Structural Biochemistry/Alzheimers
Overview
[edit | edit source]Alzheimer's is a form of dementia, a decline in mental ability that affects everyday life. This disease attacks the brain and causes problems associated with memory, thinking, and behavior. As time progresses, the symptoms usually get worse. It is usually assumed that Alzheimer's is a result of aging; however this is not the case. Aging simply increases the risk factor of obtaining this disease. As of now, there is no cure for Alzheimer's; treatments usually are only able to slow the disease from progressing.
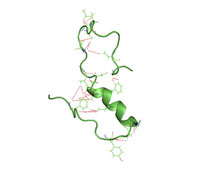
In association with Alzheimers disease are peptide proteins known as β-Amyloid. They are found in the brains of humans diagnosed with Alzheimers Disease. Researchers have been trying to study these peptides so that they can work to find a cure (or treatment) that would help the patients. To understand so, it requires understanding the structure of the proteins. Because of the complex structures, there has been limited compilations, but there has been progress. [1]
Symptoms
[edit | edit source]Memory – Memory loss associated with Alzheimer’s disease persists and only gets worse. Some symptoms may include:
- Repeating the same sentence over and over
- Forgetting conversations, past events, or future appointments
- Misplacing of possessions
- Forgetting names of family members and relationships
- Difficulty understanding surrounds, may not know when or where
Speaking, Writing, Thinking, and Reasoning
- Initially having trouble finding the right words during a conversation
- Eventually lead to the loss of speaking and writing abilities
- May eventually having trouble understanding conversation or written text
- Poor judgment and slow response
Changes in personality and behavior
Brain degradation of Alzheimer’s may affect how people feel. People with Alzheimer's may experience:
- Depression
- Anxiety
- Social withdrawal
- Mood swings
- Distrust in others
- Increased stubbornness
- Irritability and aggressiveness
- Changes in sleeping habits
As the disease progresses, the symptoms could only increase in severity to more severe memory loss, confusion about events in regards to time and space, and disorientation. To try and understand the correlation between the symptoms and the disease, there must be an understanding of the structures of the proteins that have commonly been found in all the patients- β-amyloid (Aβ) peptides. Many people who are affected with Alzheimer's require continuous attention and care since they are unable to perform even basic daily activities.
Test For Alzheimer's
[edit | edit source]A diagnosis of Alzheimer’s disease may include a complete physical and neurological exams, CT (computed tomography), and MRI(magnetic resonance imaging). Biopsy of the brain and identifying evidence of any of the following: Neurofibrillary tangles, Neuritic plaques, and Senile plaques. Neurofibrilary tangles are twisted filaments of proteins within nerve cells that clog up the cell, inhibiting neurotransmitters and the function of the nerve cell. Neuritic plaques are abnormal clusters of invalid or dying nerve cells. Senile plaques are areas of waste products around proteins that were produced by dying nerve cells. All of these may be caused by or inducing Alzheimer’s disease.
Amyloid Fibrils
[edit | edit source]
β-amyloid (Aβ) peptides segregate into different domains. Included are amyloid fibrils, protofibrils, and oligomers. These have been under study in hopes to understand Alzheimers Desease. Unfortunately, understanding their whole structure has been difficult. The B-ayloid (AB) peptide forms naturally in the human body within in the brain as a protein precursor of a proteolytic fragment. It is specifically the AB amyloid fibriles that form the core of a dense plaque within the brain leading to alzheimers disease.
Amyloid fibrils are known as fibrillar polypeptide in collection with an intermolecular cross-β structure. X-ray diffraction showed that the B-strands hydrogen bond with each other and orient in a parallel manner along an axis. The B-amyloid (AB) peptide is amphiphilic having a hydrophobic C-terminus lasting for about 37-42 residues, and a hydrophilic N-terminus. These structures twisted as crossovers, and estimated to have a length of about 1 micrometer. Unique about the AB fibrils are their polymorphism. This refers to their ability to conform to different arrangements. These arrangements include the fibrils different in the number of protofilaments, differing in their orientation, and differing in their substructure. These differences is relevant for humans because it could contribute to folding , reactions, and eventually to the level of alzheimers that the human has. [1]
Aside from the polymorphism, there is further diversity among the amyloid peptides due to structural deformations. This includes different bends and twists. These deformations allow study of nanoscale mechanical properties of the fibrils.
