Structural Biochemistry/Organic Chemistry/Stereochemistry
Stereochemistry: By definition, stereochemistry is the arrangement of different atoms in space. Stereochemistry is a 3d representation of a carbon that is sp3 hybridized. There are many different types of stereoisomers. Let's first discuss a few basic concepts.
Background
[edit | edit source]Although most people are accustomed to thinking of organic chemistry as a bunch of drawings and structures, this "paper chemistry" is not really how these molecules act in real life. Of course, these molecules are really three-dimensional shapes, and not just 2d drawings. Stereochemistry aims to explain the natural phenomena of spatial arrangements of these organic molecules.

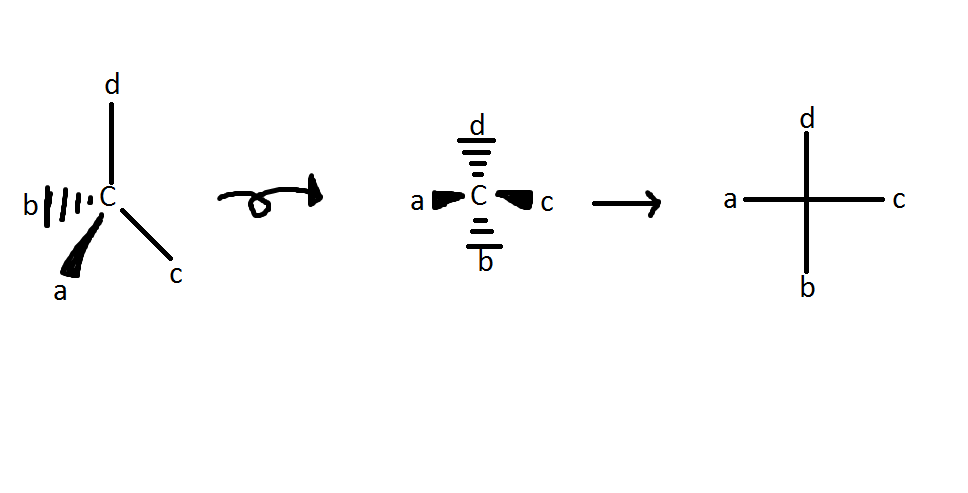
Stereocenter: Any atom in a molecule that is attached to 4 different atoms. Also known as chiral center. A chiral molecule is special in that it is not identical to its mirror image, or in other words, the only criterion for chirality is that the object and its mirror image must be non-superimposable.[1] For example, methane (CH4) is identical to its mirror image; therefore, this molecule is not chiral. In general, molecules with "n" chiral centers have 2^n stereoisomers. For example, a molecule with 3 stereocenters would give rise to a molecule with 8 stereoisomers. Stereoisomers are isomers of molecules with the same formula and connectivity, but with different arrangements of their atoms in space. Two stereoisomers have atoms linked together in the same order, but the two molecules do not have the same three-dimensional shapes. Wedges indicate bonds coming towards the viewer, while dashed lines indicate bonds going away from the viewer.
A molecule classified as achiral either is not sp3 hybridized or the molecule is sp3 hybridized but has two or more substituents that are the same. These molecules are not chiral. An achiral molecule is super-imposable on itself.
A Brief Introduction to Isomers
[edit | edit source]
Molecules that are isomers have the same molecular formulas but their structures are different from each other. There are two main categories of isomers: constitutional isomers and stereoisomers. Constitutional isomers, also known as structural isomers, are molecules that have the same molecular formulas but their connectivity is different. Oftentimes constitutional isomers have very different physical and chemical properties. Stereoisomers can be divided into two sub-categories: enantiomers and diastereomers. Enantiomers are molecules that are mirror images of one another and are not super-imposable. These molecules have the same connectivity but differ in their 3D arrangement of carbon substituents. Oftentimes enantiomers have very similar physical properties but they may differ in chemical properties. Diastereomers are molecules that are not related as mirror images but, like enantiomers, have the same connectivity but differ in their 3D arrangement of carbon substituents.
