Sensory Systems/Computer Models/Simulation of Tinnitus Development
Computational model of tinnitus-related hyperactivity development through homeostatic plasticity after hearing loss
[edit | edit source]Due to the great progress in science and technology, one might fall into belief that humanity can provide a cure to any (health) problem we might face. However, the human body is a complex system, and in many cases - especially in cases of sensory and nervous system - the damage done to the body cannot be undone. One of prominent examples of this is our hearing: since detection of sound depends on displacement of tiny hair cells (stereocilia) which human body cannot regenerate, damage to the hair cells and thus the hearing loss is permanent once it happens. Besides the imminent hearing loss however, damage to the hearing can lead to development of other pathologies, and one of these is "ringing in the ears" or tinnitus – a condition in which a person hears a sound without any external sound source actually being present.
Tinnitus
[edit | edit source]Tinnitus is a widespread otologic condition and is experienced by approximately 10% of population in the world [1] , with the phantom sound being similar to constant pure-tone ringing, roaring, hiss and even music-like tunes, and the sound intensity ranging from quiet background noise to a very loud sound [2]. For most of the patients tinnitus does not significantly affect quality of life, but for ca. 1% of patients it causes severe problems, including depression, anxiety, lack of concentration and disturbed sleep cycle – all due to the inability to perceive silence, and the (psychological) difficulties in coping with this fact.
Generally, tinnitus is characterized as either subjective or objective – with the objective tinnitus being detectable by another person as a sound coming from the ear canal, while the subjective tinnitus is imperceptible to everyone except to the patient and is not linked to any movement in the basilar membrane, that is, there is no real sound input to the auditory nerves in this case [1,2]. In both cases, tinnitus represents a symptom of other underlying conditions and not a disease itself: objective tinnitus can be caused for example by vascular or muscular pathologies or increased awareness of blood flow in the ear (pulsatile tinnitus), while the subjective tinnitus can be caused by noise-induced hearing loss, head injuries, or it can be a side effect of certain medications [2]. The exact mechanisms leading to tinnitus development are still being researched, especially in the case of subjective tinnitus, and below we shall present one possible mechanism leading to development of subjective tinnitus after hearing loss, which is believed to be its most common cause (though patients without any obvious hearing loss can also experience tinnitus) [3].
-
Metallica´s drummer Lars Ulrich suffers from tinnitus which was developed during his early career when he was not using any ear protection while drumming (as opposed to this concert which took place in 2009 in Madrid).
As with any health condition which can cause a noticeable change in one´s life (albeit a small one for most) an effective therapy for tinnitus is highly sought for, and the currently administered therapies are „talk therapies" and counseling to help patients deal with the anxiety caused by tinnitus, fitting a hearing aid and using sound maskers (white-noise sources which mask tinnitus sound while used) [2]. Quite recently, new tinnitus therapies have been made available [4] (for a high price tag [5]), and through a simple computational model of tinnitus-related hyperactivity development we shall explore one possible mechanism behind the functioning of tinnitus therapies (other mechanicsm such as pathological activity synchronisation are to be covered later).
Physiological background of computational model
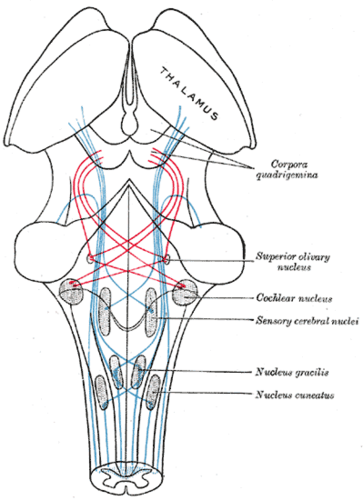
[edit | edit source]Tinnitus can be caused by various pathologies and it is considered that all levels of nervous system are involved in its development. For example, one of many contributing factors are spontaneous otoacoustic emissions (SOAEs), low intensity acoustic signals generated in the cochlea by electromotility of outer hair cells (i.e. self sustained oscillations), which can be perceived by patients as tinnitus [2]. A possible site in the central nervous system which can contribute to tinnitus development is the dorsal cochlear nucleus (DCN), which together with ventral cochlear nucleus (VCN) forms the cochlear nucleus (CN, located in the brainstem) – a point where acoustic information from auditory nerve fibers (AN) is first processed (AN only perform digitalization of input sound) and a point through which all auditory information enters the central nervous system. Quite importantly, DCN is the earliest processing stage at which tinnitus-related changes (hyperactivity, i.e. elevated spontaneous nerve activity) were observed after acoustic trauma, and only the regions of DCN innervated by lesioned parts of cochlear had shown these changes [3].
