Organic Chemistry/Alkanes
Alkanes are the simplest organic molecules, consisting solely of singly-bonded carbon and hydrogen atoms. Alkanes are used as the basis for naming the majority of organic compounds (their nomenclature). Alkanes have the general formula CnH2n+2. Although their reactivities are often rather uninteresting, they provide an excellent basis for understanding bonding, conformation, and other important concepts which can be generalized to more "useful" molecules.
Introduction
[edit | edit source]
An example of an alkane
Alkanes are the simplest and the least reactive hydrocarbon species containing only carbons and hydrogens. They are commercially very important, for being the principal constituent of gasoline and lubricating oils and are extensively employed in organic chemistry; though the role of pure alkanes (such as hexanes) is delegated mostly to solvents.
The distinguishing feature of an alkane, making it distinct from other compounds that also exclusively contain carbon and hydrogen, is its lack of unsaturation. That is to say, it contains no double or triple bonds, which are highly reactive in organic chemistry.
Though not totally devoid of reactivity, their lack of reactivity under most laboratory conditions makes them a relatively uninteresting, though very important component of organic chemistry. As you will learn about later, the energy confined within the carbon-carbon bond and the carbon-hydrogen bond is quite high and their rapid oxidation produces a large amount of heat, typically in the form of fire.
As said it is important, not considered very important component in the chemistry.
Introductory Definitions
[edit | edit source]Organic compounds contain carbon and hydrogen by definition and usually other elements (e.g. nitrogen and oxygen) as well. (CO2 is not an organic compound because it has no hydrogen).
Hydrocarbons are organic compounds that contain carbon and hydrogen only.
Alkanes are hydrocarbons or organic compounds made up of only carbon-carbon single bonds.Hence they are saturated. (as opposed to double and triple bonds). The simplest alkane is methane.
Methane
[edit | edit source]Methane, (CH4, one carbon bonded to four hydrogens) is the simplest organic molecule. It is a gas at standard temperature and pressure (STP).

|
| Methane |
This is a flattened, two-dimensional representation of methane that you will see commonly. The true three-dimensional form of methane does not have any 90 degree angles between bonded hydrogens. The bonds point to the four corners of a tetrahedron, forming cos-1(-1/3) ≈ 109.5 degree bond angles.

Ethane
[edit | edit source]Two carbons singly bonded to each other with six hydrogens is called ethane.
Ethane is the second simplest hydrocarbon molecule. It can be thought of as two methane molecules attached to each other, but with two fewer hydrogen atoms. Note that, if we were simply to create a new bond between the carbon centers of two methane molecules, this would violate the octet rule for the involved atoms.
There are several common methods to draw organic molecules. They are often used interchangeably, although some methods work better for one situation or another. It is important to be familiar with the common methods, as these are the "languages" organic chemists can use to discuss structure with one another.
Drawing alkanes
[edit | edit source]When writing out the alkane structures, you can use different levels of the shorthand depending on the needs at hand in hand. For example, pentane can be written out. Its formula is C5H12.
or CH3–CH2–CH2–CH2–CH3,
or CH3(CH2)3CH3,
Line drawing shorthand
[edit | edit source]Although non-cyclic alkanes are called straight-chain alkanes they are technically made of linked chains. This is reflected in the line-drawing method. Each ending point and bend in the line represents one carbon atom and each short line represents one single carbon-carbon bond. Every carbon is assumed to be surrounded with a maximum number of hydrogen atoms unless shown otherwise.
| Propane, butane, pentane |
Structures drawn without explicitly showing all carbon atoms are often called "skeletal" structures, since they represent the skeleton or the backbone of the molecule. In organic chemistry, carbon is very frequently used, so chemists know that there is a carbon atom at the endpoints of every line that is not specifically labeled.
Conformations
[edit | edit source]Conformers, also called conformational isomers, or rotational isomers,or rotomers are arrangements of the same molecule made transiently different by the rotation in space about one or more single bonds.
Other types of isomer can only be converted from one form to another by breaking bonds, but conformational isomers can be made simply by rotating bonds.
