Structural Biochemistry/Alcohol
Definition
[edit | edit source]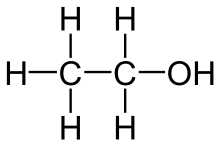
Alcohol is a psychoactive drug/beverage. It is the second most used psychoactive drug. (Caffeine is rated as the first most used.) It is considered a depressant because at medium to high concentrations, it depresses neural firing. (Though at low levels, it can have the opposite effect.)[6] It contains no vitamin or minerals. However, it does contain calories. An important thing to note is that this type of alcohol should not be generalized to mean the same as the organic chemistry term alcohol. Rather this type of alcohol, in terms of organic chemistry, is actually ethanol (or ethyl alcohol). Its chemical formula is: CH3CH2OH.
Due to its small and amphiphilic nature, ethanol is soluble in water and fat, so it is able to cross cell membranes, giving it great access to do harm to cells and affecting the entire body.[6]
History
[edit | edit source]By the tenth-century, a Persian alchemist named al-Razi discovered the first alcohol. Today this alcohol is known as ethyl alcohol. Originally, the name kuhl or kohl was given to the very fine powder that was produced through a sublimation of the natural mineral stibnite to form a compound called antimony sulfide. Today, this product is used as eyeliner and cosmetic.
In the 16th century, the word "alcohol" appears in English that means a very fine powder. In his 1657 Lexicon Chymicum, William Johnson conveys the word as antimonium sive stibium. Through time, the word came to mean any fluid obtained through a process of distillation. This type of fluid includes that of alcohol of wine. In the year 1594, Libavius in Alchymia referred to alcohol as vini alcohol vel cinum alcalisatum. This word in the end was found to mean as "spirit of wine". Until the 18th century "spirit of wine" is referred to as ethanol, and was expanded to be called as "alcohols" in the world of today.
Absorption and Distribution through the body
[edit | edit source]Unlike other drugs, ethanol does not require digestion. The body can quickly absorb it into the system. Roughly 1/5 of the alcohol is absorbed directly through the walls the stomach. Next, the upper portion of the empty stomach absorbs it. Alcohol can also be absorbed through the lungs. Within 30-90 minutes, the body is able to reach the maximal blood concentration from the alcoholic beverage.
Blood Alcohol Content
[edit | edit source]Blood alcohol content (or commonly abbreviated BAC) is a measurement of the amount of alcohol in a person's blood. More specifically, it a measure of how many grams of alcohol in 100 milliliters of a person's blood. For example, if a person registers a .20 BAC, there is 1/5th of a gram of alcohol in every 100 milliliters of that person's blood.
Zero Order Kinetics (Elimination of Alcohol from the body)
[edit | edit source]Alcohol is mainly eliminated from the body through the metabolism. This occurs mainly from the liver. A small percent (5-10%) is eliminated directly through urine and breathing. Elimination of alcohol is considered Zero Order Kinetics. This means that the amount of alcohol removed is constant over time and does not depend on the concentration intake. This is unlike many other drugs that have half-lives and depend on the amount in the body. Alcohol is constantly eliminated over time. The liver can eliminate roughly .25 ounces per hour. It takes approximately two hours to completely eliminate one (Standard) drink.
Addiction
[edit | edit source]Alcohol increases the release of dopamine in the nucleus accumbens. Dopamine is responsible for movement, attention, learning, mood, etc. Dopamine also takes a part in the positive reinforcement (reward) system of the brain. It also triggers the release of endogenous opioids. Opiate receptors also seem to be involved in the reward system of the brain. Withdrawal effects are very powerful, (i.e. seizures, increased sensitivity of NMDA receptors). Alcohol's neurological influence on the reward system of the brain is what's primarily responsible for a person's physiological addiction to alcohol.
Toxic
[edit | edit source]Since the prehistoric times, a compound known as ethanol that is commonly seen in alcoholic beverages has been consumed by human beings for many centuries. The reason for this intake is due to dietary, hygienic, medicinal, religious, and recreational reasons (special event taken place in life). The idea of intoxication is the taking in large doses of ethanol that cause drunkenness. Drunkenness that is often seen when people drink heavily can lead to a hangover as its effect wears off after a period of time. It is important to know that ethanol can cause serious respiratory failure or even death depending on how much an individual take in to their body.