-
Schematic of the brainstem, cochlear nucleus shown in center right.
Since the time scale of hyperactivity development in DCN is similar to that of homeostatic plasticity – where homeostasis aims to stabilize the mean firing rate of neurons by adjusting the strength of synapses and neuronal excitability – and since changes related to homeostatic plasticity were observed in auditory cortex and brainstem in response to sensory deprivation, the mechanism of homeostatic plasticity is a candidate for explaining the development hearing loss induced (subjective) tinnitus [3]. Said in a simpler way, a possible explanation for tinnitus development is that due to the weaker signal coming toward it, "brain" (e.g. DCN) increases the gain (of auditory circuits) in order to return the signal to its normal level – but this also increases the volume of the noise (spontaneous neuronal activity), which can then become noticeable and be heard as a phantom sound.
As DCN is the first processing stage of auditory input, the overall model can be kept simple by including only the auditory nerve fibers (AN) and dorsal cochlear nucleus fibers (DCN), and the model can be further simplified by focusing only on the phenomenological modelling rather than capturing all known physiological details of AN and DCN in detail.
Computational model
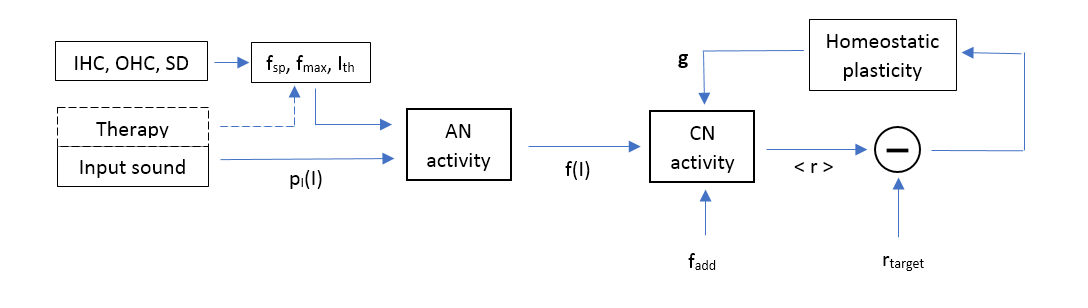
[edit | edit source]The model presented here calculates the population firing rates of AN and DCN fibers (CN in further text) under the influence of varying cochlear damage parameters (IHC, OHC loss and stereocilia damage) and under different therapies (no therapy, white-noise masker, matched sound therapy). The model and all numerical values used below are entirely adapted from the paper by Schaette, Kempter; Development of tinnitus-related neuronal hyperactivity through homeostatic plasticity after hearing loss: a computational model [2], and the schematic overview of the model can be seen in the figure below, with each segment of the model explained in further text. The numerical code was implemented in MATLAB, MathWorks Inc. and the full code is available here (Google Drive link).
-
Schematic representation of the computational model.
Distribution of environmental sound intensities
[edit | edit source]In this model it is assumed that the (daily) average of acoustic stimuli such as speech, music, environmental sound and noise can be approximated with a Gaussian probability density function of sound intensities with a mean value of and standard deviation of , as shown in the figure right below. For simplicity it shall be assumed that same probability density function is valid for all frequencies of the input sound stimulus, and unless otherwise stated, the entire following discussion of the model refers to a single input sound frequency (i.e. to a small region of cochlea which encodes this frequency).
-
Probability density function of time-averaged input sound intensity levels.
-
Probability density function with cumulative probability density between intensity values of 0 dB and 50 dB. Value of cumulative probability density is proportional to the shaded area in the graph.
Auditory nerve firing rates
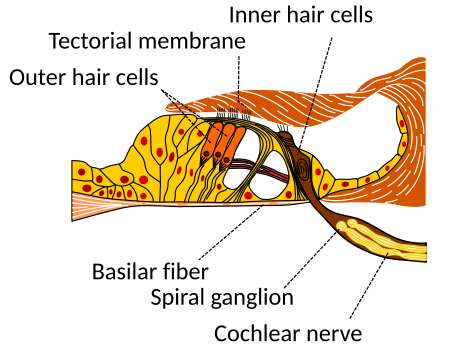
[edit | edit source]Each inner hair cells is innervated by many auditory nerve fibers (10-30) [3, 6] with varying thresholds intensities and spontaneous firing rates, and this population coding of input sound intensity enables many low dynamic range fibers (with differing threshold intensities) to encode an input sound of bigger dynamic range. In this model, the auditory nerve fiber responses are averaged over the whole population, yielding a population firing rate which includes the responses of nerve fibers ranging from low threshold / high spontaneous firing rates to high threshold / low spontaneous firing rates, and which approximates an input to downstream neurons (in cochlear nucleus) which form synapses with many AN fibers.
