Structural Biochemistry/Proteins/Purification/Isoelectric Focusing
Isoelectric Focusing[edit | edit source]
Isoelectric point, also called the pI of the protein, is the pH at which the net charge of the protein is zero. Isoelectric focusing is a separation technique which separates peptides according to their isoelectric point, or how acidic and basic their residues are. A gel with a pH gradient is used as the medium. The pH gradient is made by adding polyampholytes, which are multi-charged polymers, with different pI into the gel. Then the sample is put onto the gel and a voltage is applied. The proteins will move along the gel until they reach their isoelectric points. In other words, each protein will move until it reaches a position in the gel at which the pH is equal to the pI of the protein. a protein band that forms at a given pH can then be removed and analyzed further. This process can successfully separate proteins that have a difference in net charge greater than or equal to 1.
Isoelectric point (pI): The pH at which the net charge on the protein is zero. For a protein with many basic amino acids, the pI will be high, while for an acidic protein the pI will be lower.
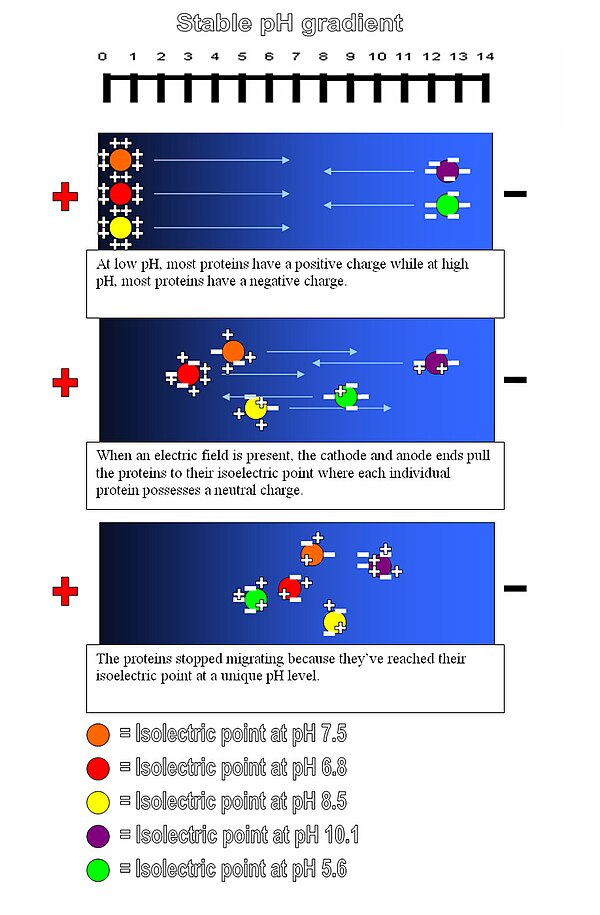
Isoelectric focusing is a type of zone electrophoresis, and it is usually performed in a gel, that takes advantage of the fact that a molecule's charge changes with the pH of its surroundings. A protein that is in a pH region below its isoelectric point (pI) will be positively charged and so will migrate towards the cathode. As it migrates, however, the charge will decrease until the protein reaches the pH region that corresponds to its pI. At this point it has no net charge and so migration ceases. As a result, the proteins become focused into the sharp stationary bands with each protein positioned at a point in the pH gradient corresponding to its pI. This technique is capable of extremely high resolution with proteins differing by a single charge being fractionated into separate bands.
Molecules to be focused are distributed over a medium that has a pH gradient (usually created by aliphatic ampholytes). An electric current is passed through the medium, creating a "positive" anode and "negative" cathode end. The negatively charged molecules migrate through the pH gradient in the medium toward the "positive" end while positively charged molecules move toward the "negative" end. As a particle moves towards the pole opposite of its charge it moves through the changing pH gradient until it reaches a point in which the pH of that molecules isoelectric point is reached. At this point the molecule no longer has a net electric charge (due to the protonation or deprotonation of the associated functional groups) and as such will not proceed any further within the gel. The gradient is initially established before adding the particles of interest by first subjecting a solution of small molecules such as polyampholytes with varying pI values to electrophoresis.
The method is applied in the study of proteins, which separate based on their relative content of acidic and basic residues, whose value is represented by the pI. Proteins are introduced into an immobilized pH gradient gel composed of polyacrylamide, starch, or agarose where a pH gradient has been established. Isoelectric focusing can resolve proteins that differ in pI value by as little as 0.01. Isoelectric focusing is the first step in two-dimensional gel electrophoresis, in which proteins are first separated by their pI and then further separated by molecular weight through SDS PAGE.
How to determine pI of amino acids[edit | edit source]
We can determine pI of each amino acid when we know its pKas by titration with NaOH. For example, glycine, the smallest amino acid, has two pKa values, which are 2.34 and 9.60, respectively.[1]
First, add strong acid and let glycine to become complete protonated form. Then gradually add NaOH until pH raises up to 2.34. At this point, we use 0.5 mol of NaOH equivalent to first protonated form of glycine. Also, There would be 0.5 mol of second protonated form generated in the solution. After using 1 mol NaOH equivalent to first pronated form, there would be solely second pronated form. We'll see that second protonated form of glycine is zwitterion, which is zero net charge molecule. Therefore pH at this point is called isoelectric point (pI) and equals 5.97. Continue adding NaOH once pH equals 9.60. At this point, 0.5 mol of third protonated form is present in the solution and total amount of NaOH is 1.5 mol. Back to pI, we see that
Then we can write in the general form:
Remark To determine pI of amino acid which has more than two pKa, we'll use two pKa values covering the range in which zwitterion would present in the solution.[2]