Structural Biochemistry/Carbohydrates/Disaccharides
In other words, disaccharides are composed by 2 sugar molecules. They are called polysaccharides.
General information
[edit | edit source]Disaccharides, the simplest polysaccharides, are the products of a condensation reaction between two monosaccharides. Disaccharide is one of four groups of Carbohydrates (monosaccharide, disaccharide, polysaccharide, and oligosaccharide).

Formation
[edit | edit source]Disaccharides are formed when two monosaccharides join together by the dehydration synthesis reaction resulting in a glycosidic bond between the two monosaccharide molecules. The reaction produces water as a side product. The glycosidic bond in the picture below is a α-glycosidic bond because the bond is formed on the side opposite of the -CH2OH group.
Reducing Sugars
[edit | edit source]Most disaccharides are hemiacetals. Hemiacetals contain a free aldehyde to be oxidized into carboxylic acid. These are classified as reducing sugar. For example: maltose, lactose.
Carbohydrates that are acetals are not oxidized because both of its anomeric carbon atoms are fixed in a glycosidic bond. These are classified as non-reducing sugar. For example: Sucrose.

Classification
[edit | edit source]
There are two basic types of disaccharides: reducing disaccharides, in which disaccharides are Hemiacetals and contain a reactive carbonyl group, they are readily oxidized to diverse products.
Non-reducing disaccharides, in which the sugar is an acetals (or ketals) that cannot readily oxidized because both anomeric carbon atoms are fixed in a glycosidic linkage in which the components bond through their anomeric centers.
Properties
[edit | edit source]The glycosidic bond can form between hydroxyl groups on the two monosaccharides. Due to the different hydroxyl groups that bond, along with the alpha(α) or beta(β) position of the anomeric carbon, there are resulting disaccharides that are diastereomers differing in chemical and physical properties, depending on the monosaccharide components. The α-glucoside is more stable than β-glucoside due to anomeric effect. The C-R bond has a δ* antibonding orbital. If the C-R bond is in axial position, the antibonding δ* overlaps with one of the orbital of the oxygen, which stabilize the molecule. If the C-R bond is in equatorial position, there is no overlap between orbitals making the β-glucoside less stable than the α-glucoside.
Common disaccharides
[edit | edit source]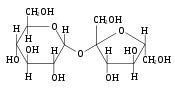


The most common disaccharides are Sucrose, Lactose, and Maltose.
Sucrose is the sugar often found in the grocery store and is produced by plants. It is a sugar derived from fructose and glucose. It is obtained from cane as a transport form of carbohydrates.
Lactose, found in milk, is formed by connecting β-D-galactose and α-D-glucose with a β-1,4-glycosidic bond.
Maltose is created by condensation reaction of the two glucoses, forming a α-1,4-O-glycosidic linkage. It is the second member of an important biochemical series of glucose chains. Maltose can be broken down into two glucose molecules by hydrolysis. In living organisms, the enzyme maltase can achieve this very rapidly.
Lactulose
[edit | edit source]galactose + fructose
Lactulose is a synthetic (man-made) sugar that is not absorbed by the body but is broken down in the colon into products that absorb water into the colon, thus softening stools.
Its primary use is to treat constipation. It is also used to reduce blood ammonia levels in persons with liver disease since lactulose absorbs ammonia into the colon (removing it from the body).
Trehalose
[edit | edit source]glucose + glucose
Trehalose is also known as tremalose or mycose. It is a natural alpha-linked disaccharide with extremely high water retention properties.
In nature, it helps plants and animals reduce long periods without water.
Cellobiose
[edit | edit source]glucose + glucose
Cellobiose is a hydrolysis product of cellulose or cellulose-rich materials, such as paper or cotton. It is formed by linking two beta-glucose molecules by a β(1→4) bond.
Table of Common Disaccharides
[edit | edit source]Here's a quick summary of the subunits of common disaccharides and how they are linked to each other.
| Dissacharide | First Unit | Second Unit | Bond |
|---|---|---|---|
| sucrose | glucose | fructose | α(1→2)β |
| lactulose | galactose | fructose | β(1→4) |
| lactose | galactose | glucose | β(1→4) |
| maltose | glucose | glucose | α(1→4) |
| trehalose | glucose | glucose | α(1→1)α |
| cellobiose | glucose | glucose | β(1→4) |
| chitobiose | glucosamine | glucosamine | β(1→4) |
There are many other disaccharides, although they are not as common, including isomaltose (2 glucose monomers), turanose (a glucose and a fructose monomer), melibiose (a galactose and a glucose monomer), xylobiose (two xylopyranose monomers), sophorose (2 glucose monomers), and mannobiose (2 mannose monomers).