-
Schematic of the Corti organ, showing the outer hair cells, inner hair cells and cochlear nerve in which auditory nerve fibers are situated.
Based on these assumptions, when the input sound intensity is below the (lowest AN fiber) threshold, there is a population spontaneous activity with a spontaneous firing rate which occurs with probability . Further assuming that AN fiber threshold are distributed (have evolved!) in such a way to produce a flat probability density function (i.e. that each AN firing rate is equally informative) relative to the assumed average sound input, the population firing rate at the input intensity is taken to be a linear function of (normalized) cumulative distribution function of with a probability of driven AN activity equal to: (see figure left above). Finally, assuming that for high input intensities the population firing rate saturates at , we obtain for AN population-averaged firing rate the following dependence on the intensity of input sound (shown in figure below on the right):
where is the maximum probability of driven activity. The above dependence produces the following probability density function (shown in figure below on the left):
where . An important point to note here is that the function of auditory nerves is only the digitalization of analog sound intensity signal into a digital firing rate signal (AD converter) and that this process is not influenced in this model by any homeostatic plasticity mechanism, but only by cochlear damage.
-
Auditory nerve fibers population-averaged firing rates versus input sound intensity. Minimum firing rate corresponds to f_sp and maximum firing rate to f_max.
-
Probability density function of auditory nerve population-averaged firing rates. Delta Dirac peak at spontaneous firing rate fsp has an intensity (area) equal to the probability of spontaneous firing P_sp , and for visual purposes the Delta Dirac peak has been replaced with a rectangular function. As expected due to underlying assumptions, probability density function is flat.
Effects of cochlear damage on auditory nerve population firing rates
[edit | edit source]Parameters that influence the population firing rates in this model are , , and , all of which can be changed by cochlear damage, and here we shall model the effects of inner hair cell loss (IHCs), outer hair cell loss (OHCs) and stereocillia damage (SD) on these parameters.
Inner hair cells loss
[edit | edit source]Since AN fibers innervate IHCs, loss of IHCs will reduce the total number of inputs to the population of AN fibers under consideration. Assuming that the population spontaneous firing rate is equal to a product of a single AN fiber spontaneous firing frequency with the total number of AN fibers in the population (), loss of IHCs can be modelled as a multiplicative reduction in both spontaneous and maximum firing rates: and . In figures below the condition with 30% IHC loss is shown, with a noticeable reduction in firing rates and no effect on spontaneous activity probability since IHC loss does not cause change in threshold intensity values.
-
AN population-averaged firing rates versus input sound intensity for healthy condition and 30% IHC loss.
-
AN population-average firing rate PDF for 30% IHC loss.
Outer hair cells loss
[edit | edit source]Function of OHCs in hearing can be simply described as amplification of input sound (due to their electromotility) by about in healthy case, and therefore their loss can be modelled as a linear increase in the threshold intensity: where [3]. Since loss of OHCs increases the threshold intensity, this leads to an increased probability of spontaneous firing and thus to reduced probability of driven activity, as shown in figures below.
-
AN population-averaged firing rates versus input sound intensity for healthy condition and 66% OHC loss.
-
AN population-average firing rate PDF for 66% IHC loss.
Sterocilia damage
[edit | edit source]Finally, the noise induced damage to stereocilia of both IHCs and OHCs can be modeled in a similar way as above, with the difference that maximum firing rate remains unchanged. Therefore, damage to stereocillia causes a decrease in AN spontaneous firing rate and an increase in AN threshold intensity, with a complete loss of all stereocilia increasing the threshold by and lowering the spontaneous firing rate by one third [3]. As shown in the figures below, this results in an increase in spontaneous activity probability (as in OHCs loss) and reduction of spontaneous firing rate (as in IHCs loss).
-
AN population-averaged firing rates versus input sound intensity for healthy condition and 50% SD.
-
AN population-average firing rate PDF for 50% SD.
