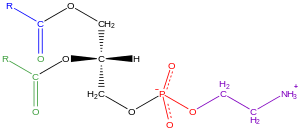
Principles of Biochemistry/Lipids
In biochemistry, a fatty acid is a carboxylic acid with a long unbranched aliphatic tail (chain), which is either saturated or unsaturated. Most naturally occurring fatty acids have a chain of an even number of carbon atoms, from four to 28. Fatty acids are usually derived from triglycerides or phospholipids. When they are not attached to other molecules, they are known as "free" fatty acids. Fatty acids are important sources of fuel because their metabolism yield large quantities of ATP. Many cell types can use either glucose or fatty acids for this purpose. In particular, heart and skeletal muscle prefer fatty acids. The brain cannot use fatty acids as a source of fuel; it relies on glucose or on ketone bodies.
Nomenclature of fatty acids
[edit | edit source]
Several different systems of nomenclature are used for fatty acids. The following table describes the most common systems.
| System | Example | Explanation |
|---|---|---|
| Trivial nomenclature | Palmitoleic acid | Trivial names (or common names) are non-systematic historical names, which are the most frequent naming system used in literature. Most common fatty acids have trivial names in addition to their systematic names (see below). These names frequently do not follow any pattern, but they are concise and often unambiguous. |
| Systematic nomenclature | (9Z)-octadecenoic acid | Systematic names (or IUPAC names) derive from the standard IUPAC Rules for the Nomenclature of Organic Chemistry, published in 1979,[1] along with a recommendation published specifically for lipids in 1977.[2] Counting begins from the carboxylic acid end. Double bonds are labelled with cis-trans isomerism-/trans- notation or E-/Z- notation, where appropriate. This notation is generally more verbose than common nomenclature, but has the advantage of being more technically clear and descriptive. |
| Δx nomenclature | cis,cis-Δ9,Δ12 octadecadienoic acid | In Δx (or delta-x) nomenclature, each double bond is indicated by Δx, where the double bond is located on the xth carbon–carbon bond, counting from the carboxylic acid end. Each double bond is preceded by a cis- or trans- prefix, indicating the conformation of the molecule around the bond. For example, linoleic acid is designated "cis-Δ9, cis-Δ12 octadecadienoic acid". This nomenclature has the advantage of being less verbose than systematic nomenclature, but is no more technically clear or descriptive. |
| n−x nomenclature | n−3 | n−x (n minus x; also ω−x or omega-x) nomenclature both provides names for individual compounds and classifies them by their likely biosynthetic properties in animals. A double bond is located on the xth carbon–carbon bond, counting from the terminal methyl carbon (designated as n or ω) toward the carbonyl carbon. For example, α-Linolenic acid is classified as a n−3 or omega-3 fatty acid, and so it is likely to share a biosynthetic pathway with other compounds of this type. The ω−x, omega-x, or "omega" notation is common in popular nutritional literature, but IUPAC has deprecated it in favor of n−x notation in technical documents.[1] The most commonly researched fatty acid biosynthetic pathways are n−3 and n−6, which are hypothesized[by whom?] to increase or decrease[citation needed] inflammation. |
| Lipid numbers | 18:3 18:3, n−6 18:3, cis,cis,cis-Δ9,Δ12,Δ15 |
Lipid numbers take the form C:D, where C is the number of carbon atoms in the fatty acid and D is the number of double bonds in the fatty acid. This notation can be ambiguous, as some different fatty acids can have the same numbers. Consequently, when ambiguity exists this notation is usually paired with either a Δx or n−x term.[1] |
Types of fatty acids
[edit | edit source]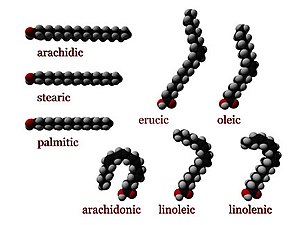
Fatty acids can be saturated and unsaturated, depending on double bonds. They differ in length as well.
Long and short fatty acids
[edit | edit source]In addition to saturation, fatty acids have different lengths, often categorized as short, medium, or long.
- Short-chain fatty acids (SCFA) are fatty acids with aliphatic tails of fewer than six carbons.
- Medium-chain fatty acids (MCFA) are fatty acids with aliphatic tails of 6–12.[3] carbons, which can form medium-chain triglycerides.
- Long-chain fatty acids (LCFA) are fatty acids with aliphatic tails longer than 12 carbons.[4]
- Very-Long-chain fatty acids (VLCFA) are fatty acids with aliphatic tails longer than 22 carbons
Unsaturated fatty acids
[edit | edit source]
Unsaturated fatty acids resemble saturated fatty acids, except that the chain has one or more double-bonds between carbon atoms. The two carbon atoms in the chain that are bound next to either side of the double bond can occur in a cis or trans configuration.
- cis
- A cis configuration means that adjacent hydrogen atoms are on the same side of the double bond. The rigidity of the double bond freezes its conformation and, in the case of the cis isomer, causes the chain to bend and restricts the conformational freedom of the fatty acid. The more double bonds the chain has in the cis configuration, the less flexibility it has. When a chain has many cis bonds, it becomes quite curved in its most accessible conformations. For example, oleic acid, with one double bond, has a "kink" in it, whereas linoleic acid, with two double bonds, has a more pronounced bend. Alpha-linolenic acid, with three double bonds, favors a hooked shape. The effect of this is that, in restricted environments, such as when fatty acids are part of a phospholipid in a lipid bilayer, or triglycerides in lipid droplets, cis bonds limit the ability of fatty acids to be closely packed, and therefore could affect the melting temperature of the membrane or of the fat.