Newman projections
[edit | edit source]Newman projections are drawings used to represent different positions of parts of molecules relative to each other in space. Remember that single bonds can rotate in space if not impeded. Newman projections represent different positions of rotating molecule parts.
 |
 |
|
|
(front end overlaps the back and also unstable) |
Conformations and energy
[edit | edit source]Different conformations have different potential energies. The staggered conformation is at a lower potential energy than the eclipsed conformation, and is favored. In ethane, the barrier to rotation is approximately 25 kJ/mol, indicating that each pair of eclipsed hydrogens raises the energy by about 8 kJ/mol. This number also applies to other organic compounds which have hydrogen atoms at similar distances from each other. At very low temperatures all conformations revert to the stabler( due to minimized vibration of atoms at it's mean position) , lower energy staggered conformation.
Steric effects
[edit | edit source]Steric effects have to do with size. Two bulky objects run into each other and invade each others space. If we replace one or more hydrogen atoms on the above Newman projections with a methyl or other group, the potential energy goes up especially for the eclipsed conformations.
Lets look at a Newman projection of butane as it rotates counterclockwise around its axes.
 |
 |
 |
 | |||
When the larger groups overlap they repel each other more strongly than hydrogen groups do, and the potential energy goes up.
Entropy
[edit | edit source]Entropy, represented as a ΔS, is a mathematical construct that represents disorder or probability. Natural systems want to find the lowest energy or organization possible, which translates to the highest entropy.
A note about potential energy: If you are rusty on this, remember the analogy of a big rock pushed to the top of a hill. At the top it has a maximum of potential energy. When you push it and allow it to roll down the hill the potential energy stored in it is transformed into kinetic energy that can be used to generate heat or smash something.
Notice that statistically, the ethane molecule has twice as many opportunities to be in the gauche conformation as in the anti conformation. However, because the Gauche configuration brings the methyl groups closer together in space, this generates high energy steric interactions and do not occur without the input of energy. Thus, the butane molecules shown will almost never be found in such unfavorable conformations.
Preparation of Alkanes
[edit | edit source]Wurtz reaction
[edit | edit source]Wurtz reaction is coupling of haloalkanes using sodium metal in solvent like dry ether
2R-X + 2Na → R-R + 2Na+X−
Mechanism
[edit | edit source]The reaction consists of a halogen-metal exchange involving the free radical species R• (in a similar fashion to the formation of a Grignard reagent and then the carbon-carbon bond formation in a nucleophilic substitution reaction.)
One electron from the metal is transferred to the halogen to produce a metal halide and an alkyl radical.
- R-X + M → R• + M+X−
The alkyl radical then accepts an electron from another metal atom to form an alkyl anion and the metal becomes cationic. This intermediate has been isolated in a several cases.
- R• + M → R−M+
The nucleophilic carbon of the alkyl anion then displaces the halide in an SN2 reaction, forming a new carbon-carbon covalent bond.
- R−M+ + R-X → R-R + M+X−
- COREY-HOUSE reactioN
- [Also called as 'coupling of alkyl halides with organo metallic compounds']
- It is a better method than wurtz reaction. An alkyl halides and a lithium dialkyl copper are reacted to give a higher hydrocarbon
- R'-X + R2CuLi---->R-R' + R-Cu + LiX
- (R and R' may be same or different)
- It
Clemmensen reduction
[edit | edit source]Clemmensen reduction is a reduction of ketones (or aldehydes) to alkanes using zinc amalgam and hydrochloric acid
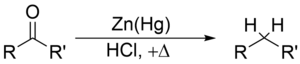
The Clemmensen reduction is particularly effective at reducing aryl-alkyl ketones. With aliphatic or cyclic ketones, zinc metal reduction is much more effective
The substrate must be stable in the strongly acidic conditions of the Clemmensen reduction. Acid sensitive substrates should be reacted in the Wolff-Kishner reduction, which utilizes strongly basic conditions; a further, milder method is the Mozingo reduction. As a result of Clemmensen Reduction, the carbon of the carbonyl group involved is converted from sp2 hybridisation to sp3 hybridisation. The oxygen atom is lost in the form of one molecule of water.
Wolff-Kishner reduction
[edit | edit source]
The Wolff–Kishner reduction is a chemical reaction that fully reduces a ketone (or aldehyde) to an alkane. Condensation of the carbonyl compound with hydrazine forms the hydrazone, and treatment with base induces the reduction of the carbon coupled with oxidation of the hydrazine to gaseous nitrogen, to yield the corresponding alkane.
Mechanism
[edit | edit source]
The mechanism first involves the formation of the hydrazone in a mechanism that is probably analogous to the formation of an imine. Successive deprotonations eventually result in the evolution of nitrogen. The mechanism can be justified by the evolution of nitrogen as the thermodynamic driving force. This reaction is also used to distinguish between aldehydes and ketones.