Cochlear nucleus firing rates
[edit | edit source]As mentioned earlier, downstream auditory neurons in the cochlear nucleus receive the inputs from AN fibers and are the first auditory processing stage (in which tinnitus related changes have been observed) [3]. Here the CN neurons are modelled as neurons which receive input from the population of AN fibers at some firing rate and an additional input from other sources (e.g. somatosensory system) at rate , and outputs a signal at a firing rate r which depends on the input firing rates and other parameters in a nonlinear way. The parameters of this nonlinear mapping are the maximum firing rate , threshold response , and most importantly, the adjustable synaptic gain factor :
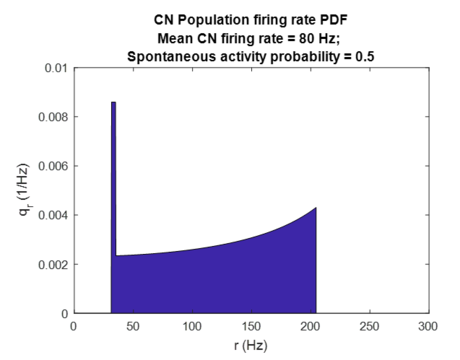
In healthy conditions the gain factor is equal to 1 and threshold is equal to the additional firing rate input, thus cancelling effects of additional input on the CN output firing rate. For the above CN firing rate function, we can derive the corresponding probability density function as follows:
and the corresponding mean CN firing rate:
-
Dependence of CN firing rate on AN firing rate for healthy (g=1, blue) and pathological case (g=1.89, red)
-
Probability density function of CN firing rates. Delta Dirac peak at r_sp has been visually augmented as in figure 8.
Homeostatic plasticity and tinnitus-related hyperactivity generation
[edit | edit source]Homeostatic plasticity is a mechanism which aims to stabilize the mean activity of a neuron around some target value, and it has a time scale of dew days [3]. Here the homeostatic response is modeled as a change in the gain factor when there is a deviation of mean CN firing rate relative the target value , as in the case of hearing loss induced changes in AN and CN firing rates. The value of gain factor needed to reach the target value of firing rate is calculated numerically, and a upper limit of is imposed to account for constraints on changing the synaptic strengths and neuron excitability through homeostatic plasticity [3]. In the case of hearing damage, mean CN firing rate will initially decrease which will trigger the homeostatic plasticity mechanism (figure below, left). After that, in a time period of few days the homeostatic plasticity will increase the value of gain factor and return the mean CN firing rate to its target value. However, this will also increase the spontaneous firing rate in the cochlear nucleus (figure below, right), and this hyperactivity of cochlear nucleus neurons can be perceived as tinnitus.
-
Probability density function of CN firing rates immediately after a lesion causing 50% stereocilia damage.
-
Probability density function of CN firing rates for 50% stereocilia damage after homeostatic plasticity has returned the mean CN firing rate to its target value of 130 Hz by increasing the gain factor from 1 to 1.89.
Tinnitus therapy
[edit | edit source]When an acoustic stimulus is present at an intensity above the threshold level , the AN firing rate will become . If this stimulus is presented constantly as an addition to the environmental sound intensities, all AN fibers with thresholds below will now become constantly stimulated and fire at a rate with probability , as opposed to the case without additional stimulus where all AN fibers below Ith constantly fire with rate and probability . Hence, the effect of additional stimulation is modeled by replacing with and with . The new value of mean CN firing rate can be then computed using these values, and in case it is different from the target value r* homeostatic plasticity will be activated, and value of gain factor will change. Using an appropriate additional stimulus, it therefore might be possible to reduce the gain factor and thus the spontaneous firing rate in the cochlear nucleus to their healthy values, thereby (possibly) eliminating the perception of tinnitus.
To demonstrate the (hypothetical) benefits of such a treatment, we consider a case of frequency-dependent stereocillia damage, with most of the damage occuring in parts of cochlea encoding high-frequency sounds (thus intensity threshold values are high for high frequencies). Please note that gain factors, mean CN firing rates and all other values are calculated for each frequency of the input sound independently (i.e. each region in cochlea is considered independently). In the figures below a side-to-side comparison of three treatments is given, with the columns corresponding to: no treatment, white-noise stimulation, and matched-noise masker, respectively, while each rows shows the following for each treatment: intensity threshold values together with the input stimulus intensity spectrum, gain factors , and CN spontaneous firing rates for each input sound frequency (i.e. region in cochlea).
Given the results shown below, we can see few interesting effects. Without any additional stimulation, homeostatic plasticity increases the gain factor in highly damaged regions of cochlea in order to return the mean CN firing rate to its target value. However, for high enough levels of damage homeostatic plasticity mechanisms saturates at and the target value is not reached, which in turn causes the overall firing rates to be less amplified as compared to the case with no saturation. Since in this model increasing stereocilia damage decreases the spontaneous firing rates (see figure above, left), together with the saturation of gain factor the overall results is that the spontaneous firing rates decrease with increasing stereocilia damage / increasing sound frequency (figure below, bottom left). The characteristic peak in the CN spontaneous firing rate vs. frequency graph that results from these effects might be what is perceived by patients as „pure-tone“ ringing.