- trans
- A trans configuration, by contrast, means that the next two hydrogen atoms are bound to opposite sides of the double bond. As a result, they do not cause the chain to bend much, and their shape is similar to straight saturated fatty acids.
In most naturally occurring unsaturated fatty acids, each double bond has three n carbon atoms after it, for some n, and all are cis bonds. Most fatty acids in the trans configuration (trans fats) are not found in nature and are the result of human processing (e.g., hydrogenation).
The differences in geometry between the various types of unsaturated fatty acids, as well as between saturated and unsaturated fatty acids, play an important role in biological processes, and in the construction of biological structures (such as cell membranes).
| Common name | Chemical structure | Δx | C:D | n−x |
|---|---|---|---|---|
| Myristoleic acid | CH3(CH2)3CH=CH(CH2)7COOH | cis-Δ9 | 14:1 | n−5 |
| Palmitoleic acid | CH3(CH2)5CH=CH(CH2)7COOH | cis-Δ9 | 16:1 | n−7 |
| Sapienic acid | CH3(CH2)8CH=CH(CH2)4COOH | cis-Δ6 | 16:1 | n−10 |
| Oleic acid | CH3(CH2)7CH=CH(CH2)7COOH | cis-Δ9 | 18:1 | n−9 |
| Linoleic acid | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | cis,cis-Δ9,Δ12 | 18:2 | n−6 |
| α-Linolenic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH | cis,cis,cis-Δ9,Δ12,Δ15 | 18:3 | n−3 |
| Arachidonic acid | CH3(CH2)4CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOHNIST | cis,cis,cis,cis-Δ5Δ8,Δ11,Δ14 | 20:4 | n−6 |
| Eicosapentaenoic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOH | cis,cis,cis,cis,cis-Δ5,Δ8,Δ11,Δ14,Δ17 | 20:5 | n−3 |
| Erucic acid | CH3(CH2)7CH=CH(CH2)11COOH | cis-Δ13 | 22:1 | n−9 |
| Docosahexaenoic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)2COOH | cis,cis,cis,cis,cis,cis-Δ4,Δ7,Δ10,Δ13,Δ16,Δ19 | 22:6 | n−3 |
Essential fatty acids
[edit | edit source]Fatty acids that are required by the body but cannot be made in sufficient quantity from other substrates, therefore must be obtained from food and are called essential fatty acids. Mammals lack the ability to introduce double bonds in fatty acids beyond carbon 9 and 10, hence ω-6 linoleic acid (18:2,9,12), abbreviated LA, and the ω-3 linolenic acid (18:3,9,12,15), abbreviated ALA, are essential for man in the diet. In humans, arachidonic acid (20:4,5,8,11,14) can be synthesized from LA by desaturation and chain elongation (though some carnivores like cats cannot do this, and require arachadonate in the diet). In addition, the human body can make some long-chain ω-3 PUFAs (EPA and DHA) from the ω-3 ALA. Between 1930 and 1950, arachidonic acid and linolenic acid were termed 'essential' because each was more or less able to meet the growth requirements of rats given fat-free diets. Further research has shown that human metabolism requires both ω-3 and ω-6 fatty acids. To some extent, any ω-3 and any ω-6 can relieve the worst symptoms of fatty acid deficiency for its class. Particular fatty acids are still needed at critical life stages (e.g. lactation) and in some disease states. In nonscientific writing, common usage is that the term essential fatty acid comprises all the ω-3 or -6 fatty acids. Conjugated fatty acids like calendic acid are not normally considered essential. Authoritative sources include the whole families, without qualification. Traditionally speaking the LC-PUFA are not essential. See (Cunnane 2003) for a discussion of the current status of the term 'essential'. Because the LC-PUFA are sometimes required, they may be considered "conditionally essential", or not essential to healthy adults. A 2005 study has shown evidence that the ω-6 fat gamma-linolenic acid, GLA has been shown to inhibit the breast cancer promoting gene of Her2/neu. Essential fatty acids should not be confused with essential oils, which are "essential" in the sense of being a concentrated essence[5].
Nomenclature and terminology
[edit | edit source]Fatty acids are straight chain hydrocarbons possessing a carboxylic acid|carboxyl (COOH) group at one end. The carbon next to the carboxylate is known as α, the next carbon β, and so forth. Since biological fatty acids can be of different lengths, the last position is labelled as a "ω", the last letter in the Greek alphabet. Since the physiological properties of unsaturated fatty acids largely depend on the position of the first unsaturation relative to the end position and not the carboxylate, the position is signified by (ω minus n). For example, the term omega-3 fatty acids (ω-3) signifies that the first double bond exists as the third carbon-carbon bond from the terminal CH3 end (ω) of the carbon chain . The number of carbons and the number of double bonds is also listed.
ω-3 18:4 (stearidonic acid) or 18:4 ω-3 or 18:4 n−3 indicates an 18-carbon chain with 4 double bonds, and with the first double bond in the third position from the CH3 end [citation needed]. Double bonds are cis and separated by a single methylene (CH2) group unless otherwise noted. So in free fatty acid form, the chemical structure of stearidonic acid is[6]:

Examples
[edit | edit source]The essential fatty acids start with the short chain polyunsaturated fatty acids (SC-PUFA):
- ω-3 fatty acids:
- α-Linolenic acid or ALA (18:3)
- ω-6 fatty acids:
- Linoleic acid or LA (18:2)
These two fatty acids cannot be synthesised by humans, as humans lack the desaturase enzymes required for their production.