Types of Stereochemistry
[edit | edit source]Enantiomers
[edit | edit source]Enantiomers are structures that are mirror images of one another that are non-superimposable. Non-superimposable means that no matter what way you rotate it, you will not be able to place it directly on top of the other facing the same way. You can easily demonstrate this with your hands because your hands are also non-superimposable, as you cannot place your hands on top of one another with your thumbs facing in the same direction, while your palms face the same way. Enantiomers have identical physical and chemical properties.

Enantiomers are generally classified with either an R or S configuration. To determine whether a stereocenter is labeled R or S, one must first rank its four substituents by molecular weight. The highest priority substituent (rank 1) will be the substituent atom with the largest molecular weight; conversely, the lowest priority substituent (rank 4) will be the substituent atom with the lowest molecular weight. For this reason, a hydrogen substituent is always given the lowest priority. Once the four substituents have been ranked by priority, a series of rules can be followed to determine the stereocenter's R or S configuration.[1]
Rule 1: The lowest priority substituent is always placed as far away as possible, or in other words, placed into the paper or board.
Rule 2: Once the lowest priority substituent (generally a hydrogen) has been placed "into the paper," there are only two possible arrangements of the remaining three substituents (R and S). Looking down the lowest priority substituent bond with the carbon, if the remaining three substituents are increasing in the clockwise direction (1, 2, 3), the stereocenter is classified as R. If the three substituents are increasing in the counter-clockwise direction, the stereocenter is classified as S.
Rule 3: Point of difference rule. This rule helps determine the priority of substituents that have the same rank when looking at the atoms directly attached to the stereocenter. A simple example is the difference between a methyl and an ethyl substituent. Following the substituents beyond the initial carbon atom, a methyl is attached to three hydrogens, while the ethyl's carbon is attached to two hydrogens and another carbon. This is the point of difference in the substituents. Since the carbon atom in the ethyl group is a higher priority than the 3rd hydrogen in the methyl group, the ethyl substituent is ranked higher in priority than the methyl group. [1]
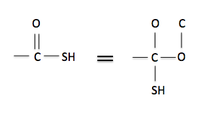
Rule 4: Double and triple bonds can be viewed as if they were single bonds, but with the extra bonds added to each end of the multiple bond as single bonds. This can be seen on the figure to the right. The carbon is attached to a thiol group and is double bonded to an oxygen. This double bond can be seen as the carbon attached to two single bonded oxygens, and the second oxygen is bonded a carbon (representing the double bond to the initial carbon).
Enantiomers are optically active, meaning they have the ability to rotate plane polarized light. A dextrorotatory (+) molecule rotates plane polarized light in a clockwise direction, while a levorotatory molecule rotates plane polarized light in a counterclockwise direction. Enantiomers differ in which direction each compound rotates plane polarized light.

As you can tell from the drawing, on the left, the methyl group is pointing out at us, while the hydroxyl group is pointing back. On the molecule in the right, this order is reversed. This is a prime example of enantiomers.
Confusion with Enantiomers
[edit | edit source]One might think that you can simply just rotate the molecule and they would be exactly the same, but this is not the case. If you rotated the molecule above, the hydroxyl and methyl will be imposed on one another, however, the fluorine and hydrogen will now be on opposite sides. This is what it means to be non-superimposable
Oftentimes there are confusions with meso compounds. For example, say there are two molecules: one characterized with 1R, 2S configurations at its stereocenters (chiral center), the other molecule characterized with 1S, 2R configurations at its respective stereocenters. One might wonder why these molecules aren't enantiomers of each other—after all, they do have opposite configurations at all their respective stereocenters. However, these molecules aren't enantiomers because they are meso compounds. In this situation, each compound has a line of symmetry dividing its two stereocenters. Due to the plane of symmetry of each molecule, each compound is considered achiral (not chiral).
Racemization of Enantiomers
[edit | edit source]Two molecules are considered to be racemic if they consist of the same amount of each enantiomer, a 50:50 ratio of the left-handed, and right handed enantiomer.
