Medical Physiology/Basic Biochemistry/Sugars
Overview
[edit | edit source]The most important source of energy for cells is the monosaccharide sugar glucose, which has the general formula C6H12O6. This is broken down in the presence of oxygen to produce energy, carbon dioxide, and water:
(36-38 mols of STP from ADP)
Glucose is taken into the body as various sugars and complex carbohydrates (such as starches). Enzymes in the digestive process (and various other cells) then produce glucose. In the body, glucose can be built up into a starch called glycogen, which can be stored in the liver and in muscles. This is the body's "reserve supply" of glucose. Energy can be also obtained from proteins and fats, but glucose is particularly important, because it is the only source of energy for brain cells. In fact, if the brain is without glucose for a period of 6-8 minutes, brain death will occur. Here is the structure of Glucose. It is usually represented as the first image, or simply a hexagonal ring.
Glucose is broken down in a two step process. First, it is broken down into two molecules of pyruvate by a process called glycolosis. This occurs in the cytoplasm of the cell. Then, if oxygen is present, the pyruvate is taken into the mitochondria, and is broken down into Acetyl-CoA (Acetyl coenzyme A), which enters the citric acid cycle, producing high energy hydrogen bonds. The mitochondria process these bonds into high energy ATP (Adenosine tri-phosphate) from ADP (Adenosine di-phosphate). This ATP powers the reactions of the cell. Some 36-38 molecules of ATP are produced for each glucose molecule so processed. This process is shown graphically in the following flow chart.
In the absence of oxygen (such as may occur during severe exercise), the pyruvate is processed into lactic acid (anaerobic tissue respiration), and only two molecules of ATP are produced. In mammals, this is insufficient for the body's needs, and, in the absence of oxygen, cell death occurs in most tissues.
Sugar Chemistry and Structure
[edit | edit source]Sugars are taken into the body mainly in the form of sugars, monosaccharides & disaccharides, and complex carbohydrates, mainly plant starches. Glucose is the main source of energy in the body, and is the most important of the sugars as its breakdown provides most of the body's energy in the form of the production of ATP from ADP.
Glucose
[edit | edit source]Glucose is a monosaccharide with the general formula C6H12O6, and when it is oxidized by the process of glycolosis and the Citric Acid Cycle, it produces 36-38 molecules of ATP from ADP.
Here is the structure of Glucose:
The first form is usually used in illustrations
This process is described in the section on Glycolosis and Energy Production below.
Other Monosaccharides & Disaccharides
[edit | edit source]Besides glucose, other important monosaccharides include fructose, the sugar found in fruits, and galactose, the sugar of milk. Note that the only difference between glucose and galactose is the arrangement of the hydroxyl groups. These are often ingested in the form of disaccharides.
Sugar polymers
[edit | edit source]Sugars are built into polymers - polysaccharides - in both plants and animals. Animals produce glycogen, plants produce carbohydrates, and insects produce chitin. Much of the sugar that is taken into the body is in the form of carbohydrates. Plants also create a structural compound called cellulose from sugars, which is unfortunately undigestible by humans, even though it is the largest source of carbohydrates!
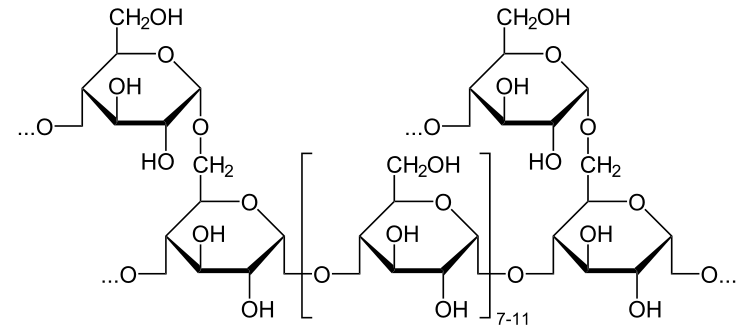
The important polysaccharide in animal physiology is glycogen, a diagram of its structure is shown here:
A detail of the bonds is also shown:
We will look at Glycogen and glycogenesis pathways in more detail elsewhere.
