Structural Biochemistry/Paclitaxel
Overview
[edit | edit source]Paclitaxel (taxol) is an important anti-cancer pharmaceutical drug. In 1967, Monreoe E. Wall and Mansukh C. Wani extracted paclitaxel from the bark of a Pacific yew tree, Taxus brevifolia.
Paclitaxel is used to treat AIDs-related Kaposi sarcoma, breast, ovarian, and lung cancer in UK. This medicine binds to the tubulin and inhibits the disassembly of microtubules. This stabilizes the microtubules and prevents further cell division.
Side Effects
[edit | edit source]Seek medical attention if you have these side effects:
- Hives
- Difficulty breathing
- Feeling like you might pass out
- Swelling of the face, lips, tongue, or throat
- Slow heart rate
- Seizure
- Pale skin, easy bruising or bleeding, unusual weak
- Fever, chills, body aches, flu symptoms
- Sores inside your mouth or on your lips
- Numbness
- Increased blood pressure
- Joint and muscle pain
- Mild nausea, and vomiting
- Hair loss
Semisynthesis
[edit | edit source]
Biosynthesis
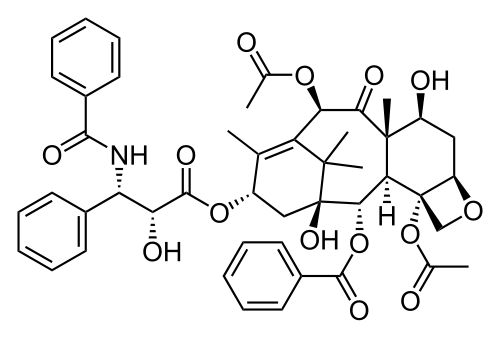
[edit | edit source]Structure
[edit | edit source]The structure of (1S,2S,3R,4S,7R,9S,10S,12R,15S)-4,12-Diacetoxy-15-{[(2R,3S)-3- (benzoylamino)-2-hydroxy-3- phenylpropanoyl]oxy}-1,9- dihydroxy-10,14,17,17-tetramethyl -11-oxo-6-oxatetracyclo [11.3.1.0~3,10~.0~4,7~] heptadec-13-en-2-yl rel-benzoate:
Functional Groups: aromatic(3), amide(1), ketone(2), esters(4), alcohol(3), and ether(5) It has 11 stereocenters.
Mechanism of Actions
[edit | edit source]
Microtubules are cellular components that act as a skeleton for the cell. For cell division to occur, microtubules need to depolymerise back to tubulin. After that, tubulins repolymerise to form the spindle of cell division. The movement of the replicated chromosomes during mitosis depends on the spindle and therefore, the depolymerization of microtubules.
Paclitaxel or Taxol enhances the polymerization of tubulin to stable microtubules and also interacts directly with microtubules, stabilizing them against depolymerization. Hence, it interferes with the spindle formation process. Chromosomes are unable to move to the opposite sides of the dividing cells. Cells division is inhibited and eventually, cell death is induced.
References
[edit | edit source]Borad, Brijesh. "CYTOTOXIC EFFECT OF TAXOL AND VITAMIN E SUCCINATE COMBINATION IN COMPARISON WITH TAXOL ALONE ON MCF-7 BREAST CANCER CELL CULTURE BY MTT ASSAY." PharmaTutor. N.p.. Web. 7 Dec 2012.