Structural Biochemistry/Lipids/Micelles
Structure
[edit | edit source]
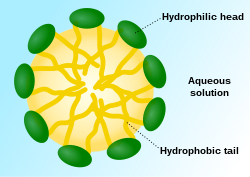
Micelles are lipid molecules that arrange themselves in a spherical form in aqueous solutions. The formation of a micelle is a response to the amphipathic nature of fatty acids, meaning that they contain both hydrophilic regions (polar head groups) as well as hydrophobic regions (the long hydrophobic chain). Micelles contain polar head groups that usually form the outside as the surface of micelles. They face to the water because they are polar. The hydrophobic tails are inside and away from the water since they are nonpolar. Fatty acids from micelles usually have a single hydrocarbon chain as opposed to two hydrocarbon tails. This allows them to conform into a spherical shape for lesser steric hindrance within a fatty acid. Fatty acids from Glycolipids and phospholipids, on the other hand, have two hydrophobic chains that are too bulky to fit into the a spherical shape as micelles do. Thus, they preferred to form glycolipids and phospholipids as "lipid bilayers", which are discussed in the next section.
Micelles form spontaneously in water, as stated above this spontaneous arrangement is due to the amphipatic nature of the molecule. The driving force for this arrangement is the hydrophobic interactions the molecules experience. When the hydrophobic tails are not sequestered from water this results in in the water forming an organized cage around the hydrophobic tail and this entropy is unfavorable. However, when the lipids form micelles the hydrophobic tails interact with each other, and this interaction releases water from the hydrophobic tail and this increases the disorder of the system, and this increase in entropy is favorable.
Bilayer sheet
[edit | edit source]The preferred structure of lipids in aqueous solutions are usually a bilayer sheet of lipids rather than spherical micelles. This is because the two fatty acid chains are too big and bulky to fit into the interior of a micelle. Therefore, micelles usually have one hydrocarbon chain instead of two. Lipid bilayers" form rapidly and spontaneously in an aqueous media and are stabilized by hydrophobic interactions, Van der Waals attractive forces, and electrostatic interactions. The function of the lipid bilayer is to form a barrier between the two sides of the membrane. Due to the fact that the lipid bilayer consists of hydrophobic fatty acid chains, ions and most polar molecules have trouble passing through the bilayer. The one exception to this rule is water because water has a high concentration, small size, and a lack of a complete charge. In order for a molecule to pass through the lipid bilayer it must move from an aqueous environment to a hydrophobic environment and then back into an aqueous environment.Therefore the permeability of small molecules is related to the solubility of said molecule in a nonpolar solvent versus the solubility of the molecule in water.
Micelles can also have a structure that is inside out of its normal structure. Instead of having the hydrocarbon chains inside, they can face outside and while the polar heads are arranged inside the sphere. This happens in a "water in oil" situation because there is so much oil surrounding the drop of water that the hydrocarbon chains face outside instead of inside.
Size
Sizes of micelles range from 2 nm (20 A) to 20 nm (200 A), depending on composition and concentration. The size of a micelle is more limited than that of a lipid bilayer. A lipid bilayer can span up to 107 A or 106 nm.
The lipid bilayer is not a rigid structures, rather they are quite fluid. The individual lipid molecules are able to move or diffuse laterally across the membrane quite easily, this process is called lateral diffusion. However, lipids have much more trouble flipping from one side of the membrane to the other, this process is called traverse diffusion or flip, because this would involve the polar head traveling through the hydrophobic core, and this interaction between polar and hydrophobic regions is unfavorable. So the lipid can move around laterally at a rate of about 2 micrometers per second, while it takes a much longer amount of time to flip flop.
the fluidity of a lipid bilayer also depends on both the temperature and the hydrocarbon chain. As the temperature is increased the fluidity of the lipid bilayer increases as well. Also the more cis double bonds the hydrocarbon tail has the more fluid the structure becomes. This is because when the hydrocarbon tail has cis double bonds it can no longer pack as well as the saturated hydrocarbon tail, so it becomes more fluid. Also the longer the hydrocarbon tail, the higher the transition temperature, which is the temperature at which the bilayer goes from rigid to fluid, this is because longer hydrocarbon tails can interact more strongly than shorter chains.