They form the starting point for the creation of longer and more desaturated fatty acids, which are also referred to as long-chain polyunsaturated fatty acids (LC-PUFA):
- omega-3 fatty acid(ω-3 fatty acids):
- eicosapentaenoic acid or EPA (20:5)
- docosahexaenoic acid or DHA (22:6)
- Omega-6 fatty acid|ω-6 fatty acids:
- gamma-linolenic acid or GLA (18:3)
- dihomo-gamma-linolenic acid or DGLA (20:3)
- arachidonic acid or AA (20:4)
ω-9 fatty acids are not essential in humans, because humans generally possess all the enzymes required for their synthesis.
Saturated fatty acids
[edit | edit source]Saturated fatty acids are long-chain carboxylic acids that usually have between 12 and 24 carbon atoms and have no double bonds. Thus, saturated fatty acids are saturated with hydrogen (since double bonds reduce the number of hydrogens on each carbon). Because saturated fatty acids have only single bonds, each carbon atom within the chain has 2 hydrogen atoms (except for the omega carbon at the end that has 3 hydrogens).
| Common name | Chemical structure | C:D |
|---|---|---|
| Lauric acid | CH3(CH2)10COOH | 12:0 |
| Myristic acid | CH3(CH2)12COOH | 14:0 |
| Palmitic acid | CH3(CH2)14COOH | 16:0 |
| Stearic acid | CH3(CH2)16COOH | 18:0 |
| Arachidic acid | CH3(CH2)18COOH | 20:0 |
| Behenic acid | CH3(CH2)20COOH | 22:0 |
| Lignoceric acid | CH3(CH2)22COOH | 24:0 |
| Cerotic acid | CH3(CH2)24COOH | 26:0 |
Synthesis of fatty acid
[edit | edit source]In humans, fatty acids are predominantly formed in the liver and lactating mammary glands, and, to a lesser extent, the adipose tissue. Most acetyl-CoA is formed from pyruvate by pyruvate dehydrogenase in the mitochondria. Acetyl-CoA produced in the mitochondria is condensed with oxaloacetate by citrate synthase to form citrate, which is then transported into the cytosol and broken down to yield acetyl-CoA and oxaloacetate by ATP citrate lyase. Oxaloacetate in the cytosol is reduced to malate by cytoplasmic malate dehydrogenase, and malate is transported back into the mitochondria to participate in the Citric acid cycle.
Fatty acid synthase (FAS)
[edit | edit source]Fatty acid synthase (FAS) is a multi-enzyme that plays a key role in fatty acid synthesis. It is not a single enzyme but a whole enzymatic system composed of 272 kDa multifunctional polypeptide, in which substrates are handed from one functional domain to the next.
There are two principal classes of fatty acid synthases.
Type I systems utilise a single large, multifunctional polypeptide and are common to both mammals and fungi (although the structural arrangement of fungal and mammalian synthases differ). A Type I fatty acid synthase system is also found in the CMN group of bacteria (corynebacteria, mycobacteria, and nocardia). In these bacteria, the FAS I system produces palmititic acid, and cooperates with the FAS II system to produce a greater diversity of lipid products.
Type II is found in archaeabacterial and eubacterial, and is characterized by the use of discrete, monofunctional enzymes for fatty acid synthesis. Inhibitors of this pathway (FASII) are being investigated as possible antibiotics. The mechanism of FAS I and FAS II elongation and reduction is the same, as the domains of the FAS II enzymes are largely homologous to their domain counterparts in FAS I multienzyme polypeptides. However, the differences in the organization of the enzymes - integrated in FAS I, discrete in FAS II - gives rise to many important biochemical differences. The evolutionary history of fatty acid synthases are very much intertwined with that of polyketide synthases (PKS). Polyketide synthases use a similar mechanism and homologous domains to produce secondary metabolite lipids. Furthermore, polyketide synthases also exhibit a Type I and Type II organization. FAS I in animals is thought to have arisen through modification of PKS I in fungi, whereas FAS I in fungi and the CMN group of bacteria seem to have arisen separately through the fusion of FAS II genes[7].
Structure of Fatty acid synthase (FAS)
[edit | edit source]Mammalian FAS consists of two identical multifunctional polypeptides, in which three catalytic domains in the N-terminal section (-ketoacyl synthase (KS), malonyl/acetyltransferase (MAT), and dehydrase (DH)), are separated by a core region of 600 residues from four C-terminal domains (enoyl reductase (ER), -ketoacyl reductase (KR), acyl carrier protein (ACP) and thioesterase (TE)).[8][9]
The conventional model for organization of FAS (see the 'head-to-tail' model on the right) is largely based on the observations that the bifunctional reagent 1,3-dibromopropanone (DBP) is able to crosslink the active site cysteine thiol of the KS domain in one FAS monomer with the phosphopantetheine prosthetic group of the ACP domain in the other monomer.[10][11] Complementation analysis of FAS dimers carrying different mutations on each monomer has established that the KS and MAT domains can cooperate with the ACP of either monomer.[12][13] and a reinvestigation of the DBP crosslinking experiments revealed that the KS active site Cys161 thiol could be crosslinked to the ACP 4'-phosphopantetheine thiol of either monomer.[14] In addition, it has been recently reported that a heterodimeric FAS containing only one competent monomer is capable of palmitate synthesis.[15]
The above observations seemed incompatible with the classical 'head-to-tail' model for FAS organization, and an alternative model has been proposed, predicting that the KS and MAT domains of both monomers lie closer to the center of the FAS dimer, where they can access the ACP of either subunit (see figure on the top right).[16]
Recently, the elucidation of the low resolution X-ray crystallography structure of both pig (homodimer)[17] and yeast FAS (heterododecamer)[18] has provided key structural and mechanistic insights into this important enzyme.
