Organic Chemistry/Ketones and aldehydes
Aldehydes (![]() ) and ketones (
) and ketones (![]() ) are both carbonyl compounds. They are organic compounds in which the carbonyl carbon is connected to conyl carbon satisfied by a H atom, while a ketone has both its vacancies satisfied by carbon.
) are both carbonyl compounds. They are organic compounds in which the carbonyl carbon is connected to conyl carbon satisfied by a H atom, while a ketone has both its vacancies satisfied by carbon.
Naming Aldehydes and Ketones
[edit | edit source]Ketones are named by replacing the -e in the alkane name with -one. The carbon chain is numbered so that the ketone carbon, called the carbonyl group, gets the lowest number. For example, ![]() would be named 2-butanone because the root structure is butane and the ketone group is on the number two carbon.
would be named 2-butanone because the root structure is butane and the ketone group is on the number two carbon.
Alternatively, functional class nomenclature of ketones is also recognized by IUPAC, which is done by naming the substituents attached to the carbonyl group in alphabetical order, ending with the word ketone. The above example of 2-butanone can also be named ethyl methyl ketone using this method.
If two ketone groups are on the same structure, the ending -dione would be added to the alkane name, such as heptane-2,5-dione.
Aldehydes replace the -e ending of an alkane with -al for an aldehyde. Since an aldehyde is always at the carbon that is numbered one, a number designation is not needed. For example, the aldehyde of pentane would simply be pentanal.
The -CH=O group of aldehydes is known as a formyl group. When a formyl group is attached to a ring, the ring name is followed by the suffix "carbaldehyde". For example, a hexane ring with a formyl group is named cyclohexanecarbaldehyde.
Boiling Points and Bond Angles
[edit | edit source]Aldehyde and ketone polarity is characterized by the high dipole moments of their carbonyl group, which makes them rather polar molecules. They are more polar than alkenes and ethers, though because they lack hydrogen, they cannot participate in hydrogen bonding like alcohols, thus making their relative boiling points higher than alkenes and ethers, yet lower than alcohols.
Typical bond angles between the carbonyl group and its substituents show minor deviations from the trigonal planar angles of 120 degrees, with a slightly higher bond angle between the O=C-R bond than the R-C-R bond on the carbonyl carbon (with R being any substituent).
Preparing Aldehydes and Ketones
[edit | edit source]Preparing Aldehydes
[edit | edit source]Partial oxidation of primary alcohols to aldehydes
[edit | edit source]This reaction uses pyridinium chlorochromate (PCC) in the absence of water (if water is present the alcohol will be oxidized further to a carboxylic acid).
From fatty acids
[edit | edit source]
Stephen reduction
[edit | edit source]
Here sulfur is used as a poisoner so that aldehyde formed doesn't get oxidised to the carboxylic acid. See the Wikipedia article for more detail.
Rosenmund reaction
[edit | edit source]
(For solvent xylene is used)
Preparing Ketones
[edit | edit source]From Grignard reagents
[edit | edit source]
R' R' OH
| | |
| | | O-R' OR' OR'
From nitriles
[edit | edit source]RCN + R'MgX ----> RCOR'(after hydrolysis) HCN does not react with RMgX as HCN has acidic hydrogen which results in RH being formed.
From gem dihalides
[edit | edit source]RCCl2R + strong base ----> RCOR
Oppenaur oxidation
[edit | edit source]Reagent is Aluminium tert. butoxide solvent is acetone
ROH + ACETONE ----> Ketone + isopropyl alcohol this oxidation does not affect double bonds in this oxidation ketone act as a oxidizing agent this is exact opposite to merrwine pondroff reduction
Friedel-Crafts acylation of aromatic compounds
[edit | edit source]An aromatic ring reacts with a carboxylic acid chlorine (RCOCl) in the presence of AlCl3 to form an aryl ketone of the form ArCOR.
Oxidation of secondary alcohols to ketones
[edit | edit source]A secondary alcohol can be oxidised into a ketone using acidified potassium dichromate(VI) and heating under reflux.
The orange dichromate(VI) ion, Cr2O72-, is reduced to the green Cr3+(aq) ion.
Ozonolysis of alkenes
[edit | edit source]It is a reaction in which the double bond is completely broken and the alkene molecule converted into two smaller molecules.

