General Chemistry/Properties of Matter/Basic Properties of Matter
What is Matter?
[edit | edit source]
Matter is defined as anything that occupies space and has mass. Black holes have mass but occupy effectively no space. Black holes have infinite density and 0 volume. Anything that has mass must be 3-dimensional, which is why, however small atoms (the stuff that makes up matter) are, they are 3-dimensional.
Mass is a measure of an object's inertia. It is proportional to weight: the more mass an object has, the more weight it has. However, mass is not the same as weight. Weight is a force created by the action of gravity on a substance while mass is a measure of an object's resistance to change in motion. For example, your weight on the moon would be one-sixth your weight on the earth, as the moons gravitational field is one-sixth that of earth's. Mass used to measured by comparing the substance of interest to a standard kilogram called the International Prototype Kilogram (IPK). The IPK is a metal cylinder for which the height and diameter both equal 39.17 millimeters and is made of an alloy of 90% platinum and 10% iridium. Thus, the standard kilogram is defined and all other masses are a comparison to this kilogram. When atom masses are measured in a mass spectrometer, a different internal standard is used. Your take home lesson with regard to mass is that mass is a relative term judged by a comparison. Mass is now defined by a Watt or Kibble balance by measuring the Planck constant. The goal of measuring mass with the Planck constant is to measure mass electronically. The reason for this is that it is a lot easier to make an electronic measurements with electronics and electricity than to weigh something big or small, so researchers have been working on accurately redefining the kilogram to a quantum standard that implements the Watt/Kibble balance for the Planck constant. This change took effect in 20 May 2019 after a historic vote among the members of the CGPM at Versailles in France to redefine the kilogram, ampere, mole, and kelvin. The redefinition of the kilogram was contingent upon continued improvement of this new method of determining mass. National metrology labs such as NIST collaborated to make the redefinition of the kilogram possible.
Volume is a measure of the amount of space occupied by an object. Volume can be measured directly with equipment designed using graduations marks or indirectly using length measurements depending on the state (gas, liquid, or solid) of the material. A graduated cylinder, for example, is a tube that can hold a liquid which is marked and labeled at regular intervals, usually every 1 or 10 mL. Once a liquid is placed in the cylinder, one can read the graduation marks and record the volume measurement. Since volume changes with temperature, graduated equipment has limits to the precision with which one can read the measurement. Solid objects that have regular shape can have their volume calculated by measuring their dimensions. In the case of a box, its volume equals length times width times height.
It is particularly interesting to note that measuring is different from calculating a specific value. While mass and volume can both be determined directly relative to either a defined standard or line marks on glass, calculating other values from measurements is not considered measuring. For example, once you have measured the mass and volume of a liquid directly, one can then calculate the density of a substance by dividing the mass by the volume. This is considered indirectly determining density. Interestingly enough, one can also measure density directly if an experiment which allows the comparison of density to a standard is set up.
Another quantity of matter directly or indirectly determined is the amount of substance. This can either represent a counted quantity of objects (e.g. three mice or a dozen bagels) or the indirectly determined number of particles of a substance being dealt with such as how many atoms are contained in a sample of a pure substance. The latter quantity is described in terms of moles. One mole used to be specifically defined as the number of particles in 12 grams of the isotope Carbon-12. This number was 6.02214078(18)x 1023 particles. The mole is now defined so the Avogadro constant N_A has the value 6.022 140 76 times 10 to the 23 reciprocal mole.
- Mass: the kilogram (kg). Also, the gram (g) and milligram (mg).
- 1 kg = 1000 g
- 1000 mg = 1 g.
- Volume: the liter (L), milliliter (mL). Also, cubic centimeters (cc) and cubic meters (m3).
- 1 cc = 1 mL
- 1000 mL = 1 L
- 1000 L = 1 m3
- Amount: the mole (mol).
- 1 mol = 6.02214078(18)x 1023 particles
Atoms, Elements, and Compounds
[edit | edit source]The fundamental building block of matter is the atom. Atoms are made of protons, electrons, and neutrons. Protons and neutrons are made of quarks and gluons.
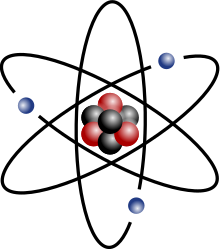
Any atom is composed of a little nucleus surrounded by a "cloud" of electrons. In the nucleus there are protons and neutrons.
However, the term "atom" just refers to a building block of matter; it doesn't specify the identity of the atom. It could be an atom of carbon, or an atom of hydrogen, or any other kind of atom.
This is where the term "element" comes into play. When an atom is defined by the number of protons contained in its nucleus, chemists refer to it as an element. All elements have a very specific identity that makes them unique from other elements. For example, an atom with 6 protons in its nucleus is known as the element carbon. When speaking of the element fluorine, chemists mean an atom that contains 9 protons in its nucleus.
- Atom: A fundamental building block of matter composed of protons, neutrons, and electrons.
- Element: A uniquely identifiable atom recognized by the number of protons in the nucleus.