Types of lipids
[edit | edit source]Fatty acyls
Fatty acyls, a generic term for describing fatty acids, their conjugates and derivatives, are a diverse group of molecules synthesized by chain-elongation of an acetyl-CoA primer with malonyl-CoA or methylmalonyl-CoA groups in a process called fatty acid synthesis. They are made of a hydrocarbon chain that terminates with a carboxylic acid group; this arrangement confers the molecule with a polar, hydrophilic end, and a nonpolar, hydrophobic end that is insoluble in water. The fatty acid structure is one of the most fundamental categories of biological lipids, and is commonly used as a building block of more structurally complex lipids. The carbon chain, typically between four to 24 carbons long, may be saturated or unsaturated, and may be attached to functional groups containing oxygen, halogens, nitrogen and sulfur. Where a double bond exists, there is the possibility of either a cis or trans geometric isomerism, which significantly affects the molecule's molecular configuration. Cis-double bonds cause the fatty acid chain to bend, an effect that is more pronounced the more double bonds there are in a chain. This in turn plays an important role in the structure and function of cell membranes. Most naturally occurring fatty acids are of the cis configuration, although the trans form does exist in some natural and partially hydrogenated fats and oils. Examples of biologically important fatty acids are the eicosanoids, derived primarily from arachidonic acid and eicosapentaenoic acid, which include prostaglandins, leukotrienes, and thromboxanes. Other major lipid classes in the fatty acid category are the fatty esters and fatty amides. Fatty esters include important biochemical intermediates such as wax esters, fatty acid thioester coenzyme A derivatives, fatty acid thioester ACP derivatives and fatty acid carnitines. The fatty amides include N-acyl ethanolamines, such as the cannabinoid neurotransmitter anandamide[19].
Glycerolipids Glycerolipids are composed mainly of mono-, di- and tri-substituted glycerols, the most well-known being the fatty acid esters of glycerol (triacylglycerols), also known as triglycerides. In these compounds, the three hydroxyl groups of glycerol are each esterified, usually by different fatty acids. Because they function as a food store, these lipids comprise the bulk of storage fat in animal tissues. The hydrolysis of the ester bonds of triacylglycerols and the release of glycerol and fatty acids from adipose tissue is called fat mobilization. Additional subclasses of glycerolipids are represented by glycosylglycerols, which are characterized by the presence of one or more sugar residues attached to glycerol via a glycosidic linkage. Examples of structures in this category are the digalactosyldiacylglycerols found in plant membranes and seminolipid from mammalian sperm cells[20].
Glycerophospholipids
[edit | edit source]Glycerophospholipids, also referred to as phospholipids, are ubiquitous in nature and are key components of the lipid bilayer of cells, as well as being involved in metabolism and cell signaling. Neural tissue (including the brain) contains relatively high amounts of glycerophospholipids, and alterations in their composition has been implicated in various neurological disorders. Glycerophospholipids may be subdivided into distinct classes, based on the nature of the polar headgroup at the sn-3 position of the glycerol backbone in eukaryotes and eubacteria, or the sn-1 position in the case of archaebacteria[21].
The term glycerophospholipid signifies any derivative of sn-glycero-3-phosphoric acid that contains at least one O-acyl, or O-alkyl, or O-alk-1'-enyl residue attached to the glycerol moiety and a polar head made of a nitrogenous base, a glycerol or an inositol unit. It contains a glycerol core with fatty acids. They can be the same or different subunits of fatty acids. Carbon 1 (tail, apolar) contains a fatty acid, typically saturated Carbon 2 (tail, apolar) contains a fatty acid, typically unsaturated and in the cis conformation, thus appearing "bent" Carbon 3 (head, polar) contains a phosphate group or an alcohol attached to a phosphate group
Nomenclature and stereochemistry In general, glycerophospholipids use a "sn" notation, which stands for stereochemical numbering. When the letters "sn" appear in the nomenclature, by convention the hydroxyl group of the second carbon of glycerol (sn-2) is on the left on a Fischer projection. The numbering follows the one of Fischer's projections, being sn-1 the carbon at the top and sn-3 the one at the bottom.
The advantage of this particular notation is that the spatial conformation (R or L) of the glycero-molecule is determined intuitively by the residues on the positions sn-1 and sn-3.
For example sn-glycero-3-phosphoric acid and sn-glycero-1-phosphoric acid are enantiomers.
Examples of glycerophospholipids
Lecithin and cephalin are more common than the others in most human membranes, but cardiolipin is quite common in the inner membranes of mitochondria.
Phosphatidylethanolamine
Examples of glycerophospholipids found in biological membranes are phosphatidylcholine (also known as PC, GPCho or lecithin), phosphatidylethanolamine (PE or GPEtn) and phosphatidylserine (PS or GPSer). In addition to serving as a primary component of cellular membranes and binding sites for intra- and intercellular proteins, some glycerophospholipids in eukaryotic cells, such as phosphatidylinositols and phosphatidic acids are either precursors of, or are themselves, membrane-derived second messengers. Typically, one or both of these hydroxyl groups are acylated with long-chain fatty acids, but there are also alkyl-linked and 1Z-alkenyl-linked (plasmalogen) glycerophospholipids, as well as dialkylether variants in archaebacteria.