Ozonolysis (cleavage "by ozone) is carried out in two stages: first, addition of ozone to the double bond to form an ozonide ; and second, hydrolysis of the ozonide to yield the cleavage products.
Ozone gas is passed into a solution of the alkene in some inert solvent like carbon tetrachloride; evaporation of the solvent leaves the ozonide as a viscous oil. This unstable, explosive compound is not purified, but is treated directly with water, generally in the presence of a reducing agent. If oxidising reagent is used, aldehyde or ketone if oxidisable can further oxidise into carboxylic acid which is not the case with reducing agents
In the cleavage products a doubly-bonded oxygen is found attached to each of the originally doubly-bonded carbons.
The function of the reducing agent, which is frequently zinc dust, is to prevent formation of hydrogen peroxide, which would otherwise react with the aldehydes and ketones. (Aldehydes, RCHO, are often converted into acids,rCOOH, for ease of isolation.)
Mechanism
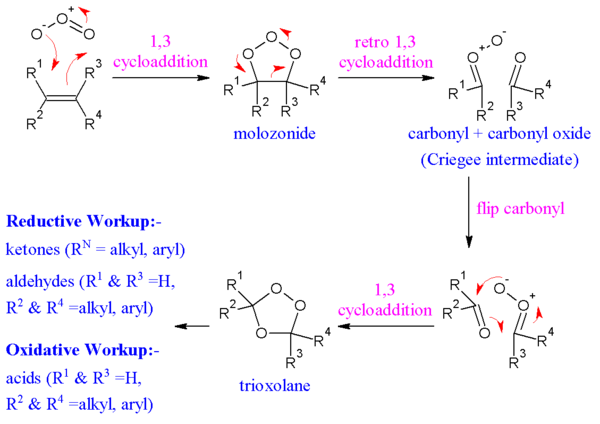
[edit | edit source]The alkene and ozone form an intermediate molozonide in a 1,3-dipolar cycloaddition. Next, the molozonide reverts to its corresponding carbonyl oxide (also called the Criegee intermediate or Criegee zwitterion) and aldehyde or ketone in a retro-1,3-dipolar cycloaddition. The oxide and aldehyde or ketone react again in a 1,3-dipolar cycloaddition or produce a relatively stable ozonide intermediate (a trioxolane)
Hydration of alkynes
[edit | edit source]Water is added to an alkyne in a strong acid. The strong acid used is sulfuric acid and mercuric acid.
Keto-enol tautomerism
[edit | edit source]In the presence of an acid (H+) or a base (OH-), the aldehyde or ketone will form an equilibrium with enols, in which the double bond of the carbonyl group migrates to form double bond between the carbonyl and the alpha (α) carbon.
In the presence of an acid, protonation of the oxygen group will occur, and water will abstract an alpha (α) hydrogen.
In the presence of a base, deprotonation of the alpha hydrogen will occur, and a hydrogen from water will be abstracted by the carbonyl oxygen.
This is an important feature of ketone and aldehydes, and is known as the keto-enolic tautomery or keto-enol tautomerism, i.e. the equilibrium of carbonyl compounds between two forms.
It must be stressed that the keto and the enol forms are two distinct compounds, not isomers. They are known as tautomers of each other. The presence of α-hydrogen is necessary for this equilibrium: those compounds not possessing it are called non-enolizable ketones.

Reactions of Aldehydes and Ketones
[edit | edit source]Reactions with the carbonyl carbon
[edit | edit source]Since aldehydes and ketones contain a polar carbonyl group, the partially positive carbon atom can act as an electrophile. Strong and weak nucleophiles are able to attack this carbonyl carbon, resulting in a net addition to the molecule.
Nucleophilic addition
[edit | edit source]With cyanide, nucleophilic addition occur to give a hydroxynitrile:
RR'C=O + CN- + H+ → RR'COHCN
e.g. propanone → 2-hydroxymethylpropanonitrile
Reactions with the carbonyl oxygen
[edit | edit source]The partially negative oxygen can act as a nucleophile, or be attacked by electrophiles.
Oxidation
[edit | edit source]Using a strong oxidizing agent such as the Tollens' Reagent (Ag2O in aqueous ammonia) acidified dichromate, Benedict's/Fehling's reagent (essentially alkaline Cu+2); aldehydes but not ketones may be oxidized into carboxylic acids. This is one way to test for the presence of an aldehyde in a sample compound: an aldehyde will become a carboxylic acid when reacted with Tollens' reagent, but a ketone will not react. when aldehydes react with fehling solution a red precipitate is obtained (due to formation of Cu2O) .
Inductive Effect and Greek letter assignment
[edit | edit source]The carbonyl group is very electron withdrawing, and adjacent carbons are effected by induction. Using the carbonyl group as a reference, adjacent carbons are named using Greek letters in order of closeness to the carbonyl group. Alpha (α) carbons are directly attached to the carbonyl group, beta (β) carbons are connected to alpha carbons, gamma (γ) to beta (β), and so on.
Due to the inductive effect of the partial positive especially prone to removal.
![{\displaystyle {\ce {Ca(COOH)2 ->[\Delta] HCHO + CaCO3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/983fac4505bd08deb768212f69be798055511c3c)
![{\displaystyle {\ce {(CH3COO)2Ca ->[\Delta] CH3COCH3 + CaCO3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/bee224b17e51525120e9299f41f75d79973725df)

![{\displaystyle {\ce {R-CN + SnCl2 + HCl -> R-CH=NH2+Cl- ->[H^+/H_2O] R-CHO}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f918cc742d9d4334dd7133ca4ccf2bbb84ded049)