Primary and secondary metabolite, acetaldehyde, and acetic acid all cause the toxicity of ethanol. It is said that all primary alcohols are broken down into aldehydes. After that it is broken down to carboxylic acids which have toxicities that is similar to that of acetic acid and acetaldehyde. Metabolite toxicity is reduced in rats that are fed with acetic acid such as thiamine.
In addition, some secondary and tertiary alcohols are not as poisonous as they are known for. In other words, these alcohols are less poisonous than that of ethanol because the liver is unable to metabolize them into these toxic by-products. Therefore, they are better for medicinal and recreational use. A good example of a tertiary alcohol would be Ethchlorvynol. Ethchlorvynol is use for both medicinal and recreational use.
However, there are also many other alcohols that are more poisonous than that of ethanol. The reason for this is because these alcohols take a longer time to metabolize and their substance that they produce is even more toxic than ethanol. A type of wood alcohol is methanol. Methanol is oxidized to formaldehyde and then through formaldehyde dehydrogenase and alcohol dehydrogenase enzymes to the poisonous formic acid in the liver. The bad thing about this is that, this can cause blindness or even death. Administer ethanol is a great way to prevent toxicity after ingestion such as ethylene glycol or methanol. Although methanol is seen as poisonous, it has a much weaker sedative effect than that of ethanol.
Typically, long chain such as isopropanol, n-butanol or n-propanol have strong sedative effects. Their toxicity is also much higher than that of ethanol. These long chains are also known as fusel alcohols and are surprisingly found to be contaiminanting to certain alcoholic beverages. Thus, this is the reason why people who drink excess or large amount of alcoholic beverage will encounter a kind of hangover. Overall, many of these long chain and even longer chains are used in the industry today as a kind of solvent and are used in alcohol that causes all kinds of serious health conditions.
Withdrawal
[edit | edit source]After a cycle of addiction, withdrawal may come after if the amount of alcohol intake is decreased or stopped. Withdrawal symptoms generally occur 24-48 hours after stoppage of intake. It can lead to symptoms that include insomnia, hyper excitability, and tremors. Furthermore, it can lead to changes in the sympathetic system that include: increased heart rate, increased blood pressure, and increased body temperature. After these early withdrawal symptoms occur, later symptoms (usually 2-4 days after) can occur. These include fits of delirium tremors, hallucinations, and high fevers. Aside from physical changes, alcohol withdrawal can lead to anxiety and dysphoria (negative mood state). One of the main treatments for withdrawal is a Benzodiazepine drug called Chlorodiazepozide(Librium). It is a sedative/hypnotic drug that helps to counteract the affects of alcohol withdrawal.
Blood Alcohol Content
[edit | edit source]Blood alcohol content (or commonly abbreviated BAC) is a measurement of the amount of alcohol in a person's blood. More specifically, it a measure of how many grams of alcohol in 100 milliliters of a person's blood. For example, if a person registers a .20 BAC, there is 1/5th of a gram of alcohol in every 100 milliliters of that person's blood.
Zero Order Kinetics (Elimination of Alcohol from the body)
[edit | edit source]Alcohol is mainly eliminated from the body through the metabolism. This occurs mainly from the liver. A small percent (5-10%) is eliminated directly through urine and breathing. Elimination of alcohol is considered Zero Order Kinetics. This means that the amount of alcohol removed is constant over time and does not depend on the concentration intake. This is unlike many other drugs that have half-lives and depend on the amount in the body. Alcohol is constantly eliminated over time. The liver can eliminate roughly .25 ounces per hour. It takes approximately two hours to completely eliminate one (Standard) drink.
General Effects
[edit | edit source]Alcohol causes a loss of inhibition and judgment, which can lead to aggressive behavior. Alcohol also causes impairment of the senses and can lead to liver and heart problems.