Black:Sphingosine
Red:Phosphocholine
Blue:Fatty acid
Sphingolipids
[edit | edit source]Sphingolipids are a complex family of compounds that share a common structural feature, a sphingoid base backbone that is synthesized de novo from the amino acid serine and a long-chain fatty acyl CoA, then converted into ceramides, phosphosphingolipids, glycosphingolipids and other compounds. The major sphingoid base of mammals is commonly referred to as sphingosine. Ceramides (N-acyl-sphingoid bases) are a major subclass of sphingoid base derivatives with an amide-linked fatty acid. The fatty acids are typically saturated or mono-unsaturated with chain lengths from 16 to 26 carbon atoms. The major phosphosphingolipids of mammals are sphingomyelins (ceramide phosphocholines), whereas insects contain mainly ceramide phosphoethanolamines and fungi have phytoceramide phosphoinositols and mannose-containing headgroups. The glycosphingolipids are a diverse family of molecules composed of one or more sugar residues linked via a glycosidic bond to the sphingoid base. Examples of these are the simple and complex glycosphingolipids such as cerebrosides and gangliosides[22].
Types of Sphingolipids
- Ceramide is the fundamental structural unit common to all sphingolipids. They consist simply of a fatty acid chain attached through an amide linkage to sphingosine.
There are three main types of sphingolipids, differing in their head groups:
- Sphingomyelins have a phosphocholine or phosphoethanolamine molecule with an ester linkage to the 1-hydroxy group of a ceramide.
Sphingomyelin (SPH, sfing-goh-mahy-uh-lin) is a type of sphingolipid found in animal cell membranes, especially in the membranous myelin sheath that surrounds some nerve cell axons. It usually consists of phosphorylcholine and ceramide. In humans, SPH represents ~85% of all sphingolipids.
- Glycosphingolipids, which differ in the substituents on their head group (see image). Glycosphingolipids are ceramides with one or more sugar residues joined in a β-glycosidic linkage at the 1-hydroxyl position[23].
- Cerebrosides have a single glucose or galactose at the 1-hydroxy position.
- Sulfatides are sulfated cerebrosides.
- Cerebrosides have a single glucose or galactose at the 1-hydroxy position.
- Gangliosides have at least three sugars, one of which must be sialic acid.
Sphingolipidoses are a class of disorders relating to Sphingolipid metabolism.
| Disease | Deficient enzyme | Accumulated products | Symptoms | Inheritance |
|---|---|---|---|---|
| Niemann-Pick disease | Sphingomyelinase | Sphingomyelin in brain and RBCs |
|
Autosomal recessive |
| Fabry disease | α-galactosidase A | Glycolipids in brain, heart, kidney |
|
X-linked[25] |
| Krabbe disease | Galactocerebrosidase | Glycolipids in oligodendrocytes |
|
Autosomal recessive |
| Gaucher disease | Glucocerebrosidase | Glucocerebrosides in RBCs, liver and spleen |
|
Autosomal recessive, prevalent in Ashkenazi Jews |
| Tay-Sachs disease | Hexosaminidase A | GM2 gangliosides in neurons |
|
Autosomal recessive, prevalent in Ashkenazi Jews |
| Metachromatic leukodystrophy | Arylsulfatase A or prosaposin | Sulfatide compounds in neural tissue | Demyelinisation in CNS and PNS:
|
Autosomal recessive[26] |
Sterol lipids Sterol lipids, such as cholesterol and its derivatives, are an important component of membrane lipids, along with the glycerophospholipids and sphingomyelins. The steroids, all derived from the same fused four-ring core structure, have different biological roles as hormones and signaling molecules. The eighteen-carbon (C18) steroids include the estrogen family whereas the C19 steroids comprise the androgens such as testosterone and androsterone. The C21 subclass includes the progestogens as well as the glucocorticoids and mineralocorticoids. The secosteroids, comprising various forms of vitamin D, are characterized by cleavage of the B ring of the core structure. Other examples of sterols are the bile acids and their conjugates, which in mammals are oxidized derivatives of cholesterol and are synthesized in the liver. The plant equivalents are the phytosterols, such as β-sitosterol, stigmasterol, and brassicasterol; the latter compound is also used as a biomarker for algal growth. The predominant sterol in fungal cell membranes is ergosterol[27].
Prenol lipids Prenol lipids are synthesized from the 5-carbon precursors isopentenyl diphosphate and dimethylallyl diphosphate that are produced mainly via the mevalonic acid (MVA) pathway. The simple isoprenoids (linear alcohols, diphosphates, etc.) are formed by the successive addition of C5 units, and are classified according to number of these terpene units. Structures containing greater than 40 carbons are known as polyterpenes. Carotenoids are important simple isoprenoids that function as antioxidants and as precursors of vitamin A. Another biologically important class of molecules is exemplified by the quinones and hydroquinones, which contain an isoprenoid tail attached to a quinonoid core of non-isoprenoid origin. Vitamin E and vitamin K, as well as the ubiquinones, are examples of this class. Prokaryotes synthesize polyprenols (called bactoprenols) in which the terminal isoprenoid unit attached to oxygen remains unsaturated, whereas in animal polyprenols (dolichols) the terminal isoprenoid is reduced[28].
Saccharolipids
Structure of the saccharolipid Kdo2-Lipid A. Glucosamine residues in blue, Kdo residues in red, acyl chains in black and phosphate groups in green.
