Structural Biochemistry/Membrane Proteins/P-Type ATPases
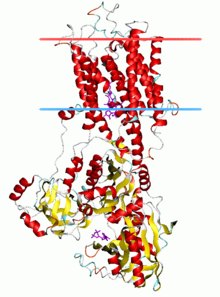
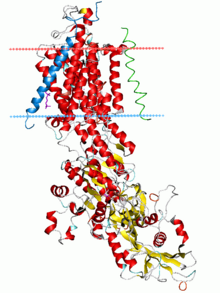
P-type ATPases are a family of cation transport enzymes present in eukaryotes, archea and bacteria. These alpha helical bundle primary transportser (P-type ATPases) are known for catalysis of self phosphorylation of a key conserved aspartate residue within an ion pump. They generate essential ion gradients that are the basis for a variety of cellular functions like signaling, energy storage and secondary transport. Prominent examples of P-type ATPases are the sodium-potassium pump(Na+,K+-ATPase), the proton pump (H+-ATPase), the proton-potassium pump (H+,K+-ATPase) and the calcium pump (Ca2+-ATPase).
Discovered in 1957 by Nobel Laureate Jens Christian Skou, the first P-type ATPase was the Na+, K+ - ATPase. As of June 2007, 400 unique and confirmed members of the P-type ATPase family have been discovered.
These enzymes have a significant impact on cellular function. Indeed, more than one third of the ATP consumed by a resting animal is used by a single ion pump, the Na+,K+-ATPase. The calcium pump (located in the sarcoplasmic reticulum (SR) membrane) also plays a significant role because it is responsible for pumping calcium from the muscle cell to cause a muscle to relax.
P-type ATPases can be divided into five phylogenetic subfamilies:
- Type I — Transition/heavy metal ATPases. These ATPases are involved in transport of metals such as K+, Cu+, Ag+, Cu2+, Zn2+, Cd2+, Pb2+ and Co2+.
- Type II — Includes Ca2+-ases, such as SERCA1a. Also includes Na+/K+ and H+/K+ ATPases.
- Type IIA and IIB transports Ca2+.
- Type IIC contains Na+/K+ and H+/K+ ATPases from animal cells.
- Type IID contains few fungal ATPases of unknown function.
- Type III — Includes plasma membrane H+-ATPases from plants and fungi and Mg2+-ATPases from three bacterial species
- Type IV — Includes ATPases have been shown to be involved in the transport of phospholipids.
- Type V — ATPases with unknown specificity. This large group are only found in eukaryotes and are believed to be involved in cation transport in the endoplasmic reticulum.