Formation
[edit | edit source]Micelles form when the polar head and the non polar tails arrange in a special way. They are usually driven to arrange either with the polar heads out (oil in water) or with the polar head in (water in oil). Micelles only form when the concentration of surfactant is greater than the critical micelle concentration (CMC). The surfactant is any surface active material that can part the surface upon entering. The CMC is the concentration above surfactant when micelles will form spontaneously. The higher the concentration, the more micelles there are. Micelle formation also depend on the Krafft temperature. This temperature is when surfactants will form micelles. If the temperature is below the Krafft temperature, then there is no spontaneous formation of micelles. As the temperature increases, the surfactant will turn into a soluble form and be able to form micelles from a crystalline state. The hydrophobic effect is also a driving force that needs to be taken into account. This effect is characterized by the fact that like to form intermolecular aggregates in aqueous substances and in intramolecular molecules. Micelle formation can be summed up by thermodynamics, driven by entropy and enthalpy.
Function and Usage
[edit | edit source]Micelles usually form in soap molecules. Soap often form as micelles because they contain only one hydrocarbon chain instead of two. Therefore they make up the soap property. Micelles act as emulsifiers that allows a compound that is usually insoluble in water to dissolve. Detergents and soap work by inserting the long hydrophobic tails from soap into the insoluble dirt (such as oil) while the hydrophilic head face outside and surround the nonpolar dirt. Then, this micelle can be washed away since the outside of the micelle is soluble with the solvent, which is usually polar. This is the reason why soap helps clean oily and waxy substances off from dishes since water alone cannot pull the oil off.
Micelles are also at work in the human body. Micelles help the body absorb lipid and fat soluble vitamins. They help the small intestine to absorb essential lipids and vitamins from the liver and gall bladder. They also carry complex lipids such as lecithin and lipid soluble vitamins (A, D, E and K) to the small intestine. Without micelles, these vitamins will not be able to be absorbed into the body which will lead to serious complications. Micelles also help clean the skin. Many facial washes use micelles to perform this task. They clean the skin by removing oil and other substances without the need of being washed afterward.
Also, studying membrane proteins often utilize detergents because micelles can isolate, solubilize, and manipulate them [1]
Vesicles
[edit | edit source]

Micelles show up as vesicles in biology. Unlike a micelle, however, vesicles contain a lipid bilayer, which is composed of two layers of phospholipids, arranged end to end with the hydrophobic layered buried between the two layers. A vesicle is a intracellular membrane bound sac that transports and stores substances within the cell. These vesicles store, transport, and digest waste and products from the cell. They can fuse with the plasma membrane to release things from the cell or come into the cell and put things in. Vesicles are important since they play a role in metabolism, transport, enzyme storage, and are chemical reaction chambers.

The picture above shows how liposomes are formed. The vesicles trap the glycine after sonication. Sonication disperses the phospholipids into equal size vesicles of about 500 A or 50 nm diameter sizes. The phospholipids form vesicles around the many molecules of glycine floating around. This is driven by the hydrophobic forces. After gel filtration, the vesicles are then separated from the rest of the glycine floating around. The function of this can be transport or storage of glycine to the appropriate targets. An enlarged view shows the single strand micelles around the hydrophobic glycine (Note that vesicles are, by definition, surrounded by a lipid bilayer so the image showing a monolayer of fatty acids or micelle surrounding the glycine is incorrect! Liposomes are vesicles, not micelles). The tails are inside with the glycine because they are hydrophobic while the heads face the outside which is surrounded by water.
Reference
[edit | edit source]Biochemistry, Berg