Saccharolipids describe compounds in which fatty acids are linked directly to a sugar backbone, forming structures that are compatible with membrane bilayers. In the saccharolipids, a monosaccharide substitutes for the glycerol backbone present in glycerolipids and glycerophospholipids. The most familiar saccharolipids are the acylated glucosamine precursors of the Lipid A component of the lipopolysaccharides in Gram-negative bacteria. Typical lipid A molecules are disaccharides of glucosamine, which are derivatized with as many as seven fatty-acyl chains. The minimal lipopolysaccharide required for growth in E. coli is Kdo2-Lipid A, a hexa-acylated disaccharide of glucosamine that is glycosylated with two 3-deoxy-D-manno-octulosonic acid (Kdo) residues[29].
Polyketides Polyketides are synthesized by polymerization of acetyl and propionyl subunits by classic enzymes as well as iterative and multimodular enzymes that share mechanistic features with the fatty acid synthases. They comprise a large number of secondary metabolites and natural products from animal, plant, bacterial, fungal and marine sources, and have great structural diversity. Many polyketides are cyclic molecules whose backbones are often further modified by glycosylation, methylation, hydroxylation, oxidation, and/or other processes. Many commonly used anti-microbial, anti-parasitic, and anti-cancer agents are polyketides or polyketide derivatives, such as erythromycins, tetracyclines, avermectins, and antitumor epothilones[30].
Galactolipids and Sulfolipids
[edit | edit source]Galactolipids are a type of glycolipid whose sugar group is galactose. They're different from glycosphingolipids in that they do not have nitrogen in their composition. They are the main part of plant membrane lipids where they substitute phospholipids to conserve phosphate for other essential processes. These chloroplast membranes contain a high quantity of monogalactosyldiacylglycerol (MGDG) and digalactosyldiacylglycerol (DGDG). They probably also assume a direct role in photosynthesis, as they have been found in the X-ray structures of photosynthetic complexes. The galactolipid galactocerebroside (GalC) and its sulfated derivative sulfatide is also in abundance present (together with a small group of proteins) in myelin, the membrane around the axons in the nervous system of vertebrates.
Sulfolipids are a class of lipids which possess a sulfur-containing functional group. One of the most common consituents of sulfolipids is sulfoquinovose, which is acylated to form sulfoquinovosyl diacylglycerols. In plants, sulfolipids are important intermediates in the sulfur cycle.
What is Sulfoquinovose?
Sulfoquinovose, also known as 6-sulfoquinovose and 6-deoxy-6-sulfo-D-glucopyranose is a monosaccharide sugar that is found as a building block in the sulfolipid sulfoquinovosyl diacylglycerol (SQDG). Sulfoquinovose is a sulfonic acid derivative of glucose, the sulfonic acid group is introduced into the sugar by the enzyme UDP-sulfoquinovose synthase (SQD1).
Lipoprotein
[edit | edit source]A lipoprotein is a biochemical assembly that contains both proteins and lipids water-bound to the proteins. Many enzymes, transporters, structural proteins, antigens, adhesins and toxins are lipoproteins. Examples include the high density (HDL) and low density (LDL) lipoproteins which enable fats to be carried in the blood stream, the transmembrane proteins of the mitochondrion and the chloroplast, and bacterial lipoproteins.
Classification
[edit | edit source]Classification of lipoproteins acoording to their density
[edit | edit source]Lipoproteins may be classified as follows, listed from larger and less dense to smaller and denser. Lipoproteins are larger and less dense, if they consist of more fat than of protein. They are classified on the basis of electrophoresis and ultracentrifugation.
Chylomicrons carry triglycerides (fat) from the intestines to the liver, skeletal muscle, and to adipose tissue.
Very low density lipoproteins (VLDL) carry (newly synthesised) triacylglycerol from the liver to adipose tissue.
Intermediate density lipoproteins (IDL) are intermediate between VLDL and LDL. They are not usually detectable in the blood.
Low density lipoproteins (LDL) carry cholesterol from the liver to cells of the body. LDLs are sometimes referred to as the "bad cholesterol" lipoprotein.
High density lipoproteins (HDL) collect cholesterol from the body's tissues, and bring it back to the liver. HDLs are sometimes referred to as the "good cholesterol" lipoprotein.
Alpha and beta
[edit | edit source]It is also possible to classify lipoproteins as "alpha" and "beta", according to the classification of proteins in serum protein electrophoresis. This terminology is sometimes used in describing lipid disorders such as Abetalipoproteinemia.
Lipoprotein(a)
Lipoprotein(a) – Lp(a), Cardiology diagnostic tests < 14 mg/dL : Normal 14-19 mg/dL : ? > 19 mg/dL : High risk
How to lower: aerobic exercise, niacin, aspirin, guggulipid.
WAX
[edit | edit source]Waxes are organic compounds that characteristically consist of a long alkyl chains. Natural waxes are typically esters of fatty acids and long chain alcohols. Synthetic waxes are long-chain hydrocarbons lacking functional groups.
Types of waxe
Plant and animal waxes Waxes are biosynthesized by many plants or animals. They typically consist of several components, including wax esters, wax acids, wax alcohols, and hydrocarbons. Wax esters are typically derived from a variety of carboxylic acids and a variety of fatty alcohols. The composition depends not only on species, but also on geographic location of the organism. Because they are mixtures, naturally produced waxes are softer and melt at lower temperatures than the pure components. Animal waxes The best known animal wax is beeswax, but other insects secrete waxes. A major component of beeswax is the ester myricyl palmitate substance which is used in constructing their honeycombs. Its melting point is 62-65 °C. Spermaceti occurs in large amounts in the head oil of the sperm whale. One of its main constituents is cetyl palmitate, another ester of a fatty acid and a fatty alcohol. Lanolin is a wax obtained from wool, consisting of esters of sterols[31]. eg.Beeswax - produced by honey bees
Chinese wax - produced by the scale insect Ceroplastes ceriferus[check spelling]
Earwax - found in the human ear.
