Sensory Systems/General Features/Association Cortex
The Role of the Association Cortices in Sensory Processing
[edit | edit source]Introduction
[edit | edit source]The sensory systems described in the Human Anatomy and Physiology section of this Wikibook perform specific functions within the nervous system. However, an understanding of these systems does not paint the whole picture of what enables our species to make decisions that increase its chance of survival.
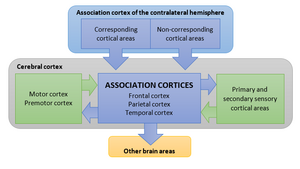
To manage the many elaborate signals from its sensory subsystems, the nervous system contains various higher-order processes. These are responsible for more complex processing of sensory stimuli, and for generating behaviour. Collectively, these higher-order brain areas are called the association cortices, and comprise the greater part of the neocortex. The wide range of functions performed by the association cortices is often referred to as “cognition”: the “ability to attend to external stimuli or internal motivation; to identify the significance of such stimuli; and to make appropriate responses”[1].
Function of the Association Cortices
[edit | edit source]Our knowledge of the role of the human association cortices is fairly limited. Most inferences about their function were derived from observations of patients with cortical lesions. These inferences were subsequently verified by observing patterns of neural activity in homologous regions in the brains of experimental animals and humans [1]. Over the last several decades, the development of technologies such as PET (positron emission tomography), MRI (magnetic resonance imaging), EEG (electroencephalography), TMS (transcranial magnetic stimulation), TES (transcranial electrical stimulation), MEG (magnetoencephalography) and NIRS (near infrared spectroscopy) has significantly advanced our understanding of the neural basis of cognitive control [2].
The parietal association cortex governs attention and perceptual awareness. Patients with parietal lobe lesions experience an inability to attend to objects in a portion of space, even though their visual, somatosensory and motor systems are intact. This condition is known as contralateral neglect syndrome [1]. The parietal cortex, and more specifically, the inferior parietal lobe, is the main (but not the only) part of the brain mediating attention [1].
The temporal association cortex is responsible for the recognition and identification of stimuli. Unlike patients with neglect syndromes, patients with damage to the temporal cortex are aware of objects on the side contralateral to the lesion, but experience difficulty recognising and naming them [1]. These disorders are collectively known as agnosias. Damage to a particular part of the inferior temporal cortex results in an inability to identify faces, a condition known as prosopagnosia [1].
The frontal association cortex is responsible for planning and decision making. It governs a wider range of functions than any other neocortical region [1], and integrates information from sensory and motor cortices, as well as from the parietal and temporal association cortices [1]. Within the human frontal cortex is the brain structure that evolved the most from our ancestors [2]: the prefrontal cortex. The prefrontal cortex plays a critical role in cognitive control [2], and governs what we call an individual’s “personality” [1].
Function of the Prefrontal Cortex
[edit | edit source]A famous medical case in the history of neuroscience first drew attention to the function of the prefrontal cortex: in 1848, Phineas Gage, a railroad construction foreman, had an iron rod – about 1 meter in length and 5 centimetres in diameter – propelled through his head in a work accident [2]. Gage did not even lose consciousness, but sustained severe damage to his frontal cortex. Regions of the frontal lobe involved in motor control, eye movements and language remained intact, and Gage did not experience impairments in his basic function for the remaining eleven years of his life following the accident [2]. However, Gage’s prefrontal cortex was severely damaged, resulting in dramatic changes to his personality.
According to the reports of his doctor and friends, Gage became irrational and immature in his behaviour, and was incapable of controlling his impulses or planning for the future [2]. Similar symptoms were also reported for patients who underwent frontal lobotomies, a medical procedure performed in the twentieth century on patients suffering from various psychiatric conditions, in which the prefrontal cortex was purposefully destroyed [2].
Although these cases revealed the role of the prefrontal cortex in guiding human behaviours, its executive function and importance in cognitive control only became clear in the mid-twentieth century as a result of neuropsychological testing [2].
We now know that the prefrontal cortex interacts with many other brain regions across an intricate neural network [2]. The prefrontal cortex integrates and influences information from sensory inputs, internal states and motor outputs [3], and can thus be regarded as the brain’s control centre.