When we provide (for long period of time) an additional stimulation with flat spectrum (white-noise, in figures), the input stimulus will only cause stimulation in regions of cochlea where AN population intensity thresholds are below (figure below, top center, low/mid frequency and low/mid sterocilia damage regions). This in turn causes only AN fiber populations in these regions to be stimulated and to increase their spontaneous activity during stimulation (figure below, bottom center) and their mean CN firing rate - homeostatic response then activates and reduces the gain of these fibers, leading to gain factors lower than unity (figure below, center; high threshold/damage regions remain unaffected). When additional stimulus if removed, these changes then lead to lowered CN spontaneous firing rates in low/mid threshold regions, thus making the „pure-tone“ peak even more pronounced and possibly worsening the perception of tinnitus.
Now we consider the case of matched-noise stimulus. Here the input stimulus spectrum is chosen such that stimulus intensity is always slightly above the threshold intensity of the respective region of cochlea / respective sound frequency ( difference shown in figure below in top right), and this is performed only for the high threshold/damage regions of cochlea. Following the same logic as in the previous case, under the influence of this stimulation (for prolonged period of time) the CN spontaneous firing rates and mean CN firing rates corresponding to these regions will increase (figure below, bottom right), activating the homeostatic plasticity response which lowers the corresponding CN gain factors (figure below, center right). Immediately after the stimulation is removed, that is before homeostatic plasticity changes the newly reached gain factors, lowering of gain factors in highly damaged regions of cochlea causes the CN spontaneous firing rates to be much closer to their healthy values and largerly eliminates the „pure-tone“ peak. This in turn suggest that it might be possible to eliminate tinnitus by regularly administering a matched-noise stimulation to the patient – this will not revert the hearing damage sustained by the patient, but it will prevent the homeostatic response to cause tinnitus-related hyperactivity in CN.
-
Comparison of effects of various additional stimuli on CN gain factors and CN spontaneous firing rates.
Final remarks
[edit | edit source]A simplified model of interaction between input sound, auditory neurons and cochlear nucleus discussed above is able to produce hyperactivity in the cochlear nucleus (increased spontaneous firing rates) in the presence of cochlear damage through the mechanism of homeostatic plasticity which aims to restore mean CN firing rates to its target value. However, whether this hyperactivity indeed leads to perception of tinnitus in humans is not clear although animal models do seem to point with this, as mentioned in [3]. Modeling of further downstream neurons is possible as an addition to this model, but the basic premise of homeostatic response causing hyperactivity would stay the same.
As for the novel tinnitus therapies being offered, some of them do seem to be similar to matched-noise therapy described above, however the most promising (and/or advertised) approach seems to be the "CR® Neuromodulation therapy" which disrupt the „pathological synchronous oscillatory activity in the brain” [7] by administering “short sequences of high-frequency pulses” to groups of neurons affected [8] (i.e. groups of neurons which have pathologically started to fire together are reset by this therapy). Description of this approach to tinnitus therapy is left as further work in this chapter, with additional details available in the references.
References:
[edit | edit source][1] Tinnitus, Wikipedia Article, 30.09.2017. https://en.wikipedia.org/wiki/Tinnitus
[2] Han et al.; Tinnitus: Characteristics, Causes, Mechanisms, and Treatments; Journal of Clinical Neurology, 5(1), 11–19, 2009
[3] Schaette, Kempter; Development of tinnitus-related neuronal hyperactivity through homeostatic plasticity after hearing loss: a computational model; European Journal of Neuroscience, Vol. 23, p. 3124 - 3138, 2006
[4] Tinnitus Treatment - Causes and treatment of tinnitus, The Tinnitus Clininc Youtube channel, Youtube video, 01.09.2017. https://www.youtube.com/watch?v=fIAjJogJ0cs
[5] The Tinnitus Clinic, Website, 01.09.2017. https://thetinnitusclinic.co.uk/tinnitus-treatment/?field=YTL
[6] Auditory system, Wikipedia article, 01.09.2017. https://en.wikipedia.org/wiki/Auditory_system
[7] Hoare et al.; Evaluation of the acoustic coordinated reset (CR ®) neuromodulation therapy for tinnitus: study protocol for a double-blind randomized placebo-controlled trial; Trials 2013 14:207.
[8] Peter A. Tass; A model of desynchronizing deep brain stimulation with a demand-controlled coordinated reset of neural subpopulations; Biol. Cybern. 89, 81–88 (2003)