School Science/Gram staining
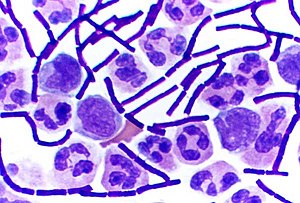
Gram staining, or Gram's method, is an empirical method of differentiating bacterial species into two large groups based on the chemical and physical properties of their cell walls.
The method is named after the inventor, the Danish scientist Hans Christian Gram, who developed the technique in 1884 to discriminate between pneumococci and Klebsiella pneumoniae bacteria.
Uses
[edit | edit source]Gram stains are performed on body fluid or biopsy when infection is suspected. It yields results much quicker than culture, and is especially important when infection would make an important difference in the patient's treatment and prognosis; examples are cerebrospinal fluid for meningitis and synovial fluid for septic arthritis.
Mechanism
[edit | edit source]Gram-positive bacteria have a thick mesh-like cell wall made of peptidoglycan that is capable of retaining the violet dye/iodine complex. Gram-negative bacteria have a thin cell wall made of a layer of peptidoglycan. In addition to an inner membrane, they also have an outer membrane which contains lipids, and is separated from the cell wall by the periplasmic space.
The decolorizing mixture causes dehydration of the multilayered peptidoglycan in the Gram-positive cell wall, thus decreasing the space between the molecules and causing the cell wall to trap the crystal violet-iodine complex within the cell. But in Gram-negative bacteria, the decolorizing mixture acts as a lipid solvent and dissolves the outer membrane of the Gram-negative cell wall. The thin layer of peptidoglycan is unable to retain the crystal violet-iodine complex and the Gram-negative cell is decolorized. The decolorization step is the crucial one, and requires some degree of skill, as Gram-positivity is not an all-or-none phenomenon.
As a rule of thumb (which has exceptions), Gram-negative bacteria are more dangerous as disease organisms, because their outer membrane is often hidden by a capsule or slime layer which hides the antigens of the cell and so acts as "camouflage" - the human body recognises a foreign body by its antigens; if they are hidden, it becomes harder for the body to detect the invader. Often the presence of a capsule will increase the virulence of a pathogen. Additionally, Gram-negative bacteria have lipopolysaccharide in their outer membrane. Lipopolysaccharide is an endotoxin which increases the severity of inflammation. This inflammation may be so severe that septic shock may occur. Gram-positive infections are generally less severe because the human body does not contain peptidoglycan, and in fact the human body produces an enzyme called lysozyme which attacks the open peptidoglycan layer of Gram-positive bacteria. Gram-positive bacteria are also much more susceptible to beta-lactam antibiotics, such as penicillin.
Procedure
[edit | edit source]A description of Gram staining
[edit | edit source]- First, an inoculum is taken from a culture using an inoculation loop and put on a slide and then allowed to air dry. If the culture is solid, it is diluted by adding a drop of water or sterile saline on the slide and mixing with the loop. It is important here to take a very small inoculum so that the end result is a sparse single layer of bacteria. It is a common mistake for beginners to put far too much inoculum at this step.
- The specimen is heat-fixed by passing the slide, inoculum side up, through a bunsen flame 1-2 times, without allowing the slide to become hot to the touch. This prevents the bacteria from being washed away later and it also kills the bacteria.
- A basic dye, crystal violet or gentian violet, is used to stain the slide. This dye is taken up by both Gram-positive and Gram-negative bacteria. Allow to stain for 1 minute. The slide should look purple to the unaided eye, and if examined microscopically at this point both Gram-positive and Gram-negative bacteria are purple. Lugol can also be used instead of crystal violet.
- Rinse off with water for a maximum of 5 seconds.
- Add iodine (Gram's iodine) solution (1% iodine, 2% potassium iodide in water) for 1 minute. This acts as a mordant and fixes the dye.
- Rinse with water.
- Apply 95% ethanol or a mixture of acetone and alcohol several times until no more colour appears to come from the sample. This washes away all the unbound basic dye, (usually crystal violet) and leaves Gram-positive organisms stained purple and Gram-negative organisms unstained (colourless).
- Rinse with water immediately to prevent over-decolourisation.
- Apply a suitable counterstain. Opinions vary as to the best choice but suitable stains include safranin or fuchsin. This stain is taken up by both Gram-positive and Gram-negative organisms, but does not alter the colour of Gram-positive organism much, as they are already purple. It does, however, make the Gram-negative organisms pinkish-red.
- Blot gently and allow to dry. Do not smear.
Interpretation
[edit | edit source]Inspect the slide under a microscope
- Gram-positive organisms will appear blue-black or purple.
- Gram-negative organisms will appear red or pink.
Organisms that cannot reliably be differentiated by this staining technique are said to be Gram-variable
References
[edit | edit source]- Gram HC. Über die isolierte Färbung der Schizomyceten in Schnitt- und Trockenpreparäten. Fortschr Med 1884;2:185-89.
- Bergey, John G.; Holt, Noel R.; Krieg, Peter H.A. Sneath (1994). Bergey's Manual of Determinative Bacteriology, 9th ed. Lippincott Williams & Wilkins. ISBN 0-683-00603-7.
- Madigan, Michael T.; Martinko, John; Parker, Jack (2004). Brock Biology of Microorganisms, 10th Edition. Lippincott Williams & Wilkins. ISBN 0-130-66271-2.