Perception-Action Cycle
[edit | edit source]The prefrontal cortex modulates sensory inputs by creating internal representations of visual, auditory, tactile and olfactory information [3]. These cortical representations of the external world are referred to as perceptions [3].
Motor outputs, such as controlled bodily movements and speech, are also the result of prefrontal cortex modulation [2]. Furthermore, the prefrontal cortex regulates thoughts and emotions [3]. Prefrontal cortex functions thus form a feedback loop between the brain and the environment. This two-way communication is termed the perception-action cycle [2].
Feedback loops already existed in the brains of our species’ recent ancestors (and in fact still exist in many more primitive brains). However, these were reflexive, bottom-up loops. An example of a bottom-up perception-action cycle that has remained in humans is the ‘knee jerk’ reflex [2]. A tap to one’s knee triggers an abrupt, automatic leg movement. Reflexive perception-action cycles like the knee-jerk reflex occur without evaluation and decision making [2].
The perception-action cycle governed by the prefrontal cortex, by contrast, involves top-down processing. This is a result of a short pause in the cycle, between perception and action. The executive (or higher-order) cognitive functions are engaged during this delay period [2]. The evolution of this delay period between perception and action coincided with the development of the prefrontal cortex in the human brain [4]. The pause enables selective, top-down processing [5], and as a result, more complex interactions with the environment.
Selective attention allows us to focus on specific aspects of the environment in order to pursue a goal, while at the same time suppressing or ignoring irrelevant sensory inputs [6]. The ability to maintain task-relevant information in mind for brief periods of time, to guide subsequent behaviour, is known as working memory [6]. Working memory is believed to link perception and future action through the expectation of upcoming events [2].
Current research suggests a significant amount of overlap between selective attention and working memory, with the prefrontal cortex mediating both of these cognitive processes [6].

Cognitive Control
[edit | edit source]In governing the perception-action cycle, the prefrontal cortex plays a critical part in cognitive control. Cognitive control refers to the coordination of an individual’s thoughts and actions in relation to internal goals.
As described by the neuroscientists Earl Miller and Jonathan Cohen, “cognitive control stems from the active maintenance of patterns of activity in the prefrontal cortex that represents goals and means to achieve them” [3]. Cognitive control thus comprises the three domains of selective attention, working memory and goal management.
Signals from the prefrontal cortex guide the neural activity in other brain areas, such as sensory regions. Top-down modulation influences this activity, resulting in a bias in the expectations of stimuli present in the environment. In this way, the features, locations and events with the most immediate relevance to an individual’s goals have greater representation in their specific brain areas, and can be detected more rapidly.
Equally important, however, is the ability to disregard stimuli of less importance. This perceptual inhibition is essential to minimize goal interference, which can interrupt the process of accomplishing the desired task [2]. Humans are prone to goal interference in the form of distractions and interruptions – both internal and external [2].
Thus, focusing and ignoring are both critical to the success of facilitating top-down goals, maintaining selectivity of attention in the brain, and shaping the way in which we perceive the world.
References
[edit | edit source]- ↑ a b c d e f g h i Neuroscience. 5 ed. 2012, Sunderland: Sinauer Associates, Inc.
- ↑ a b c d e f g h i j k l m n o p q Gazzaley, A. and L.D. Rosen, The distracted mind: ancient brains in a high-tech world. 2016, Cambridge, MA: MIT Press.
- ↑ a b c d e Miller, E.K. and J.D. Cohen, An integrative theory of prefrontal cortex function. Annu Rev Neurosci, 2001. 24: p. 167-202.
- ↑ Fuster, J.M., Upper processing stages of the perception-action cycle. Trends Cogn Sci, 2004. 8(4): p. 143-5.
- ↑ Corkin, S., Permanent Present Tense. 2013, London: Penguin Books.
- ↑ a b c Gazzaley, A. and A.C. Nobre, Top-down modulation: bridging selective attention and working memory. Trends Cogn Sci, 2012. 16(2): p. 129-35.