Biochemistry/Electron Transport Chain
The electron transport chain is a system of molecules through which electrons are transferred to generate ATP. It has an important role in both photosynthesis and cellular respiration.
Oxidative phosphorylation[edit | edit source]
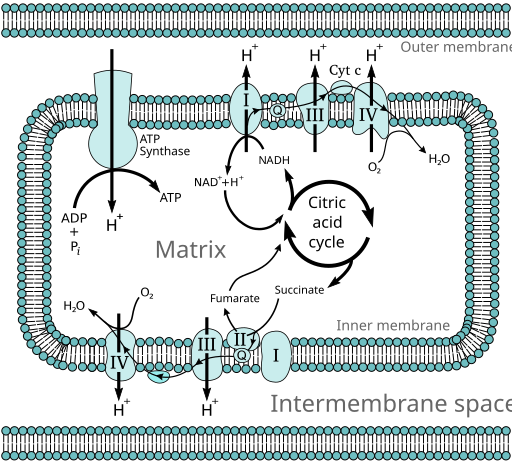
Mitochondria is a double membraned organelle where most of the ATP, of the cell, is harvested by means of oxidative phosphorylation. The outer membrane of the mitochondria is highly permeable to most of the small molecules (<500 g/mol) and ions, as opposed to inner one. The inner membrane consists of folds, which have special name cristae, providing more surface area for ETC (electron transfer chain) elements. The matrix contains pyruvate dehydrogenase complex alongside citric acid cycle enzymes, fatty acids beta-oxidation enzymes and amino acid oxidation elements. Thus, except glycolysis happens at cytoplasm, oxidation of most of the fuels occurs at mitochondrial matrix. Therefore, most of the electrons transferred to electron carriers such as NAD and FAD directly available for processing of ETC. Electron transfer to NAD and NADPH is achieved by the help of Nicotinamide Nucleotide-Linked Dehydrogenases. Reactions catalyzed via Nicotinamide Nucleotide-Linked Dehydrogenases can be processed to reverse direction, by again by these enzyme.
Standard reduction potential value implies that how much a molecule is disposed to be reduced. The greater value infers the greater tendency to be reduced.
For NAD and NADP standard reduction potential is majorly determined molecules themselves. FAD is similar to NAD and NADP however it differs at its relation with its linked protein. NAD and NADP are water-soluble after reaction occurs NAD and NADPH dissociates from the enzyme. As segregate from NAD and NADP, FAD found as bound to the enzyme, meaning it is a prosthetic group. Because of this permanent link, standard reduction potential of FAD is decided by the enzyme FAD associated.
There are differences between NAD and NADPH, too. After electron acceptance, NADH is directed in order to enter ETC whereas NADPH contributes anabolic reactions. In addition to this, NADP and NAD do not stored at the same place, instead they deposit at different parts of the cells.
Electrons carried via NADH and FADH2 are transported to complexes some of which pumps H+ from matrix to intermembrane space. But passage between complexes also requires some electron carriers. These carriers are respectively Ubiquinone (Coenzyme Q, shortly just Q), Iron sulphur proteins and cytochromes.
There are four unique electron carrier complexes in the respiratory chain:
- Complex I: NADH dehydrogenase
- Composed of 42 different polypeptide chains.
- Has several Fe-S centers.
- NADH + H + FMN <---> NAD + FMNH2
- Transfers 2 electrons from NADH to Q.
- Pumps 4 protons from matrix to IMS.
- Complex II: Succinate Dehydrogense
- Transfers 2 electrons from succinate to coenzyme Q.
- Contains 4 different protein subunits:
- Subunit A: FAD, succinate binding
- Subunit B: 3 Fe-S centers
- Subunit C: integral membrane protein, Q binding
- Subunit D: does not participate in electron transfer to Q, instead reduces the frequency of some leaking electrons form reactive oxygen Species: ROS.
- Complex III: Ubiquinone Cytochrome C Oxidoreductase
- Composed of 22 subunits: heme groups and Fe-S centers
- Transfers of 2 electrons QH2 to Cytochrome C.
- Pumps 4 protons into IMS.
- Complex IV: Cytochrome C Oxidase
- Contains 13 subunits, prosthetic groups such as cytochromes and Fe-S centers.
- Transfers 2 electrons from cytochrome C to oxygen, reducing it to H2O.
- Pumps 2 protons into IMS
By the pumping electrons to IMS, proton motive force is generated. Then, via the backflow of hydrogen ions into matrix (mediated by ATP synthase) phosphorylation of ADP to ATP occurs.
For oxidative phosphorylation, oxidation and phosphorylation are said to be coupled. the base idea for this is the event that if one of them does not function, other one cannot proceed. Oxidation of electron carriers provides proton passage form matrix to IMS, thus promotes the activity of ATP synthase. ATP synthase, in concaminant to ATP production, replenishes matrix protons by pumping electrons from IMS to matrix. In conclusion ETC and ATP synthase promotes their action, in relation looks like mutualism.
ETC in photosynthesis[edit | edit source]
In photosynthesis, when light is absorbed in photosystem 2, electrons are energized. They are transferred to the reaction center. From the reaction center, the electrons enter the electron transport chain and pass the etransport[check spelling] chain molecules. Then the 2 deenergized electrons are reenergized in Photosystem 1 (is second because PSII was discovered first) and they go to the NADP+ reductase which transfers their electrons to a coenzyme, converting it to NADPH+ The two electrons used must be replaced, so water is broken down producing 2 protons (H+) which concentrate in the thylakoid membrane, 2 electrons replaced in PSII and oxygen released as O2. Protons go down proton pumps and a concentration gradient forms as protons move from the stroma into the thylakoid space. Protons move down gradient through ATP synthase which forms ATP. Energy is stored.