Galactose Metabolism
[edit | edit source]Galactose is the sugar in milk. Infants have an enzyme in the small intestine that metabolizes Lactose to galactose and glucose. In areas where milk products are regularly consumed, adults have also evolved this enzyme. Galactose is converted in the liver to Glucose-6-phosphate, and can thus enter the Glycolysis pathway.
galacto- uridyl phosphogluco-
kinase transferase mutase
gal --------> gal 1 P ------------------> glc 1 P -----------> glc 6 P
^ \
/ v
UDP-glc UDP-gal
^ /
\___________/
epimerase
Fructose Metabolism
[edit | edit source]Fructose is absorbed from the small intestine, and passes to the liver where it is metabolised. Most fructose is converted to Glycogen.
The following description and the above flow sheet is taken from the 2009 wiki:
"Although the metabolism of fructose and glucose share many of the same intermediate structures," they have very different metabolic fates in human metabolism. Fructose is metabolized almost completely in the liver in humans, and is directed toward replenishment of liver glycogen and triglyceride synthesis, while much of dietary glucose passes through the liver where it is metabolized in skeletal muscle to CO2, H2O and ATP, and to fat cells where it is metabolized primarily to glycerol phosphate for triglyceride synthesis as well as energy production. The products of fructose metabolism are liver glycogen and de novo lipogenesis of fatty acids and eventual synthesis of endogenous triglyceride can be divided into two main phases: The first phase is the synthesis of the trioses, dihydroxyacetone(DHAP) and glyceraldehyde; the second phase is the subsequent metabolism of these trioses either in the gluconeogenic pathway for glycogen replenishment and/or the complete metabolism in the fructolytic pathway to pyruvate, which enters the Krebs cycle, is converted to citrate and subsequently directed toward ’’de novo’’ synthesis of the free fatty acid palmitate. " (From http://en.wikipedia.org/wiki/Fructolysis)
Glycolysis & Energy Production
[edit | edit source]Energy is for the most part utilized by cells as ATP which is broken down into ADP with the release of energy. The main source of energy in the cell is glucose, although both fats and amino acids can produce energy by entering into the glycolysis pathway, as is illustrated below. The end product of the glycolysis pathway is pyruvate. In the presence of oxygen, pyruvate is taken into the mitochondria, and processed in the citric acid cycle (CAC), producing some ATP but also a lot of high-energy hydrogen bonds in the form of FADH and NADH. These bonds are then taken into the mitochondrial membrane space and are processed into ATP via the electron transport system. A feature of this process is the way that the various metabolic pathways occur in different parts of the cell. Glycolysis occurs in the cell cytoplasm; the citric acid cycle occurs in the inner mitochondrial space; and the High Energy Transport system occurs in the intermembraneous space of the mitochondria. A summary of the process is shown in the following flow diagram:
Between 36 and 38 molecules of high energy ATP are produced in the breakdown of a single glucose molecule in the process. A more detailed flow diagram is given in the flow charts below.
Glycolysis and the Citric Acid Cycles
[edit | edit source]In the flow sheets each black dot represents a Carbon atom. We will see how glycerol, fats and amino acids enter the system when we look at these molecules.
Glucose is first broken down into Pyruvate in the Glycolosis pathway:

The pyruvate is taken into the Mitochondria, where it is processed in the Citric Acid Cycle:

Localisation of Glycolysis and the Citric Acid Cycles
[edit | edit source]The organelles of the cell localise the place where reactions take place. Glycolysis occurs in the cytoplasm of the cell. In the presence of oxygen pyruvate is transfered into the interior of the mitochondria where it is processed in the Citric Acid Cycle to produce some ATP, and many High Energy Hydrogen bonds. These hydrogen bonds are transported into the inter-membranous space of the mitochondria where the Electron Transport System produces ATP by utilizing the enzyme ATPase.