A racemic molecule is not shown to rotate plane-polarized light. Individually each enantiomer rotates plane-polarized light, but does so in equal and opposite directions, thus leading to a net rotation of 0.
Optical activity is directly proportional to the ratio of the two enantiomers of a compound. When an equal ratio of the two enantiomers of a compound are present, the sample is considered optically inactive or racemic. If only one of the enantiomers is present, then the sample is considered optically pure. To describe a mixture that falls in-between these two extremes, one can use the enantiomer excess (ee) relation. This is used for when one enantiomer is in excess of the other enantiomer in a mixture.[1] The enantiomer excess equation is:
Enantiomer excess (ee) = % of major enantiomer - % of minor enantiomer
The enantiomer excess equation tells us how much of one enantiomer is in excess of a racemic solution (since in a racemate the ratio of enantiomers is 1:1). The enantiomer excess is also known as optical purity and can be found by the relation:
Enantiomer excess (ee) = optical purity = ([a]Mixture/[a]Pure enantiomer) X 100%
Example
A solution of (+)-alanine from an artifact has a value of [a]Mixture = 3.7; [a]pure enantiomer = 8.5. Find optical purity and actual enantiomer composition of the sample.[1]
Solution
Enantiomer excess = optical purity = (3.7/8.5) X 100% = 43.5 % Therefore, 56.5% of the sample is racemic and 43.5% of the sample is the pure (+) isomer. Thus, it can be concluded that the artifact under examination has an actual composition of 71.75% (+) and 28.25% (-) alanine.
Optical Activity
[edit | edit source]If two enantiomers of a chiral molecule were isolated into two different containers in pure form, it would be extremely difficult to distinguish the two based on just their physical properties such as boiling points, melting points, and densities. Because only certain enantiomers of certain drugs are effective while the other enantiomers are ineffective or even detrimental to the body, it is very important to be able to distinguish enantiomers.
Fortunately chiral molecules possess a unique property that rotates the chiral molecules in a particular direction when a special kind of light called plane-polarized light is passed through a sample containing the enantiomers. This unique reaction that chiral molecules have with plane-polarized light is called optical activity which results in enantiomers often being referred to as optical isomers. The rotation of a particular chiral molecule as seen from the perspective of the viewer facing the light source can either be clockwise, dextrorotatory (“dexter” is Latin for “right”), or counterclockwise, levorotatory (“laevus” is Latin for “left”) [2]. Molecules that rotate clockwise are referred to as (+) enantiomers while molecules that rotate counterclockwise are referred to as (-) enantiomers [2]. The direction of rotation of any particular chiral molecule will always be the same and the enantiomer of that chiral molecule will have an opposite direction of rotation.
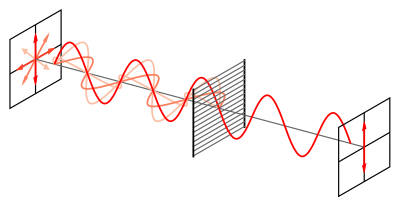
Plane-polarized light result from passing a beam of ordinary light through a special material called a polarizer. A polarizer serves as a filter for the light waves that pass through it and filters out all but one light wave, the plane-polarized light wave [2]. Once the light has been filtered to just the plane-polarized light, the light wave travels through the chiral molecules. The electric field of the light wave interacts with the electrons around the chiral molecule and causes the molecule to rotate at a particular direction; this rotation is called optical rotation [2]. If there is optical rotation in the sample, then the sample is referred to as optically active. To determine if a sample is optically active, optical rotations have to be measured with a polarimeter.
Oftentimes reactions that yield chiral molecules are not stereospecific which may lead to products that have a 1:1 mixture of enantiomers. This reaction mixture is referred to as a racemic mixture because when a plane-polarized light passes through this sample, half of the chiral molecules would be rotating clockwise while the other half would be rotating counterclockwise. This results in a sample with no net optical activity. However, when the ratio of enantiomers is not 1:1, then there will be optical activity. One of the major goals of pharmaceutical companies is to perform reactions that yield optically pure products because this saves them very much time from the purification process of optically impure samples, not to mention money on the reactants used during production.
