Organic Chemistry/Chirality
Introduction
[edit | edit source]Chirality (pronounced kie-RAL-it-tee) is the property of handedness. If you attempt to superimpose your right hand on top of your left, the two do not match up in the sense that your right hand's thumb overlays your left hand's pinky finger. Your two hands cannot be superimposed identically, despite the fact that your fingers of each hand are connected in the same way. Any object can have this property, including molecules.
An object that is chiral is an object that can not be superimposed on its mirror image. Chiral objects don't have a plane of symmetry. An achiral object has a plane of symmetry or a rotation-reflection axis, i.e. reflection gives a rotated version.
Optical isomers or enantiomers are stereoisomers which exhibit chirality. Optical isomerism is of interest because of its application in inorganic chemistry, organic chemistry, physical chemistry, pharmacology and biochemistry.
They are often formed when asymmetric centers are present, for example, a carbon with four different groups bonded to it. Every stereocenter in one enantiomer has the opposite configuration in the other.
When a molecule has more than one source of asymmetry, two optical isomers may be neither perfect reflections of each other nor superimposeable: some but not all stereocenters are inverted. These molecules are an example of diastereomers: They are not enantiomers. Diastereomers seldom have the same physical properties. Sometimes, the stereocentres are themselves symmetrical. This causes the counterintuitive situation where two chiral centres may be present but no isomers result. Such compounds are called meso compounds.
A mixture of equal amounts of both enantiomers is said to be a racemic mixture.
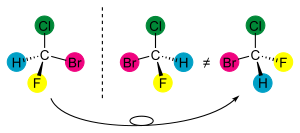
It is the symmetry of a molecule (or any other object) that determines whether it is chiral or not. Technically, a molecule is achiral (not chiral) if and only if it has an axis of improper rotation; that is, an n-fold rotation (rotation by 360°/n) followed by a reflection in the plane perpendicular to this axis which maps the molecule onto itself. A chiral molecule is not necessarily dissymmetric (completely devoid of symmetry) as it can have, e.g., rotational symmetry. A simplified rule applies to tetrahedrally-bonded carbon, as shown in the illustration: if all four substituents are different, the molecule is chiral.
It is important to keep in mind that molecules which are dissolved in solution or are in the gas phase usually have considerable flexibility and thus may adopt a variety of different conformations. These various conformations are themselves almost always chiral. However, when assessing chirality, one must use a structural picture of the molecule which corresponds to just one chemical conformation – the one of lowest energy.
Chiral Compounds With Stereocenters
[edit | edit source]Most commonly, chiral molecules have point chirality, centering around a single atom, usually carbon, which has four different substituents. The two enantiomers of such compounds are said to have different absolute configurations at this center. This center is thus stereogenic (i.e., a grouping within a molecular entity that may be considered a focus of stereoisomerism), and is exemplified by the α-carbon of amino acids.
The special nature of carbon, its ability to form four bonds to different substituents, means that a mirror image of the carbon with four different bonds will not be the same as the original compound, no matter how you try to rotate it. Understanding this is vital because the goal of organic chemistry is understanding how to use tools to synthesize a compound with the desired chirality, because a different arrangement may have no effect, or even an undesired one.
A carbon atom is chiral if it has four different items bonded to it at the same time. Most often this refers to a carbon with three heteroatoms and a hydrogen, or two heteroatoms plus a bond to another carbon plus a bond to a hydrogen atom. It can also refer to a nitrogen atom bonded to four different types of molecules, if the nitrogen atom is utilizing its lone pair as a nucleophile. If the nitrogen has only three bonds it is not chiral, because the lone pair of electrons can flip from one side of the atom to the other spontaneously.
Any atom in an organic molecule that is bonded to four different types of atoms or chains of atoms can be considered "chiral".
If a carbon atom (or other type of atom) has four different substituents, that carbon atom forms a chiral center (also known as a stereocenter). Chiral molecules often have one or more stereocenters. When drawing molecules, stereocenters are usually indicated with an asterisk near the carbon.
Example:
Left: The carbon atom has a Cl, a Br, and 2 CH3. That's only 3 different substituents, which means this is not a stereocenter.
Center: The carbon atom has one ethyl group (CH2CH3), one methyl group (CH3) and 2 H. This is not a stereocenter.
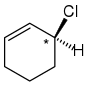
Right: The carbon atom has a Cl and 1 H. Then you must look around the ring. Since one side has a double bond and the other doesn't, it means the substituents off that carbon are different. The 4 different substituents make this carbon a stereocenter and makes the molecule chiral.
A molecule can have multiple chiral centers without being chiral overall: It is then called a meso compound. This occurs if there is a symmetry element (a mirror plane or inversion center) which relates the chiral centers.
Fischer projections
[edit | edit source]Fischer projections (after the German chemist Hermann Emil Fischer) are an ingenious means for representing configurations of carbon atoms. Considering the carbon atom as the center, the bonds which extend towards the viewer are placed horizontally. Those extending away from the viewer are drawn vertically. This process, when using the common dash and wedge representations of bonds, yields what is sometimes referred to as the "bowtie" drawing due to its characteristic shape. This representation is then further shorthanded as two lines: the horizontal (forward) and the vertical (back), as showed in the figure below:

Operations on Fischer projections
[edit | edit source]- in a Fischer projection, exchange two substituent positions results in the inversion of the stereocenter
- rotation by 90° of the Fischer projection results in inversion
- rotation by 180° of the Fischer projection preserves the configuration
Naming conventions
[edit | edit source]There are three main systems for describing configuration: the oldest, the relative whose use is now deprecated, and the current, or absolute. The relative configuration description is still used mainly in glycochemistry. Configuration can also be assigned on the purely empirical basis of the optical activity.
By optical activity: (+)- and (-)-
[edit | edit source]An optical isomer can be named by the direction in which it rotates the plane of polarized light. If an isomer rotates the plane clockwise as seen by a viewer towards whom the light is traveling, that isomer is labeled (+). Its counterpart is labeled (-). The (+) and (-) isomers have also been termed d- and l-, respectively (for dextrorotatory and levorotatory). This labeling is easy to confuse with D- and L- and is therefore not encouraged by IUPAC.
The fact that an enantiomer can rotate polarised light clockwise (d- or +- enantiomer) does not relate with the relative configuration (D- or L-) of it.
By relative configuration: D- and L-
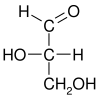
[edit | edit source]Fischer, whose research interest was in carbohydrate chemistry, took glyceraldehyde (the simplest sugar, systematic name 2,3-dihydroxypropanal) as a template chiral molecule and denoted the two possible configurations with D- and L-, which rotated polarised light clockwise and counterclockwise, respectively.

All other molecules are assigned the D- or L- configuration if the chiral centre can be formally obtained from glyceraldehyde by substitution. For this reason the D- or L- naming scheme is called relative configuration.
|
An optical isomer can be named by the spatial configuration of its atoms. The D/L system does this by relating the molecule to glyceraldehyde. Glyceraldehyde is chiral itself, and its two isomers are labeled D and L. Certain chemical manipulations can be performed on glyceraldehyde without affecting its configuration, and its historical use for this purpose (possibly combined with its convenience as one of the smallest commonly-used chiral molecules) has resulted in its use for nomenclature. In this system, compounds are named by analogy to glyceraldehyde, which generally produces unambiguous designations, but is easiest to see in the small biomolecules similar to glyceraldehyde.