The following flow sheet illustrates how The high energy hydrogen ions move through the High energy transport system, finally using ATPase to produce ATP from ADP
The 'spent' (now low-energy) hydrogen ions combine with oxygen to produce water.
Anerobic Respiration
[edit | edit source]In the absence of oxygen, such as can occur in heavily exercising muscle, pyruvate does not enter the mitochondria, but the pyruvate is converted to lactic acid.
Pyruvate ---> Lactic Acid
The net gain of ATP by this process is 2 molecules - as opposed to 38 in aerobic respiration. This is not enough to sustain cell activity in most cells. In the absence of oxygen, cell death will rapidly occur. As few as 4 minutes of anoxia will cause irreversible damage to the brain cells.
Lipolysis
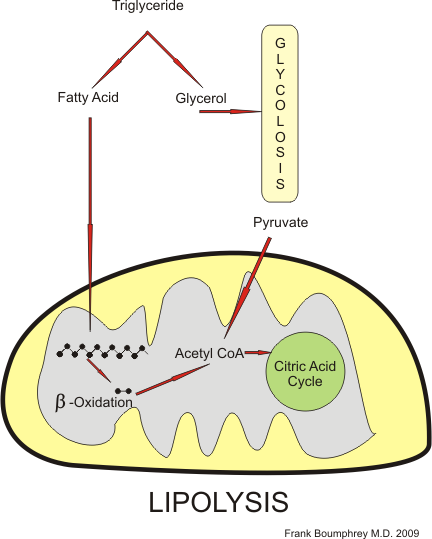
[edit | edit source]Fats are broken down by a complex process to produce energy. The following flow chart shows the pathways involved.
Fats are first broken down into glycerol and fatty acids. The glycerol molecule enters directly into the glycolysis pathway, and the fatty acid is taken into the mitochondria. There, by a complex process called beta-oxidation, it is broken down into two carbon atom units and is then converted to Acetyl coA
Protein Catabolism
[edit | edit source]Proteins are broken down by hydrolysis of the terminal amino acid. This amino acid is then deaminated, and the resulting organic acid either enters the glycolysis pathway or the Citric Acid Cycle.
Glycogen & Gluconeogenesis
[edit | edit source]Glycogen is a polysaccharide of glucose and is used to produce glucose. Although it is stored in all cells, most of it is stored in the liver and muscle cells. It is broken down to produce about 10% Glucose and 90% Glucose-6-phosphate. It is important to note that because only liver cells have an enzyme for converting G-6-P into glucose, only the liver glycogen can supply glucose to the circulating glucose pool.
Structure
[edit | edit source]Glycogen is a branching chain of glucose molecules.
Glycogen Catabolism
[edit | edit source]A flow chart of glycogen catabolism is shown here:
The reverse reaction, the synthesis of glycogen from glucose, is quite complex, and the details need not bother us. However it is useful to consider the reaction from glucose to glycogen as reversible:
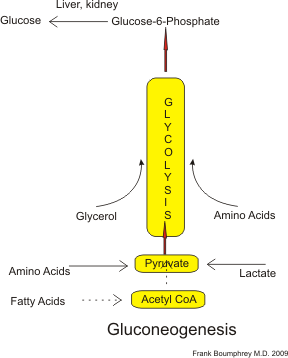
Gluconeogenesis
[edit | edit source]Gluconeogenesis is the production of glucose from precursors, and follows the opposite pathways to glycolysis. Note that the conversion of glucose-6-phosphate can only take place in the kidney and liver, so in practice gluconeogenesis is confined to the liver. A flow sheet is shown:
Although theoretically glucose can be made from fatty acids via Acetyl CoA, because cells can metabolise fatty acids directly as a source of ATP, it is doubtful if much or any glucose is made this way.