Neurological Effects
[edit | edit source]Alcohol interferes with neural adhesion protein-protein that helps to guide growth of neurons in developing brain. Alcohol is an agonist of GABA receptors and antagonist of NMDA receptors. The GABA (gamma-aminobutyric acid) neurotransmitter is responsible for inhibition in the brain and GABA receptors activate chloride ion channels. Glutamate is the normal neurotransmitter for the NMDA receptors, but NMDA can also activate these receptors in a similar manner. However, NMDA does not bind to other glutamate receptors. NMDA is an excitatory neurotransmitter that is responsible for learning and memory. NMDA receptors activate ion channels that allow the influx of sodium and calcium ions.
Diseases
[edit | edit source]Some diseases associated with drinking alcohol are Korsakoff’s Syndrome and Fetal Alcohol Syndrome. Korsakoff’s Syndrome is characterized by a deficiency in thiamine (Vitamin B1), which leads to damage in the mammillary bodies in the hypothalamus and hippocampus.
Fetal alcohol syndrome is caused when a woman drinks alcohol during pregnancy and the alcohol impairs the baby’s development. Two traits that are common in babies with fetal alcohol syndrome physical deformity and mental retardation. It causes mild to moderate retardation. It can also lead to hyperactivity in childhood as well as growth deficiency. This syndrome is also has characteristic facial abnormalities. Ethanol's affects on fetal development include, but are not limited to, disruption of production of cell-adhesion molecules, druption of normal apoptosis (programmed cell death), and disruption of neurotrophic support.[6]
Alcohol Dehydrogenase
[edit | edit source]Alcohol dehydrogenase is the primary enzyme that converts alcohol into acetaldehyde. This enzyme is part of the primary metabolic system for alcohol. The activity of alcohol dehydrogenase determines the rate of alcohol metabolism. Therefore, myths such as exercise or coffee consumption do not play a role in speeding up the metabolism system. [5]
Having a deficiency in alcohol dehydrogenase decreases the speed of the metabolic process, therefore taking longer to break down alcohol. There is evidence that women have less alcohol dehydrogenase enzymes than men. There has been some evidence that fluctuations in women's menstrual cycles, gonadal hormone levels can influence the rate of their alcohol metabolic system. Women may have elevated blood alcohol concentrations at different times throught their cycle.[7]
Acetaldehyde Dehydrogenase
[edit | edit source]
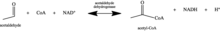
Acetaldehyde Dehydrogenase (ALDH) are dehydrogenase enzymes that help convert acetyldehyde into acetic acid. More specifically, this enzyme is present in the metabolism and helps to break down alcohol. When a person drinks, the alcohol is first converted to acetaldehyde by the alcohol dehydrogenase enzyme. Acetaldehyde dehydrogenase is needed to further break the new acetyldehyde into more harmless acetic acid.
If an individual has a deficiency in acetaldehyde dehydrogenase, this breakdown cannot occur, leading to elevated acetaldehyde levels which results in vomiting, hyperventilation, nausea, increased heart rate, and alcohol flush reaction. It has been found that people of East Asian descent usually have genes that code for an inactive variant of acetaldehyde dehydrogenase 2.[2]
References
[edit | edit source]- “Alcohol: Balancing Risks and Benefits.” Harvard School of Public Health (2010). n. pag. Web. 22 May 2010.
- "Alcohol Flush Signals Increased Cancer Risk among East Asians." National Institutes of Health. 23 March 2009. http://www.nih.gov/news/health/mar2009/niaaa-23.htm.
- “Alcoholic Beverage." Wikipedia, The Free Encyclopedia. Wikimedia Foundation, Inc. 21 May 2010. Web. 22 May. 2010.
- Carlson, Neil R. Physiology of Behavior. Boston: Pearson Education, Inc., 2007.
- Hart, Carl. Drugs, Society, and Human Behavior. 13th. McGraw-Hill Humanities, 2008. Print.
- Pinel, John. "Biopsychology." Pearson, 7th Edition, 2009. Print.
- "Alcohol and Women" National Institute of Health. Oct. 1990. http://pubs.niaaa.nih.gov/publications/aa10.htm
- http://en.wikipedia.org/wiki/Alcohol#History_and_etymology