Structure of β-amyloid
[edit | edit source]The β-amyloid peptide is a natural forming proteolytic peptide found in the human brain. It is intrinsically unstructured, meaning that it lacks a stable tertiary structure. Many β-amyloid peptides have disordered and unfolded structures that can only be observed using NMR (nuclear magnetic resonance). The peptide is amphipathic, possessing a hydrophilic N-terminus and a hydrophobic C-terminus. The C-terminus can bind up to 36-43 amino residues, which creates the overall structure of the peptide chain. A great number of β-amyloid isoforms differ by one amino residue; many are closely related to Alzheimer’s. β-amyloid undergoes many complex fibrillation pathways, creating intermediate structure such as oligomers, amyloid derived diffusible ligands, globulomers, paranuclei, and protofibrils. When any of these intermediates decide to plaque the walls of cerebral blood vessels, Alzehimer’s disease may be underdevelopment.
The cross-β sheet structure of a β Amyloid
[edit | edit source]Many different amyloid-like polypeptides show a common cross β-sheet structure. These β-sheets are perpendicularly attached microcrystal backbone through non-covalent and hydrogen bonds, and they have parallel conformations between sheets. Recent studies have shown crystallographic evidence of these microcrystal called steric zippers, which are present in many amyloid fibrils. Steric zipper is a structure of a pair of two cross-β sheets with side chains that resemble a zipper. There are dry and wet interfaces of the cross β-sheet conformation. The wet interface is covered by water molecules, which create a greater distance between two adjacent sheets. The dry interface does not contain water so the distance between two adjacent sheets is much closer. While the polar side-chains of the wet interface is stabilized by hydrogen bond interaction, the side-chains of the dry interface are integrated by adjacent side-chains by stacking the previously mentioned steric zippers. Different β-amyloids with different lengths of residues favor either the parallel or antiparallel form. A β-amyloid can also have segmented parallel and antiparallel structures. For example, residues 1-25 would have one conformation. Residues 26-43 would have the other. From These β-sheet structures assign many distinguishable properties to β-amyloid. For example, β-amyloids have a high affinity to specific dyes such as Congo red and Thioflavin T. These dyes can help mark and track the activity of β-amyloids inside the brain.
Models of Amyloids
[edit | edit source]There have been two forms of Aβ peptides that have been under study: Aβ(1-40) and Aβ (1-42). The numbers 40 and 42 refer to their respective amount of residues.
The Aβ(1-40) is proposed to be more pathogenic than the Aβ(1-42) form. When experimented with the model Drosophilia melanogaster, the Aβ(1-42) showed to be toxic and result in a shorter life-span. Aβ fibrils are a big factor leading to the alzheimers disease. It’s been hypothesized that it is toxic and eventually kills the cells that come in contact by penetrating their membranes. It’s suggested that the activity of these peptides are intracellular rather than extracellular. AB amyloid fibrils are complex units segregate into different populations. To try and understand Alzheimers Disease would mean having to understand the population of the Aβ amyloid fibrils. Doing so will allow researchers and scientist work for the disease treatment
It is often difficult to isolate the Aβ amyloid peptides, thus the amount of information obtained from them is very limited. The full-length structure of the AB amyloid fibrils have yet to be uncovered, even with use of X-ray crystallography. Many other forms of measurements have been used to study the Aβ- amyloids. These include infrared spectroscopy, NMR, mass spectrometry, electron paramagnetic resonance. Unfortunately, the data received are rather indirect. The most direct way would be to use solid state NMR and electros cryomicroscopy (cryo-EM). These allow the distinction of Aβ amyloid fibrils at near-atomic resolution. They give chemical shifts and even the bond angles. From here it allows the researchers to ID the residues and their sheet structure.
