OCR Advanced GCE in Chemistry/Periodic Table: Transition elements

Electronic structures
[edit | edit source]A transitional element is defined as a d-block element forming one or more stable ions with incompletely filled d-orbitals.
If we look at some examples of the transitional elements electronic configurations we can see how this works:
Scandium (Sc) - [Ar] 3d1 4s2
titanium (Ti) - [Ar] 3d2 4s2
Remembering that electrons are first removed from the 4s sub-shell:
titanium can form 2+ 3+ (and 4+) both of which result in ions with incomplete d-orbitals
However, scandium only forms 3+ ions and they have an empty d sub-shell. Therefore scandium is not classified as a transitional element. Zinc is the same because it only form 2+ ions, loosing two electrons from the 4s sub-shell and maintaining a full d sub-shell.
So now we have a nice pattern of increasing electron number in the 3d sub-shell in the group 4 transition elements. However as always there are exceptions to this rule:
One would expect chromium to have an electronic structure as follows:
[Ar] 3d4 4s2
but actually it has the structure:
[Ar] 3d5 4s1
This is because half filled sub-shells and completely filled sub-shells are the most stable so they are favoured. This is the same in copper:
[Ar] 3d10 4s1
Where having a complete d sub-shell is preferred to having 9 electrons and 2 in the 4s sub-shell
Oxidation states, coloured ions and catalytic behaviour
[edit | edit source]The transitional elements can form compounds in multiple oxidation sates. Most elements only form compounds in one oxidation state so transitional elements are rare. The reason that this happens is to do with the energy levels between the 4s sub-shell and within the d sub-shell; they are all very similar so movement of multiple electrons doesn't require too much energy.
The main oxidation states of iron and copper compounds (the ones that you must know) are as follows:
Fe +2 +3 +4 +5 +6
Cu +1 +2 +3
With the most common oxidation states in bold
Transitional metal ions are coloured in aqueous solutions because they have partly filled d orbitals (this is discovered more in the transitional elements exam). This therefore suggests that compounds of zinc and scandium will not have colour - this is true.
The most important coloured ions to learn are:
Fe2+ green
Fe3+ yellow
Cu2+ blue
By having partly fill d orbitals transitional metals can absorb light in our visible spectrum. This means that the light that is reflected (the light we see) will have the absorbed wavelength of light missing. Therefore the wavelength and colour of absorbed light is different to the wavelength and colour of the light we see.

This colour wheel demonstrates which colour a compound will appear if it only has one absorption in the visible spectrum. For example, if the compound absorbs red light, it will appear green.
λ absorbed versus colour observed
400nm Violet absorbed, Green-yellow observed (λ 560nm)
450nm Blue absorbed, Yellow observed (λ 600nm)
490nm Blue-green absorbed, Red observed (λ 620nm)
570nm Yellow-green absorbed, Violet observed (λ 410nm)
580nm Yellow absorbed, Dark blue observed (λ 430nm)
600nm Orange absorbed, Blue observed (λ 450nm)
650nm Red absorbed, Green observed (λ 520nm)
Precipitation reactions
[edit | edit source]There are 3 precipitation reactions in this section which must be learnt with appropriate observations remembered. They are all very similar with a solution of transition metal ions reacting with aqueous sodium hydroxide:
- Cu2+(aq) + 2OH-(aq) ---> Cu(OH)2(s)
(blue) --------> (pale blue)
- Fe2+(aq) + 2OH-(aq) ---> Fe(OH)2(s)
(green) --------> (green)
- Fe3+(aq) + 3OH-(aq) ---> Fe(OH)3(s)
(yellow) --------> (rust)
These reactions produce solid 'gelatinous' precipitates (called this because they are jelly-like)
Complexes
[edit | edit source]
What is a complex ion?
[edit | edit source]Transition metals have a tendency to form complexes (coordination compounds) this is due to their partially filled d sub-shell accepting donated electron pairs from other ions or molecules.