Mozingo Reduction
[edit | edit source]A thioketal is first produced by reaction of the ketone with an appropriate thiol. The product is then hydrogenolyzed to the alkane, using Raney nickel

Properties of Alkanes
[edit | edit source]Alkanes are not very reactive when compared with other chemical species. This is because the backbone carbon atoms in alkanes have attained their octet of electrons through forming four covalent bonds (the maximum allowed number of bonds under the octet rule; which is why carbon's valence number is 4). These four bonds formed by carbon in alkanes are sigma bonds, which are more stable than other types of bond because of the greater overlap of carbon's atomic orbitals with neighboring atoms' atomic orbitals. To make alkanes react, the input of additional energy is needed; either through heat or radiation.
Gasoline is a mixture of the alkanes and unlike many chemicals, can be stored for long periods and transported without problem. It is only when ignited that it has enough energy to continue reacting. This property makes it difficult for alkanes to be converted into other types of organic molecules. (There are only a few ways to do this). Alkanes are also less dense than water, as one can observe, oil, an alkane, floats on water.
Alkanes are non-polar solvents. Since only C and H atoms are present, alkanes are nonpolar. Alkanes are immiscible in water but freely miscible in other non-polar solvents. Alkanes consisting of weak dipole dipole bonds can not break the strong hydrogen bond between water molecules hence it is not miscible in water. The same character is also shown by alkenes. Because alkanes contain only carbon and hydrogen, combustion produces compounds that contain only carbon, hydrogen, and/or oxygen. Like other hydrocarbons, combustion under most circumstances produces mainly carbon dioxide and water. However, alkanes require more heat to combust and do not release as much heat when they combust as other classes of hydrocarbons. Therefore, combustion of alkanes produces higher concentrations of organic compounds containing oxygen, such as aldehydes and ketones, when combusting at the same temperature as other hydrocarbons.
The general formula for alkanes is CNH2N+2; the simplest possible alkane is therefore methane, CH4. The next simplest is ethane, C2H6; the series continues indefinitely. Each carbon atom in an alkane has sp³ hybridization.
Alkanes are also known as paraffins, or collectively as the paraffin series. These terms are also used for alkanes whose carbon atoms form a single, unbranched chain. Branched-chain alkanes are called isoparaffins.
Methane through Butane are very flammable gases at standard temperature and pressure (STP). Pentane is an extremely flammable liquid boiling at 36 °C and boiling points and melting points steadily increase from there; octadecane is the first alkane which is solid at room temperature. Longer alkanes are waxy solids; candle wax generally has between C20 and C25 chains. As chain length increases ultimately we reach polyethylene, which consists of carbon chains of indefinite length, which is generally a hard white solid.
Chemical properties
[edit | edit source]Alkanes react only very poorly with ionic or other polar substances. The pKa values of all alkanes are above 50, and so they are practically inert to acids and bases. This inertness is the source of the term paraffins (Latin para + affinis, with the meaning here of "lacking affinity"). In crude oil the alkane molecules have remained chemically unchanged for millions of years.
However redox reactions of alkanes, in particular with oxygen and the halogens, are possible as the carbon atoms are in a strongly reduced condition; in the case of methane, the lowest possible oxidation state for carbon (−4) is reached. Reaction with oxygen leads to combustion without any smoke; with halogens, substitution. In addition, alkanes have been shown to interact with, and bind to, certain transition metal complexes.
Free radicals, molecules with unpaired electrons, play a large role in most reactions of alkanes, such as cracking and reformation where long-chain alkanes are converted into shorter-chain alkanes and straight-chain alkanes into branched-chain isomers.
In highly branched alkanes and cycloalkanes, the bond angles may differ significantly from the optimal value (109.5°) in order to allow the different groups sufficient space. This causes a tension in the molecule, known as steric hinderance, and can substantially increase the reactivity. The same is preferred for alkenes too.
Introduction to Nomenclature
[edit | edit source]Before we can understand reactions in organic chemistry, we must begin with a basic knowledge of naming the compounds. The IUPAC nomenclature is a system on which most organic chemists have agreed to provide guidelines to allow them to learn from each others' works. Nomenclature, in other words, provides a foundation of language for organic chemistry.