One example is the amino acid alanine: alanine has two optical isomers, and they are labeled according to which isomer of glyceraldehyde they come from. Glycine, the amino acid derived from glyceraldehyde, incidentally, does not retain its optical activity, since its central carbon is not chiral. Alanine, however, is essentially methylated glycine and shows optical activity.
The D/L labeling is unrelated to (+)/(-); it does not indicate which enantiomer is dextrorotatory and which is levorotatory. Rather, it says that the compound's stereochemistry is related to that of the dextrorotatory or levorotatory enantiomer of glyceraldehyde. Nine of the nineteen L-amino acids commonly found in proteins are dextrorotatory (at a wavelength of 589 nm), and D-fructose is also referred to as levulose because it is levorotatory.
The dextrorotatory isomer of glyceraldehyde is in fact the D isomer, but this was a lucky guess. At the time this system was established, there was no way to tell which configuration was dextrorotatory. (If the guess had turned out wrong, the labeling situation would now be even more confusing.)
A rule of thumb for determining the D/L isomeric form of an amino acid is the "CORN" rule. The groups:
- COOH, R, NH2 and H (where R is an unnamed carbon chain)
are arranged around the chiral center carbon atom. If these groups are arranged clockwise around the carbon atom, then it is the L-form. If counter-clockwise, it is the D-form.This rule only holds when the hydrogen atom is pointing out of the page.[1]
By absolute configuration: R- and S-
[edit | edit source]Main article: R-S System
The absolute configuration system stems from the Cahn-Ingold-Prelog priority rules, which allow a precise description of a stereocenter without using any reference compound. In fact the basis is now the atomic number of the stereocenter substituents.
The R/S system is another way to name an optical isomer by its configuration, without involving a reference molecule such as glyceraldehyde. It labels each chiral center R or S according to a system by which its ligands are each assigned a priority, according to the Cahn Ingold Prelog priority rules, based on atomic number.
This system labels each chiral center in a molecule (and also has an extension to chiral molecules not involving chiral centers). It thus has greater generality than the D/L system, and can label, for example, an (R,R) isomer versus an (R,S) — diastereomers.
The R/S system has no fixed relation to the (+)/(-) system. An R isomer can be either dextrorotatory or levorotatory, depending on its exact ligands.
The R/S system also has no fixed relation to the D/L system. For example, one of glyceraldehyde's ligands is a hydroxy group, -OH. If a thiol group, -SH, were swapped in for it, the D/L labeling would, by its definition, not be affected by the substitution. But this substitution would invert the molecule's R/S labeling, due to the fact that sulfur's atomic number is higher than carbon's, whereas oxygen's is lower. [Note: This seems incorrect. Oxygen has a higher atomic number than carbon. Sulfur has a higher atomic number than oxygen. The reason the assignment priorities change in this example is because the CH2SH group gets a higher priority than the CHO, whereas in glyceraldehyde the CHO takes priority over the CH2OH.]
For this reason, the D/L system remains in common use in certain areas, such as amino acid and carbohydrate chemistry. It is convenient to have all of the common amino acids of higher organisms labeled the same way. In D/L, they are all L. In R/S, they are not, conversely, all S — most are, but cysteine, for example, is R, again because of sulfur's higher atomic number.
The word “racemic” is derived from the Latin word for grape; the term having its origins in the work of Louis Pasteur who isolated racemic tartaric acid from wine.
Chiral Compounds Without Stereocenters
[edit | edit source]It is also possible for a molecule to be chiral without having actual point chirality (stereocenters). Commonly encountered examples include 1,1'-bi-2-naphthol (BINOL) and 1,3-dichloro-allene which have axial chirality, and (E)-cyclooctene which has planar chirality.
For example, the isomers which are shown by the following figure are different. The two isomers cannot convert from one to another spontaneously because of restriction of rotation of double bonds.

Other types of chiral compounds without stereocenters (like restriction of rotation of a single bond because of steric hindrance) also exist. Consider the following example of the R and S binol molecules:
The biphenyl C-C bond cannot rotate if the X and Y groups cause steric hindrance.
This compound exhibits spiral chirality.
Properties of optical isomers
[edit | edit source]Enantiomers have – when present in a symmetric environment – identical chemical and physical properties except for their ability to rotate plane-polarized light by equal amounts but in opposite directions. A solution of equal parts of an optically-active isomer and its enantiomer is known as a racemic solution and has a net rotation of plane-polarized light of zero.
Enantiomers differ in how they interact with different optical isomers of other compounds. In nature, most biological compounds (such as amino acids) occur as single enantiomers. As a result, different enantiomers of a compound may have substantially different biological effects. Different enantiomers of the same chiral drug can have very different pharmological effects, mainly because the proteins they bind to are also chiral.
For example, spearmint leaves and caraway seeds respectively contain L-carvone and D-carvone – enantiomers of carvone. These smell different to most people because our taste receptors also contain chiral molecules which behave differently in the presence of different enantiomers.

D-form Amino acids tend to taste sweet, whereas L-forms are usually tasteless. This is again due to our chiral taste molecules. The smells of oranges and lemons are examples of the D and L enantiomers.
Penicillin's activity is stereoselective. The antibiotic only works on peptide links of D-alanine which occur in the cell walls of bacteria – but not in humans. The antibiotic can kill only the bacteria, and not us, because we don't have these D-amino acids.
The electric and magnetic fields of polarized light oscillate in a geometric plane. An axis normal to this plane gives the direction of energy propagation. Optically active isomers rotate the plane that the fields oscillate in. The polarized light is actually rotated in a racemic mixture as well, but it is rotated to the left by one of the two enantiomers, and to the right by the other, which cancel out to zero net rotation.
Chirality in biology
[edit | edit source]Many biologically-active molecules are chiral, including the naturally-occurring amino acids (the building blocks of proteins), and sugars. Interestingly, in biological systems most of these compounds are of the same chirality: most amino acids are L and sugars are D. The origin of this homochirality in biology is the subject of much debate.