There have been many models of the Aβ peptides proposed. But because it must be considered that in different conditions different fibrils can conform, it’s critical to have much caution. The general model of an Aβ fibril is a U-shaped peptide told, refered as a β-arc. [2]
A Molecular Link Between the Active Component of Marijuana and Alzheimer's Disease Pathology
[edit | edit source]Recent studies have shown that the active component of marijuana, Δ9-tetrahydrocannabinol (THC), inhibits AChE-induced β-amyloids aggregation in the pathology of Alzheimer’s disease. Some studies have demonstrated the ability of THC to provide neuroprotection against the toxicity of β-amyloid peptide. One of the causes of Alzheimer’s disease is the deposition of β-amyloid in portions of the brain that are important for memory and cognition. This deposition and formation of a plaque in the brain is caused by enzyme acetylcholinesterase (AChE). AchE is an enzyme that degrades acetylcholine, which in turn increase the amount of neurotransmitters released into the synaptic cleft. It also functions as an allosteric effector that accelerates the formation of amyloid fibrils in the brain. In vitro studies have demonstrated the inhibition of AChE has decreased β-amyloid deposition in the brain, and THC is a very good inhibitor.
THC binding to AChE using AutoDock revealed that THC has a high binding affinity to AChE. Not only do they bind well, interactions were observed between THC and the carbonyl backbone of AChE, residues of Phe123 and Ser125. Furthermore, the ability of THC to inhibit AChE catalytic activities were tested using steady-state kinetic. The results have shown that THC inhibits AChE at a Ki of 10.2 uM. This number is relatively competitive with the current drugs in the market that treat Alzheimer’s disease. While THC shows competitive inhibition relative to the substrate, this does not necessitate a direct interaction between THC and the AChE active site. In fact, enzymes can bind to the PAS allosteric site on AChE while still blocks the entry into the active site of AChE, preventing it from depositing plaque. This why THC serves as an uncompetitive inhibitor of AChE substrate.
References
[edit | edit source]"What is Alzheimer's?." Alzheimer's and Dementia. Alzheimer's Association, 2012. Web. 20 Nov. 2012. <http://www.alz.org/alzheimers_disease_what_is_alzheimers.asp>. Fandrich, Marcus, Schmidt, Matthias, and Nikolaus Griforieff: Trends Biochem Sci. Author Manuscrpt: Recent Progress in Understanding Alzheimer's B-amyloid Structures. 2011 June; 36(6) 338-345.
Sipe, J.D. Amyloidosis. A. Rev. Biochem. 61, 947−76 (1992).
Glenner, G.G. Amyloid deposits and amyloidosis: The -fibrilloses (first of two parts). New. Engl. J. Med. 302, 1283−1292 (1980). | PubMed | ISI | ChemPort |
- ↑ a b Fandrich, Marcus, Matthias Schmidt, Mikolaus Grigorieff (February 2011). "Recent Profress in Understanding Alzheimer's B-amyloid Structures". Trends Biochemistry. 36 (6): 338–45. doi:10.1002/ana.410380312. PMID 7668828.
{{cite journal}}: CS1 maint: multiple names: authors list (link) Invalid<ref>tag; name "pmid7668828" defined multiple times with different content - ↑ Roher AE, Lowenson JD, Clarke S, Woods AS, Cotter RJ, Gowing E, Ball MJ (November 1993). "beta-Amyloid-(1-42) is a major component of cerebrovascular amyloid deposits: implications for the pathology of Alzheimer disease". Proc. Natl. Acad. Sci. U.S.A. 90 (22): 10836–40. Bibcode:1993PNAS...9010836R. doi:10.1073/pnas.90.22.10836. PMC 47873. PMID 8248178.
{{cite journal}}: CS1 maint: multiple names: authors list (link)