Stereochemistry of Amino Acids
[edit | edit source]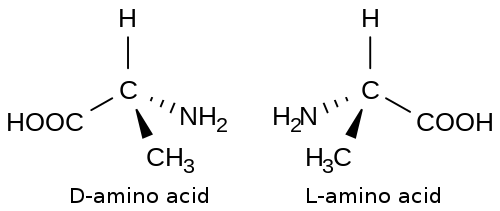
Amino acids are building blocks of protein. The general structure of an amino acid has an sp3 hybridized carbon atom linked to four other substituents. Three of the four substituents attached to the carbon of all amino acids are an amino group and a carboxylic acid group and a hydrogen atom. The last substituent is often represented with an R if the amino acid isn’t specified. R can represent one of the twenty different side chains the twenty different amino acids have.
The stereochemistry of amino acids are represented with an L,D system. The L,D system is essentially the same as the R,S system for absolute configuration with the exception that the L,D system is for amino acids. For the L,D system, L is the equivalent of the S absolute configuration leaving D to be the equivalent of the R absolute configuration.
Amino acids exist dominantly in the L absolute confirmation. There is no conclusive evidence for the dominance of the L isomer over the D isomer at the moment. Many speculate that the L isomer became the dominant amino acid configuration due to chance rather than any physical or chemical property. There have been experiments done trying to elucidate the reason behind the dominance of the L isomer. In the experiments, a protein would be artificially synthesized with only amino acids with the D absolute configuration. The conclusion was that the proteins made of only D amino acids were equally as active as the proteins made of L amino acids with the only difference being that the proteins made of D amino acids had reactions that were the reverse of the proteins made of L amino acids.
Diastereomers
[edit | edit source]Diastereomers are any stereoisomers that are not enantiomers. Thus, diastereomers are not mirror images of each other. Because diastereomers are stereoisomers that aren't mirror images of each other, they can be distinguished by different physical and chemical properties. Thus, it is possible to separate diastereomers by standard laboratory techniques. Diastereomers arise when stereoisomers of a molecule have different configurations at at least one of their stereocenters (but not all, for they would then be enantiomers). Each stereocenter possesses two configurations, so the number of stereoiosmers is increased by a factor of two. Cis/trans isomerism is a form of diastereomerism (cis-two substituents pointing in the same direction; trans-two substituents pointing in opposite directions).
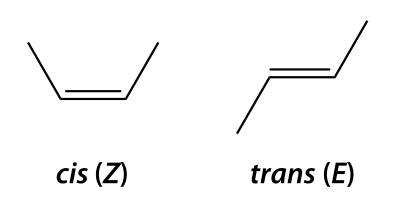

While enantiomers have identical chemical properties, diastereomers do not.
There are 3 stereocenters in the molecule below. The atoms have been switched in the first and third carbon. This is an example of diastereomers. If the second carbons atoms were switched also, then these two molecules would be enantiomers.
Stereochemistry for Alkenes
[edit | edit source]Cis and trans prefixes are essentially absent when naming alkenes with an E,Z system being used instead. The E,Z system sets priorities for carbon substituents the same way as the R,S system with the difference being that the double-bond having no bearing on the priority, only the two substituents that are single-bonded to the carbon. An E isomer (E comes from the German word “entgegen” which means “opposite”) would be where the highest priority substituents on each carbon on the double bond are opposite of each other [1]. A Z isomer (Z comes from the German word “zusammen” which means “together”) would be where the highest priority substituents on each carbon on the double bond are on the same side of each other [2].
Epimers
[edit | edit source]Epimers are diastereomers that differ at only ONE stereocenter.
There are 3 stereocenters in the molecule below. The atoms have been switched at the stereocenter in ONLY the third carbon(on right). This is an example of epimers.

Anomers
[edit | edit source]"Anomers are isomers that differ at a new asymmetric carbon atom formed on ring closure."[2] An example of this is Alpha-D-Glucose and Beta-D-Glucose (shown below).