A complex ion is an ion containing a central atom or ion to which other atoms, ions or molecules are bonded. The atoms, ions or molecules are bonded to it with dative covalent bonds and are called ligands
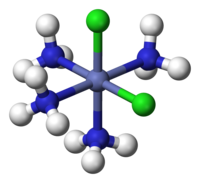
On the right is an example of the complex ion Hexaaquavanadium(III). The central vanadium 3+ metal ion has formed dative bonds from the lone pairs of electrons provided by the water molecule ligands. As water is neutral the overall charge will stay as 3+ but with other ligands that are charged such as Cl- and CN- the overall charge will be affected.
The number of dative covalent (coordinate) bonds that are formed between the central metal and ligands is called the coordination number.
Shapes and Names
[edit | edit source]Naming complexes can seem tricky at first but once you get used to the systematic way of naming the compounds it is easy to stick to the rules:
1. Identify the number of ligands and use the appropriate prefix - 1 mono, 2 di, 3 tri, 4 tetra, 5 penta, 6 hexa
2. Identify the name of the ligands:
- If the ligand is an anion the name will end with an 'o' e.g. cloro (Cl-), cyano (CN-), hydroxo (OH-)
- Most neutral ligands use their usual names but water is aqua, ammonia is ammine and carbon monoxide is carbonyl.
3. Identify the metal in the centre and consider the overall charge of the complex:
- If neutral or positive use the normal name of the metal e.g. copper
- If negative the name will end in 'ate' e.g. ferrate (iron), cuprate (copper), plumbate (lead)
4. Identify the oxidation state of the central metal ion and show in roman numerals in brackets at the end.

On the left we have an example of a complex with an overall negative charge (2-) so cuprate is used instead of copper.
On the right there is an example of a complex with two different types of ligands, this does not affect the naming procedure other than having to do steps 1 and 2 twice.
Here are some more examples of complexes:
[Co(H2O)6]2+ hexaaquacobalt(ll) ion
[CuCl4]2- tetrachlorocuprate(ll) ion
[FeCN6]4- hexacyanoferrate(ll) ion
[CrCl2(NH3)4]+ dichlorotetraamminechromium(lll) ion
Shapes - The shape of Hexaaquavanadium(III) as seen above is octahedral and the shape of Tetrachlorocuprate(II) is tetrahedral
Ligand substitution reactions
[edit | edit source]When a more stable complex is possible, ligands can be substituted by other ligands. For the exam 3 ligand substitutions must be known:
[Cu(H2O)6]2+(aq) + 4Cl-(aq) ---> [CuCl4]2-(aq) + 6H2O(l)
In this reaction blue hexaaquacopper(ll) undergoes ligand substitution when concentrated hydrochloric acid is added drop by drop and yellow tetrachlorocuprate(ll) is formed.
[CuCl4]2-(aq) + 4NH3(aq) + 2H2O(l) ---> [Cu(NH3)4(H2O)2]2+(aq) + 4Cl-(aq)
The yellow tetrachlorocuprate(ll) will then react with conc ammonia and water to form deep-blue tetraamminediaquacopper(ll)
Therefore out of the three complex ions that we have mentioned tetraamminediaquacopper(ll) is the most stable and hexaaquacopper(ll) is the least stable
[Fe(H2O)6]3+(aq) + SCN-(aq) ---> [Fe(H2O)5SCN]2+(aq) + H2O(l)
Here yellow hexaaquairon(lll) becomes blood-red pentaaquathiocyanoiron(lll)
Colorimetry
[edit | edit source]We can use the colour changes that we have just learned about in the ligand substitution reactions to work out the ratio of ligand to metal in a complex. This can be done by adding different volumes of the new ligand to the original complex. A narrow beam of light can then be shone through a filter which allows only one wavelength of light to be absorbed by the solution (otherwise white light would be absorbed by all colours and the colour change would make no difference). The remaining light is then detected by a photocell the other side of the solution and a meter records the results.
The colour of the filter should be that of the colour that the new complex will absorb (opposite to the colour reflected). This means that as the reaction proceeds, more and more light will be absorbed.