Despite the fact that we define an element as a unique identifiable atom, when we speak, for example, 5 elements, we don't usually mean those 5 atoms are of the same type (having the same number of protons in their nucleus). We mean 5 'types' of atoms. It is not necessary there are only 5 atoms. There may be 10, or 100, etc. atoms, but those atoms belong to one of 5 types of atoms. I'd rather define 'element' as 'type of atom'. I think it is more precise. If we'd like to refer to 5 atoms having the same 6 protons in their nucleus, I'd say '5 carbon atoms' or '5 atoms of carbon'.
It is important to note that if the number of protons in the nucleus of an atom changes, so does the identity of that element. If we could remove a proton from nitrogen (7 protons), it is no longer nitrogen. We would, in fact, have to identify the atom as carbon (6 protons). Remember, elements are unique and are always defined by the number of protons in the nucleus. The Periodic Table of the Elements shows all known elements organized by the number of protons they have.
An element is composed of the same type of atom; elemental carbon contains any number of atoms, all having 6 protons in their nuclei. In contrast, compounds are composed of different type of atoms. More precisely, a compound is a chemical substance that consists of two or more elements. A carbon compound contains some carbon atoms (with 6 protons each) and some other atoms with different numbers of protons.
Compounds have properties different from the elements that created them. Water, for example, is composed of hydrogen and oxygen. Hydrogen is an explosive gas and oxygen is a gas that fuels fire. Water has completely different properties, being a liquid that is used to extinguish fires.
The smallest representative for a compound (which means it retains characteristics of the compound) is called a molecule. Molecules are composed of atoms that have "bonded" together. As an example, the formula of a water molecule is "H2O": two hydrogen atoms and one oxygen atom.
Properties of Matter
[edit | edit source]Properties of matter can be divided in two ways: extensive/intensive, or physical/chemical.
According to the International Union of Pure and Applied Chemistry (IUPAC), an intensive property or intensive quantity is a quantity whose magnitude is independent of the size of the system (the part of the environment under study). Intensive properties include temperature, refractive index (for example, the refactive index of air is 1.000293), and mass density. The reciprocal or multiplicative inverse of mass density, specific volume, is also an intensive property. Boiling point is an intensive property. Here is a list of intensive properties:
- charge density
- linear charge density is amount of electric charge per unit length and typically represented by with units of
- surface charge density is amount of electric charge per unit surface area and represented by and measured with the unit
- volume charge density is amount of electric charge per unit volume and typically represented by rho, and measured with the unit .
- color
- concentration
- mass concentration in
- molar concentration in
- number concentration in
- energy density in
- electric permeability as a measure of magnetization produced in a material in response to an applied magnetic field typically represented by mu, and measured with either or
- specific gravity
- melting point
- boiling point
- molality measured with
- pressure
- refractive index
- electrical resistivity measured with
- electrical conductivity measured with
|
|
States of Matter
[edit | edit source]One important physical property is the state of matter. Three are common in everyday life: solid, liquid, and gas. The fourth, plasma, is observed in special conditions such as the ones found in the sun and fluorescent lamps. Substances can exist in any of the states. Water is a compound that can be liquid, solid (ice), or gas (steam).


Solids
[edit | edit source]Solids have a definite shape and a definite volume. Most everyday objects are solids: rocks, chairs, ice, and anything with a specific shape and size. The molecules in a solid are close together and connected by intermolecular bonds. Solids can be amorphous, meaning that they have no particular structure, or they can be arranged into crystalline structures or networks. For instance, soot, graphite, and diamond are all made of elemental carbon, and they are all solids. What makes them so different? Soot is amorphous, so the atoms are randomly stuck together. Graphite forms parallel layers that can slip past each other. Diamond, however, forms a crystal structure that makes it very strong.
Liquids
[edit | edit source]Liquids have a definite volume, but they do not have a definite shape. Instead, they take the shape of their container to the extent they are indeed "contained" by something such as beaker or a cupped hand or even a puddle. If not "contained" by a formal or informal vessel, the shape is determined by other internal (e.g. intermolecular) and external (e.g. gravity, wind, inertial) forces. The molecules are close, but not as close as a solid. The intermolecular bonds are weak, so the molecules are free to slip past each other, flowing smoothly. A property of liquids is viscosity, the measure of "thickness" when flowing. Water is not nearly as viscous as molasses, for example.
Gases
[edit | edit source]Gases have no definite volume and no definite shape. They expand to fill the size and shape of their container. The oxygen that we breathe and steam from a pot are both examples of gases. The molecules are very far apart in a gas, and there are minimal intermolecular forces. Each atom is free to move in any direction. Gases undergo effusion and diffusion. Effusion occurs when a gas seeps through a small hole, and diffusion occurs when a gas spreads out across a room. If someone leaves a bottle of ammonia on a desk, and there is a hole in it, eventually the entire room will reek of ammonia gas. That is due to the diffusion and effusion. These properties of gas occur because the molecules are not bonded to each other. The molecules in gas, are free and they can move around unlike solid molecules.
Technically, a gas is called a vapor if it does not occur at standard temperature and pressure (STP). STP is 0° C and 1.00 atm of pressure. This is why we refer to water vapor rather than water gas. |
- In gases, intermolecular forces are very weak, hence molecules move randomly colliding with themselves, and with the wall of their container, thus exerting pressure on their container. When heat is given out by gases, the internal molecular energy decreases; eventually, the point is reached when the gas liquifies.















