case is water. The measure temperature change is partially determined by the purity of the water. The purer the water is, the more amount of heat is need for the temperature to increase by one unit.
In the twentieth century, more and more scientists realized that heat is just another form of energy that is being transferred by making contact of two objects. Thus it is convenient and practical to measure heat by using an energy unit.
The Modern Concept of Enthalpy
[edit | edit source]The modern concept of enthalpy or heat was developed largely based on the series of experiments carried out by James P. Joule (1818–1889). Joule’s experiment involves carefully measured amounts of water, oil, and mercury in an insulated container. He placed known amounts of these afore mentioned substances in the container, and agitated the fluids with a rotating stirrer. The mechanical work needed to carry out the rotation was carefully calculated, and the temperature changes of the fluid temperatures were accurately measured. In his result, Joule stated that for each fluid that a fixed amount of work was required per unit mass for every degree of temperature rise caused by the mechanical work provided. Furthermore, the original temperature of the fluids could be restored by the transfer of heat through contact as simple as to cooler objects. The quantitative relationship between heat and work was thus firmly established by Joule’s experiment, which proved definitively that heat is a form of energy.
In order to give heat a more concrete definition and moreover, to recognize a common basis for all energy units, international steam table calorie is defined in relation to joule, the SI unit of energy. Joule is the mechanical work done when a force of one Newton acts through a distance of one meter. Upon the establishment of the SI unit, the international steam table calorie was defined as equivalent to 4.1868 Joule (exact, by definition) and thermochemical calorie is equivalent to 4.184 Joule (exact, by definition). As a reference, one international steam table Btu is equivalent to 1055.056 Joule, and one thermochemical Btu is equivalent to 1054.35 Joule.
Mathematical Expression of Enthalpy
[edit | edit source]Enthalpy(H) is a measure of the heat energy of a reaction. The Enthalpy H of a thermodynamic system whose internal energy, pressure, and volume are U, P, and V is defined as H =U+PV. Since U, P, and V are state functions, H is a state function. note from dw=-PdV that the product of P and V has the dimensions of work and hence of energy. Therefore it is legitimate to add U and PV. Naturally, H has units of energy.
For example, a reaction that is exothermic will have a negative change in enthalpy. This is because the enthalpy of the products is less that that of the reactants.
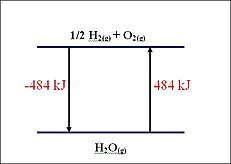
In mathematical terms enthalpy of a reaction can be known as the following:
ΔH = (sums of bonds broken) - (sums of bonds formed)
ΔH = ΣnHproducts - ΣnHreactants
When ΔH is negative then the reaction is exothermic and more bonds are formed than broken.
If ΔH is positive then the reaction is endothermic. Chemical bonds tend to form spontaneously that the negative value of ΔH represents exothermic reaction.
The enthalpy for the reverse reaction is equal in magnitude, but opposite in mathematical sign.
Enthalpy is a state function and thus according to Hess's Law, the overall enthalpy of the reaction is equal to the sum of the enthalpies of the individual reactions or steps for which the overall reaction can be divided.
Since enthalpy is a function defined for the sake of calculations, it is difficult to measure the actual enthalpy of a substance. The change in enthalpy however, is easily measured and is an important quantity in many calculations.
When enthalpy changes, it signifies there is a change of state happening in the system. But enthalpies are reversible in the sense that the physical state changes are usually reversible. Therefore, since the enthalpy for formation of everything is always given, then at any given change, the new enthalpy can be calculated.
Reference
[edit | edit source]Slonczewski, Joan L.. Microbiology "An Evolving Science." Second Edition.
Smith, J.M. (2005). Introduction to Chemical Engineering Thermodynamics. McGraw Hill. ISBN 978-007-127055-7. {{cite book}}: Text "coauthors+ H.C. Van Ness, M.M. Abbott" ignored (help)
Engel, Thomas. Physical Chemistry. Third Edition.