Providing enough samples are used, a pattern should appear where there is a limit to the absorbance after a certain volume is reached. This is because all of the ligands have been substituted. Using n=cv the number of moles of both the new ligand used and the original complex for the first of the maximum absorbance values can be calculated. The ratio between these two values is the ration of ligand to metal in a complex.
Redox behaviour
[edit | edit source]Redox reactions
[edit | edit source]A redox reaction is one where reduction and oxidation both occur simultaneously. Transition metals have a variety of different oxidation states so can be reduced or oxidised easily. Constructing redox equations isn't very difficult once you have two half-equations:
e.g. Redox reaction between iron(ll) ions and manganate(Vll) ions in acidified aqueous solution
The half-equation for the oxidation of Fe(ll)
Fe2+(aq) ---> Fe3+(aq) + e-
The half-equation for the reduction of potassium manganate(Vll) in acidified solution
MnO4-(aq) + 8H+(aq) + 5e- ---> Mn2+(aq) + 4H2O(l)
To construct the full equation the number of electrons must be equal so the Fe equation must be multiplied by 5:
5Fe2+(aq) ---> 5Fe3+(aq) + 5e-
The full equation can now be constructed by adding both sides from the two equations:
5Fe2+(aq) + MnO4-(aq) + 8H+(aq) + 5e- ---> 5Fe3+(aq) + 5e- + Mn2+(aq) + 4H2O(l)
And without the electrons:
5Fe2+(aq) + MnO4-(aq) + 8H+(aq) ---> 5Fe3+(aq) + Mn2+(aq) + 4H2O(l)
Observations: Colour change from purple to very pale pink (almost colourless) and the Fe3+ ions cannot be seen as they are too faint.
Redox titrations
[edit | edit source]Using the equation learnt in the previous section we can work out (for example) the percentage by mass of iron in iron tablets bought from a chemist:
In a titration reaction 2.13g of iron tablets were dissolved in sulphuric acid, filtered and washed with water into a standard flask making 250cm3. Then 25cm3 was titrated with 0.01M potassium manganate(Vll). The average titre value after 3 consistent titrations was 12.0cm3.
Remembering that the equation below will confirm the reactions completion at the first permanent pink tinge.
5Fe2+(aq) + MnO4-(aq) + 8H+(aq) ---> 5Fe3+(aq) + Mn2+(aq) + 4H2O(l)
First the number of moles of MnO4- must be calculated using n=cv:
0.01 x 12 = 1.2x10-4 moles
Then the number of Fe2+ can be calculated by looking at the molar ratios in the equation:
1.2x10-4 x 5 = 6 x 10-4
This is the number of moles for the 25cm3 so to find the total for all the tablets (in the 250cm3) the value must be multiplied by 10 to give 6 x 10-3 moles
Finally the number of moles in the solution is multiplied by the mass of Iron 56 and divided by 5 to give the value of one tablet only:
6 x 10-3 x 56 x 1/5 = 0.00672
Therefore: 0.0672/0.425 x 100 = 15.77%
Unfortunately this value is not the same as on the box and this is due to inaccuracies in the execution of the procedure
Questions
[edit | edit source]1. Why do the transition metals Cu and Cr not follow the usual electronic structure pattern?
2. What is a transitional metal? which period 4 d-block elements aren't classified as transitional metals? why?
3. Why are transitional metal ions coloured?
4. What colour are aqueous Fe2+ ions? Which colour and wavelength do they therefore absorb most of?
5. Fe3+(aq) + 3OH-(aq) ---> Fe(OH)3(s) what do you see?
6. name this : [CrCl2(NH3)4]+
7. Write down two ligand substitution reactions involving Cu and describe how colorimetry could be used to determine the ligand:metal ratio
8. 1.7g of steel was dissolved in dilute sulphuric acid and the solution was made up to 250cm3 in a standard flask with distilled water. 25cm3 samples were titrated with 0.02M potassium manganate(Vll) solution. 26.05cm3 of potassium manganate(Vll) solution was required for complete reaction. Calculate the percentage of iron in the steel sample. (Ar: Fe = 56)