Fischer Projections
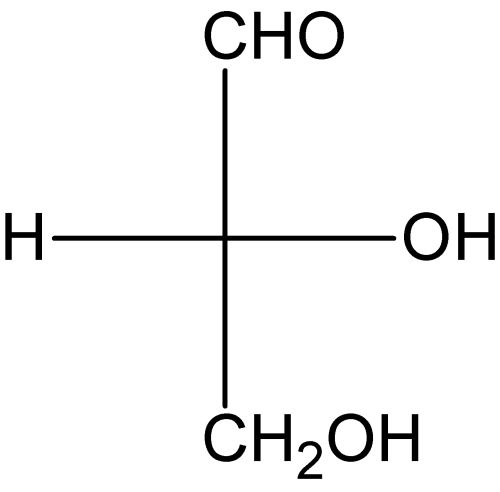
[edit | edit source]In Fischer projections, bonds attaching substituents to the central carbon are shown using horizontal and vertical lines. Similar to the hashed-wedged line structures, horizontal lines are indicative of bonds coming towards the viewer, while vertical lines indicate bonds going away from the viewer. It is a simplified way of showing tetrahedral carbon atoms and their substituents in 2 dimensions. Converting hashed-wedged structures to Fischer projections is a simple process. Here is an example shown below.
Shortcuts for Stereochemistry
[edit | edit source]A simple method to differentiate between an enantiomer and a diastereomer is to look at the molecules being compared and, assuming they both are in fact stereoisomers, look at each stereocenter. If the carbon substituents of each stereocenter have been switched once, then the molecules are related as enantiomers. If not all of the carbon substituents of each stereocenter have been switched once, then the molecules are related as diastereomers. In this case, the switching of carbon substituents means, for example, that a dashed OH substituent and a wedged H substituent of the same carbon switched to a wedged OH substituent and a dashed H substituent while the other two carbon substituents remain in the same positions. By this logic, if the carbon substituents of the stereocenters have been switched twice, then the molecules are related as enantiomers, not diastereomers.
Chiral Drugs
[edit | edit source]Today, the majority of chiral medicines are sold as racemic mixtures. In two enantiomers, there would be one that is inactive. According to Vollhardt[3], the other enantiomer would stop the activity of the other enantiomer by becoming the blocker of the biological receptor site. As a result, the FDA started to produce individual enantiomers of the medicine. Consequently, they began to test the pure enantiomers to help increase the performance of the medicine and it may help to lengthen the patent of medicine. Also, they applied the "chiral switch" method to develop different ways of enantioselective synthesis. "Chiral switch" is the method of switching achiral reactant to a chiral product in a reaction that can be catalyzed by a chiral catalyst. Vollhardt states that such methods have been applied to the production of medicine such as antiarthritic, analgesic naproxen, and antihypertensive propranolol.
References
[edit | edit source]1. Schore, Neil E. (2011). Organic Chemistry Structure and Function 6th edition. W. H. Freeman.
2. Berg, Jeremy Mark, John L. Tymoczko, and Lubert Stryer.Biochemistry. 6th. New York: W H Freeman & Co, 2012.
3. Schore, Neil E. (2007). Organic Chemistry Structure and Function 5th edition. W. H. Freeman.
3. Berg, Jeremy M. (2002). Biochemistry 5th edition. W. H. Freeman.
4. Vollhardt, Peter and Schore, Neil. (2009). Organic Chemistry 9th Edition. W.H. Freeman and Company. ISBN 978-1-4292-0494-1.
- ↑ a b c d e Schore, Neil E. (2011). Organic Chemistry Structure and Function 6th Edition. W. H. Freeman
- ↑ Berg, Jeremy Mark, John L. Tymoczko, and Lubert Stryer.Biochemistry. 6th. New York: W H Freeman & Co, 2012. Print.
- ↑ Vollhardt, Peter and Schore, Neil. (2009). Organic Chemistry 9th Edition. W.H. Freeman and Company. ISBN 978-1-4292-0494-1.