Lanolin (wool wax) - from the sebaceous glands of sheep
Shellac wax - from the lac insect Kerria lacca
Spermaceti - from the head cavities and blubber of the sperm whale
Plant waxes Especially in warm climates, plants secrete waxes as a way to control evaporation and hydration.[2] From the commercial perspective, the most important wax is Carnauba wax, a hard wax obtained from the Brazilian palm. Containing the ester myricyl cerotate, it has many applications. Other more specialized vegetable waxes include candelilla wax, ouricury wax, sugarcane wax, retamo wax, jojoba oil. The epicuticular waxes of plants are mixtures of substituted long-chain aliphatic hydrocarbons, containing alkanes, fatty acids, primary and secondary alcohols, diols, ketones, aldehydes.
Petroleum derived waxes
Although most natural waxes are esters, paraffin waxes are hydrocarbons, mixtures of alkanes usually in a homologous series of chain lengths. These materials represent a significant fraction of petroleum. They are refined by vacuum distillation. Paraffin waxes are mixtures of saturated of n- and isoalkanes, naphthenes, and alkyl- and naphthene-substituted aromatic compounds. The degree of branching has an important influence on the properties. Millions of tons of paraffin waxes are produced annually. They are used in adhesives, in foods (such as chewing gum and cheese wrapping), in cosmetics, and as coatings.
Montan wax Montan wax is a fossilized wax extracted from coal and lignite. It is very hard, reflecting the high concentration of saturated fatty acids and alcohols, not esters that characterize softer waxes. Although dark brown and smelly, they can be purified and bleached to give commercially useful products.
Polyethylene and related derivatives Some waxes are obtained by cracking polyethylene at 400 °C. The products have the formula (CH2)nH2, where n ranges between about 50 and 100. As of 1995, about 200 million kilograms/y were consumed[32].
HOW LIPID DROPLETS ARE ANALOGOUS TO LIPOPROTEINS
There are common structural features that lipid droplets and plasma lipoproteins both share. Also neutral lipid cores encased in polar lipid monolayers are found in both lipid droplets and plasma lipoproteins. Another common thing these two both have is their decoration with specific surface proteins which often compose of amphipathic α-helices. However, lipid droplets (with diameters of 100nm up to 100 microns) are found to be generally larger than lipoproteins (diameters ranging from less than 20nm to approximatedly 500nm) Although both lipid droplets and lipoproteins are formed via the linkage to neutral lipid synthesis in the endoplasmic reticulum, they function differently in the sense of physiology: lipids are primarily stored in lipid droplets while they are distributed in the body by lipoproteins. Also, a few cell types such as enterocytes, hepatocytes and yolk sac endodermal cells) express the required proteins (like the microsomal TG transfer protein and apolipoprotein B) for lipoprotein assembly, while most cells create lipid droplets. Therefore, there may be an evolution of lipoproteins by adapting lipid droplets to secretion. It is mostly unknown how cells can regulate the storage in lipid droplets versus secretion on lipoproteins. Only after secretory cells exceed the capacity to assemble and secrete lipoproteins, lipids in lipid droplets may be stored in these secretory cells. Alternatively, it is possible that when a storage threshold is achieved secretion may be activated. It is also possible that the lipid droplet pool of TG may be directed toward lipoprotein formation by some specific lipid droplet proteins. It is thought that secretion of TG via lipoproteins involves their hydrolysis not only at lipid droplets but also at resynthesis in the endoplasmic reticulum.
REMOVAL OF LIPID DROPLET PROTEINS Due to their analogy, it is quite similar in the mechanisms to remove lipid droplet proteins as well as those to remove membrane proteins. In lyposomes there is a degradation of endocytosed membrane proteins or autophagic vesicles. Proteasomes can also degrade the endoplasmic reticulum membrane proteins ( it is called endoplasmic reticulum – associated degradation). Lipid droplet proteins may be degraded by these both mechanisms. Lipid droplets are brought to autophagosomes before the modification of some Lipid droplet proteins by ubiquitin for proteasomal degradation can occur. Additionally, proteasome components are identified as required for normal Lipid droplet morphology by a nonbiased screen. It was recently identified how lipid droplets and proteasomal degradation are linked molecularly. Among lipid droplets proteins, ubiquitin E2 ligase (Ube2g2) is bound to the ancient ubiquitouos protein 1(Aup1) which is located either directly on lipid droplets or in the endoplasmic reticulum. The ubiquitin E3 ligase (spartin/SPG20) localizes to lipid droplets, and the lipid droplets accumulation is led by its depletion or overexpression. Then it is implied from these findings that lipid droplet – associated proteins are degraded by a specific machinery but the details of this remain unknown. It is likely that some endoplasmic reticulum – associated degradation substrate may localize to lipid droplets but at least it is not required for endoplasmic reticulum – associated degradation to have lipid droplets.