The names of all alkanes end with -ane. Whether or not the carbons are linked together end-to-end in a ring (called cyclic alkanes or cycloalkanes) or whether they contain side chains and branches, the name of every carbon-hydrogen chain that lacks any double bonds or functional groups will end with the suffix -ane.
Alkanes with unbranched carbon chains are simply named by the number of carbons in the chain. The first four members of the series (in terms of number of carbon atoms) are named as follows:
- CH4 = methane = one hydrogen-saturated carbon
- C2H6 = ethane = two hydrogen-saturated carbons
- C3H8 = propane = three hydrogen-saturated carbons
- C4H10 = butane = four hydrogen-saturated carbons
Alkanes with five or more carbon atoms are named by adding the suffix -ane to the appropriate numerical multiplier, except the terminal -a is removed from the basic numerical term. Hence, C5H12 is called pentane, C6H14 is called hexane, C7H16 is called heptane and so forth.
Straight-chain alkanes are sometimes indicated by the prefix n- (for normal) to distinguish them from branched-chain alkanes having the same number of carbon atoms. Although this is not strictly necessary, the usage is still common in cases where there is an important difference in properties between the straight-chain and branched-chain isomers: e.g. n-hexane is a neurotoxin while its branched-chain isomers are not.
Number of hydrogens to carbons
[edit | edit source]This equation describes the relationship between the number of hydrogen and carbon atoms in alkanes:
- H = 2C + 2
where "C" and "H" are used to represent the number of carbon and hydrogen atoms present in one molecule. If C = 2, then H = 6.
Many textbooks put this in the following format:
- CnH2n+2
where "Cn" and "H2n+2" represent the number of carbon and hydrogen atoms present in one molecule. If Cn = 3, then H2n+2 = 2(3) + 2 = 8. (For this formula look to the "n" for the number, the "C" and the "H" letters themselves do not change.)
Progressively longer hydrocarbon chains can be made and are named systematically, depending on the number of carbons in the longest chain.
Naming carbon chains up to twelve
[edit | edit source]- methane (1 carbon)
- ethane (2 carbons)
- propane (3 carbons)
- butane (4 carbons)
- pentane (5 carbons)
- hexane (6 carbons)
- heptane (7 carbons)
- octane (8 carbons)
- nonane (9 carbons)
- decane (10 carbons)
- undecane (11 carbons)
- dodecane (12 carbons)
The prefixes of the first three are the contribution of a German Chemist, August Wilhelm Hoffman, who also suggested the name quartane for 4 carbons in 1866. However, the but- prefix had already been in use since the 1820s and the name quartane never caught on. He also recommended the endings to use the vowels, a, e, i (or y), o, and u, or -ane, -ene, -ine or -yne, -one, and -une. Again, only the first three caught on for single, double, and triple bonds and -one was already in use for ketones. Pent, hex, hept, oct, and dec all come from the ancient Greek numbers (penta, hex, hepta, octa, deka) and oddly, non, from the Latin novem. For longer-chained alkanes we use the special IUPAC multiplying affixes. For example, pentadecane signifies an alkane with 5+10 = 15 carbon atoms. For chains of length 30, 40, 50, and so on the basic prefix is added to -contane. For example, C57H116 is named as heptapentacontane. When the chain contains 20-29 atoms we have an exception. C20H42 is known as icosane, and then we have, e.g. tetracosane (eliding the "i" when necessary). For the length 100 we have "hecta" but for 200, 300 ... 900 we have "dicta", "tricta", and so on, eliding the "i" on "icta" when necessary; for 1000 we have "kilia" but for 2000 and so on, "dilia", "trilia", and so on, eliding the "i" on "ilia" when necessary.
We then put all of the prefixes together in reverse order. The alkane with 9236 carbon atoms is then hexatridinoniliane.
Isomerism
[edit | edit source]The atoms in alkanes with more than three carbon atoms can be arranged in many ways, leading to a large number of potential different configurations (isomers). So-called "normal" alkanes have a linear, unbranched configuration, but the n- isomer of any given alkane is only one of potentially hundreds or even possibly millions of configurations for that number of carbon and hydrogen atoms in some sort of chain arrangement.
Isomerism is defined as the compound having same moleculer formula the formula which present the different moleculer formula arrangement are called as Isomerism.
e.g.- The molecular formula for butane is C4H10.
The number of isomers increases rapidly with the number of carbon atoms in a given alkane molecule; for alkanes with as few as 12 carbon atoms, there are over three hundred and fifty-five possible forms the molecule can take!