Chiral objects have different interactions with the two enantiomers of other chiral objects. Enzymes, which are chiral, often distinguish between the two enantiomers of a chiral substrate. Imagine an enzyme as having a glove-like cavity which binds a substrate. If this glove is right handed, then one enantiomer will fit inside and be bound while the other enantiomer will have a poor fit and is unlikely to bind.
Chirality in inorganic chemistry
[edit | edit source]
Many coordination compounds are chiral; for example the well-known [Ru(2,2'-bipyridine)3]2+ complex in which the three bipyridine ligands adopt a chiral propeller-like arrangement [7]. In this case, the Ru atom may be regarded as a stereogenic centre, with the complex having point chirality. The two enantiomers of complexes such as [Ru(2,2'-bipyridine)3]2+ may be designated as Λ (left-handed twist of the propeller described by the ligands) and Δ (right-handed twist). Hexol is a chiral cobalt compound.
More definitions
[edit | edit source]- Any non-racemic chiral substance is called scalemic
- A chiral substance is enantiopure or homochiral when only one of two possible enantiomers is present.
- A chiral substance is enantioenriched or heterochiral when an excess of one enantiomer is present but not to the exclusion of the other.
- Enantiomeric excess or ee is a measure for how much of one enantiomer is present compared to the other. For example, in a sample with 40% ee in R, the remaining 60% is racemic with 30% of R and 30% of S, so that the total amount of R is 70%.
Enantiopure preparations
[edit | edit source]Several strategies exist for the preparation of enantiopure compounds. The first method is the separation of a racemic mixture into its isomers. Louis Pasteur in his pioneering work was able to isolate the isomers of tartaric acid because they crystallize from solution as crystals with differing symmetry. A less common and more recently discovered method is by enantiomer self-disproportionation, which is an advanced technique involving the separation of a primarily racemic fraction from a nearly enantiopure fraction via column chromatography.
In a non-symmetric environment (such as a biological environment) enantiomers may react at different speeds with other substances. This is the basis for chiral synthesis, which preserves a molecule's desired chirality by reacting it with or catalyzing it with chiral molecules capable of maintaining the product's chirality in the desired conformation (using certain chiral molecules to help it keep its configuration). Other methods also exist and are used by organic chemists to synthesize only (or maybe only mostly) the desired enantiomer in a given reaction.
Enantiopure medications
[edit | edit source]Advances in industrial chemical processes have allowed pharmaceutical manufacturers to take drugs that were originally marketed in racemic form and divide them into individual enantiomers, each of which may have unique properties. For some drugs, such as zopiclone, only one enantiomer (eszopiclone) is active; the FDA has allowed such once-generic drugs to be patented and marketed under another name. In other cases, such as ibuprofen, both enantiomers produce the same effects. Steroid receptor sites also show stereoisomer specificity.
Examples of racemic mixtures and enantiomers that have been marketed include:
- Ofloxacin (Floxin) and Levofloxacin (Levaquin)
- Bupivacaine (Marcaine) and Ropivacaine (Naropin)
- Methylphenidate (Ritalin) and Dexmethylphenidate (Focalin)
- Cetirizine (Zyrtec) and Levocetirizine (Xyzal)
- Albuterol (Ventolin) and Levalbuterol (Xopenex)
- Omeprazole (Prilosec) and Esomeprazole (Nexium)
- Citalopram (Celexa / Cipramil) and Escitalopram (Lexapro / Cipralex)
- Zopiclone (Imovane) and Eszopiclone (Lunesta)
- Modafinil (Provigil) and Armodafinil (Nuvigil) — sulfur is the chiral center in modafinil, instead of carbon.
Many chiral drugs must be made with high enantiomeric purity due to potential side-effects of the other enantiomer. (The other enantiomer may also merely be inactive.)
Consider a racemic sample of thalidomide. One enantiomer was thought to be effective against morning sickness while the other is now known to be teratogenic. Unfortunately, in this case administering just one of the enantiomers to a pregnant patient would still be very dangerous as the two enantiomers are readily interconverted in vivo. Thus, if a person is given either enantiomer, both the D and L isomers will eventually be present in the patient's serum and so chemical processes may not be used to mitigate its toxicity.

See also
[edit | edit source]
<< Haloalkanes | Stereochemistry | Alcohols >>