Chemical Sciences: A Manual for CSIR-UGC National Eligibility Test for Lectureship and JRF/Named Reactions/Schmidt Rearrangement
| This page was imported and needs to be de-wikified. Books should use wikilinks rather sparsely, and only to reference technical or esoteric terms that are critical to understanding the content. Most if not all wikilinks should simply be removed. Please remove {{dewikify}} after the page is dewikified. |
The Schmidt reaction is an organic reaction involving alkyl migration over the carbon-nitrogen chemical bond in an azide with expulsion of nitrogen.[1] A key reagent introducing this azide group is hydrazoic acid and the reaction product depends on the type of reactant: carboxylic acids form amines through an isocyanate intermediate (1) and ketones form amides (2):
A catalyst is required which can be a protic acid usually sulfuric acid or a Lewis acid. The reaction was discovered in 1924 by Karl Friedrich Schmidt (1887–1971),[2] who successfully converted benzophenone and hydrazoic acid to benzanilide. It is a tool regularly used in organic chemistry for the synthesis of new organic compounds for example in that of the unusual 2-quinuclidone.
Reaction mechanism
[edit | edit source]The carboxylic acid Schmidt reaction starts with acylium ion 1 obtained from protonation and loss of water. Reaction with hydrazoic acid forms the protonated azido ketone 2 which goes through a rearrangement reaction with the alkyl group R migrating over the C-N bond with expulsion of nitrogen. The protonated isocyanate is attacked by water forming carbamate 4 which after deprotonation loses carbon dioxide to the amine.
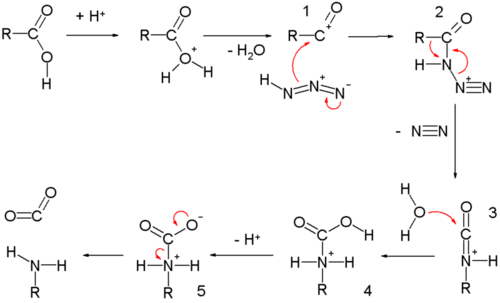
The reaction is related to the Curtius rearrangement except that in this reaction the azide is protonated and hence with different intermediates.
In the reaction mechanism for the ketone Schmidt reaction the carbonyl group is activated by protonation for nucleophilic addition by the azide forming intermediate 3 which loses water in a elimination reaction to temporary imine 4 over which one of the alkyl groups migrates from carbon to nitrogen with loss of nitrogen. A similar migration is found in the Beckmann rearrangement. Attack by water and proton loss converts 5 to 7 which is a tautomer of the final amide.
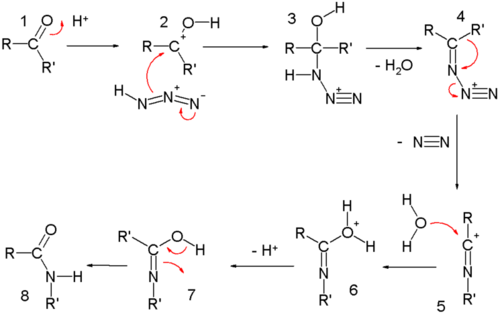
Reactions involving alkyl azides
[edit | edit source]The scope of this reaction has been extended to reactions of carbonyls with alkyl azides R-N3. This extension was first reported by J.H. Boyer in 1955 [3] (hence the name Boyer reaction) for example the reaction of m-nitrobenzaldehyde with β-azido-ethanol:

Variations involving intramolecular Schmidt reactions have been known since 1991.[4] An intramolecular reaction has been applied to the synthesis of novel bicyclic lactams:[5]

References
[edit | edit source]- ↑ Plagens, Andreas; Laue, Thomas M. (2005). Named organic reactions (2nd ed.). Chichester: John Wiley & Sons. ISBN 0-470-01041-X.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ doi:10.1002/cber.19240570423
- ↑ J. H. Boyer and J. Hamer (1955). "The Acid-catalyzed Reaction of Alkyl Azides upon Carbonyl Compounds". J. Am. Chem. Soc. 77 (4): 951–954. doi:10.1021/ja01609a045.
- ↑ Jeffrey Aube and Gregory L. Milligan (1991). "Intramolecular Schmidt reaction of alkyl azides". J. Am. Chem. Soc. 113 (23): 8965–8966. doi:10.1021/ja00023a065.
- ↑ Lei Yao and Jeffrey Aubé (2007). "Cation-pi Control of Regiochemistry of Intramolecular Schmidt Reactions en Route to Bridged Bicyclic Lactams". J. Am. Chem. Soc. (Communication). 129 (10): 2766–2767. doi:10.1021/ja068919r.
{{cite journal}}:|format=requires|url=(help)
Further reading
[edit | edit source]- Scott Grecian and Jeff Aubé (2007), "Synthesis of an N-Substituted Lactam using an Intramolecular Schmidt Reaction", Org. Synth., 84: 347–358
- Jade D. Nelson, Dilip P. Modi, and P. Andrew Evans (2004), "Photo-induced Ring Expansion of 1-Triisopropylsilyloxy-1-azidocyclohexane", Org. Synth.
{{citation}}: CS1 maint: multiple names: authors list (link); Coll. Vol., 10: 207{{citation}}: Missing or empty|title=(help) - Leo A. Paquette (1973), "1,3-Dihydro-3,5,7-trimethyl-2h-azepin-2-one", Org. Synth.; Coll. Vol., 5: 408
{{citation}}: Missing or empty|title=(help)