References
[edit | edit source]- ↑ a b c Rigaudy, J. (1979). Nomenclature of Organic Chemistry. Pergamon. ISBN 0080223699. OCLC 5008199.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ↑ "The Nomenclature of Lipids. Recommendations, 1976". European Journal of Biochemistry. 79 (1): 11–21. 1977. doi:10.1111/j.1432-1033.1977.tb11778.x.
{{cite journal}}: More than one of|work=and|journal=specified (help) - ↑ Medscape: Free CME, Medical News, Full-text Journal Articles & More
- ↑ Christopher Beermann1, J Jelinek1, T Reinecker2, A Hauenschild2, G Boehm1, and H-U Klör2, "Short term effects of dietary medium-chain fatty acids and n-3 long-chain polyunsaturated fatty acids on the fat metabolism of healthy volunteers"
- ↑ http://en.wikipedia.org/w/index.php?title=Essential_fatty_acid&oldid=422687249
- ↑ http://en.wikipedia.org/w/index.php?title=Essential_fatty_acid&oldid=422687249
- ↑ http://en.wikipedia.org/w/index.php?title=Fatty_acid_synthase&oldid=416140503
- ↑ Chirala SS, Jayakumar A, Gu ZW, Wakil SJ (2001). "Human fatty acid synthase: role of interdomain in the formation of catalytically active synthase dimer". Proc. Natl. Acad. Sci. U.S.A. 98 (6): 3104–8. doi:10.1073/pnas.051635998. PMC 30614. PMID 11248039.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Smith S (1994). "The animal fatty acid synthase: one gene, one polypeptide, seven enzymes". FASEB J. 8 (15): 1248–59. PMID 8001737.
{{cite journal}}: Unknown parameter|month=ignored (help) - ↑ Stoops JK, Wakil SJ (1981). "Animal fatty acid synthetase. A novel arrangement of the beta-ketoacyl synthetase sites comprising domains of the two subunits". J. Biol. Chem. 256 (10): 5128–33. PMID 6112225.
{{cite journal}}: Unknown parameter|month=ignored (help) - ↑ Stoops JK, Wakil SJ (1982). "Animal fatty acid synthetase. Identification of the residues comprising the novel arrangement of the beta-ketoacyl synthetase site and their role in its cold inactivation". J. Biol. Chem. 257 (6): 3230–5. PMID 7061475.
{{cite journal}}: Unknown parameter|month=ignored (help) - ↑ Joshi AK, Rangan VS, Smith S (1998). "Differential affinity labeling of the two subunits of the homodimeric animal fatty acid synthase allows isolation of heterodimers consisting of subunits that have been independently modified". J. Biol. Chem. 273 (9): 4937–43. doi:10.1074/jbc.273.9.4937. PMID 9478938.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Rangan VS, Joshi AK, Smith S (2001). "Mapping the functional topology of the animal fatty acid synthase by mutant complementation in vitro". Biochemistry. 40 (36): 10792–9. doi:10.1021/bi015535z. PMID 11535054.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Witkowski A, Joshi AK, Rangan VS, Falick AM, Witkowska HE, Smith S (1999). "Dibromopropanone cross-linking of the phosphopantetheine and active-site cysteine thiols of the animal fatty acid synthase can occur both inter- and intrasubunit. Reevaluation of the side-by-side, antiparallel subunit model". J. Biol. Chem. 274 (17): 11557–63. doi:10.1074/jbc.274.17.11557. PMID 10206962.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Joshi AK, Rangan VS, Witkowski A, Smith S (2003). "Engineering of an active animal fatty acid synthase dimer with only one competent subunit". Chem. Biol. 10 (2): 169–73. doi:10.1016/S1074-5521(03)00023-1. PMID 12618189.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Asturias FJ, Chadick JZ, Cheung IK, Stark H, Witkowski A, Joshi AK, Smith S (2005). "Structure and molecular organization of mammalian fatty acid synthase". Nat. Struct. Mol. Biol. 12 (3): 225–32. doi:10.1038/nsmb899. PMID 15711565.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Maier T, Leibundgut M, Ban N (2008). "The crystal structure of a mammalian fatty acid synthase". Science. 321 (5894): 1315–22. doi:10.1126/science.1161269. PMID 18772430.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ Lomakin IB, Xiong Y, Steitz TA (2007). "The crystal structure of yeast fatty acid synthase, a cellular machine with eight active sites working together". Cell. 129 (2): 319–32. doi:10.1016/j.cell.2007.03.013. PMID 17448991.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Sphingolipid&oldid=422417411
- ↑ http://en.wikipedia.org/w/index.php?title=Sphingolipid&oldid=422417411
- ↑ If not otherwise specified, reference is: Marks, Dawn B.; Swanson, Todd; Sandra I Kim; Marc Glucksman (2007). Biochemistry and molecular biology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. ISBN 0-7817-8624-X.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ Banikazemi M, Desnick RJ, Astrin KH (2009-07-08). "Fabry Disease". eMedicine Pediatrics: Genetics and Metabolic Disease. Medscape. Retrieved 2010-12-31.
{{cite web}}: Cite has empty unknown parameter:|coauthors=(help)CS1 maint: multiple names: authors list (link) - ↑ Gieselmann V, Zlotogora J, Harris A, Wenger DA, Morris CP (1994). "Molecular genetics of metachromatic leukodystrophy". Hum. Mutat. 4 (4): 233–42. doi:10.1002/humu.1380040402. PMID 7866401.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Lipid&oldid=424764580
- ↑ http://en.wikipedia.org/w/index.php?title=Wax&oldid=423902940
- ↑ http://en.wikipedia.org/w/index.php?title=Wax&oldid=423902940