# Carbon Atoms # Isomers of Alkane 1 1 2 1 3 1 4 2 5 3 6 5 7 9 8 18 9 35 10 75 11 159 12 355
Branched chains
[edit | edit source]Carbon is able to bond in all four directions and easily forms strong bonds with other carbon atoms. When one carbon is bonded to more than two other carbons it forms a branch.
 |
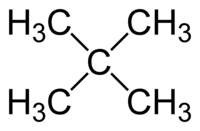 |
Above you see a carbon bonded to three and four other carbons.
- Note: a methane group is called a methyl group when it is bonded to another carbon instead of a fourth hydrogen. –CH3
The common system has naming convention for carbon chains as they relate to branching.
- n-alkanes are linear
- iso-alkanes have one branch R2CH—
- neo-alkanes have two branches R3C—
Note: "R" in organic chemistry is a placeholder that can represent any carbon group.
Constitutional isomers
[edit | edit source]One of the most important characteristics of carbon is its ability to form several relatively strong bonds per atom. It is for this reason that many scientists believe that carbon is the only element that could be the basis for the many complicated molecules needed to support a living being.
One carbon atom can have attached to it not just the one or two other carbons needed to form a single chain but can bond to up to four other carbons. It is this ability to bond multiply that allows isomerism.
Isomers are two molecules with the same molecular formula but different physical arrangements. Constitutional isomers have their atoms arranged in a different order. A constitutional isomer of butane has a main chain that is forked at the end and one carbon shorter in its main chain than butane.
 |
 |
Naming Alkanes
[edit | edit source]There are several ways or systems for the nomenclature, or naming, of organic molecules, but just two main ones.
- The traditional, non-systematic names. Many of these linger on, especially for simpler or more common molecules.
- The systematic IUPAC (eye-YOU-pack (International Union of Pure And Applied Chemistry)) names.
The IUPAC system is necessary for complicated organic compounds. It gives a series of unified rules for naming a large compound by conceptually dividing it up into smaller, more manageable nameable units.
Many traditional (non-IUPAC) names are still commonly used in industry, especially for simpler and more common chemicals, as the traditional names were already entrenched.
IUPAC naming rules
[edit | edit source]- Find the longest carbon chain, identify the end near which the most substituents are located, and number the carbons sequentially from that end. This will be the parent chain.
- Consider all other carbon groups as substituents.
- Alphabetize the substituents.
- Number the substituents according to the carbon to which they are attached. If numbering can be done in more than one way, use the numbering system that results in the smallest numbers.
Substituents are named like a parent, and replacing the -ane ending with -yl.
Numbering
[edit | edit source]The above molecule is numbered as follows:
2,3,7-Trimethyloctane
Not 2,6,7-Trimethyloctane. Remember, number so as to give the smallest numbers to the substituents.
Alphabetizing
[edit | edit source]3-Ethyl-3-methylpentane
Ethyl is listed before methyl for alphabetizing purposes.
Branched Substituents
[edit | edit source]Naming branched substituents
[edit | edit source]3-(1-methylethyl)-2,4-dimethylpentane
The main chain in the drawing is numbered 1-5. The main part of the branched substituent, an ethyl group, is numbered 1' and 2'. The methyl substituent off of the ethyl substituent is not numbered in the drawing.
To name the compound, put the whole branched substituent name in parentheses and then number and alphabetize as if a simple substituent.
Common system
[edit | edit source]Some prefixes from the common system are accepted in the IUPAC system.
For alphabetization purposes, iso- and neo- are considered part of the name, and alphabetized. Sec- and tert- are not considered an alphabetizable part of the name.
(In the following images, the R- represents any carbon structure.)
Iso-
[edit | edit source]Iso- can be used for substituents that branch at the second-to-last carbon and end with two methyls. An isobutyl has four carbons total:
Sec-
[edit | edit source]Sec- can be used for substituents that branch at the first carbon .
Neo-
[edit | edit source]Neo- refers to a substituent whose second-to-last carbon of the chain is trisubstituted (has three methyl groups attached to it). A neo-pentyl has five carbons total.
Tert-
[edit | edit source]Tert- is short for tertiary and refers to a substituent whose first carbon has three other carbon groups attached to it.
See also
[edit | edit source]![]() || << Foundational concepts | Alkanes | Stereochemistry >>
|| << Foundational concepts | Alkanes | Stereochemistry >>







