Structural Biochemistry/Chemical Bonding/Dipole interaction
Nature of Dipole Bonding
[edit | edit source]
The nature of dipole bonding begins when atoms differ in their electronegativity, which quantifies an individual atom's ability to attract electrons to itself. A classic example of an electronegative atom is fluorine.
When a covalent bond forms between two atoms, the electrons will be distributed between the two atoms unequally; the more electronegative atoms will have the larger electron density. This unequal sharing of electrons creates a charge separation, and the molecule under inspection will develop partial charges where the electronegative atom will develop a partial negative charge and its adjacent atom will develop a partial positive charge. The molecule is then said to be polarized due to this charge separation. When molecules exhibit this charge separation, there is a pseudo-electrostatic force between the partial charges of molecules.
The key to dipole bonding is charge separation within a molecule.
Permanent dipole-dipole interactions
[edit | edit source]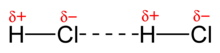
- Dipole moment
- When the covalent bonds in a molecule are polarized so that one portion of the molecule experiences a positive charge and the other portion of the molecule experiences a negative charge. This separation of opposite charges creates an electric dipole. Depending on the orientation of two dipoles, molecules can be attracted to each other as the partial negative charge is attracted to the partial positive charge.
- Ex. Carbon Monoxide (CO)
The permanent and induced dipole interaction of nonpolar molecules bonded to nonpolar molecules in polar molecules, which forms more stable structure.
Besides, when the polar molecules lie near one another, as in liquids and solids, their partial charges act as tiny electric fields that orient them and give rise to dipole-dipole forces. So, the dipole-dipole forces is the positive pole of one molecule that attracts the negative pole of another. The greater the dipole moment, the greater the dipole-dipole forces between the molecules are.
Induced dipole interactions
[edit | edit source]Induced dipoles occur due to the fact that electrons are in constant motion within a molecule. For nonpolar molecules, the averaged charge distribution within a molecule would suggest that there is no permanent dipole. However, instantaneous charge distributions indicate the presence of a transient dipole. Imagine that if you take a photograph of a molecule, you can see an electron and a nucleus in a fixed arrangement. This fixed arrangement implies that there is a dipole moment, if only for a brief moment. We call this type of instantaneous dipole an induced dipole. Brief partial negative charges will be attracted to brief partial positive charges.
Induced dipoles are typical of nonpolar molecules such as fatty acid chains, aliphatic chains, and aromatic hydrocarbons.
Hydrophobic interactions
[edit | edit source]- Hydrophobic effect
- When nonpolar molecules clump together when surrounded by polar molecules.
- The nonpolar molecules are not held together by bonds which is why it is called an instantaneous dipole-induced dipole interaction. This phenomenon is due to the reduction of the number of water molecules surrounding the hydrophobic portion of molecules.
- clathrate
- The structure that is formed when hydrophobic interactions take place (when the hydrophilic molecules line the outside of the hydrophobic molecules). This forces the hydrophobic molecules to become highly ordered
- Example: When water molecules surround lipids