General Chemistry/Phase Changes
Phase Diagrams
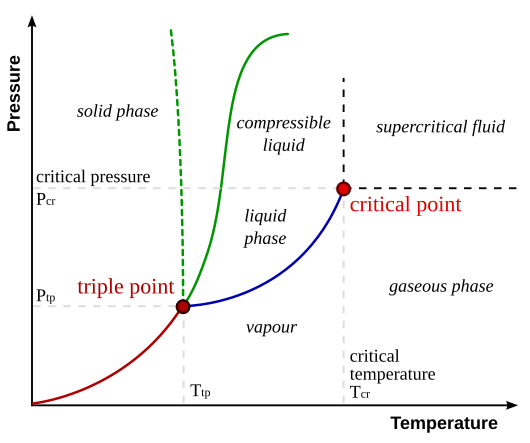
[edit | edit source]Phase diagrams predict the phase of a substance at a certain pressure and temperature.
The triple point is where all three phases of matter can exist in equilibrium. Points that are on a line are where two phases of matter can coexist.
The critical point is the highest pressure and temperature that the three normal phases can exist. Any further, and the characteristics become a blend of liquid and gas.
Changes in State
[edit | edit source]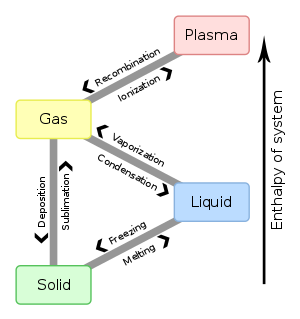
If the temperature and pressure change and move across a line in the diagram, the phase will also to change. There are six ways this can happen:
- Solid to liquid: melting
- Liquid to solid: freezing
- Liquid to gas: evaporation
- Gas to liquid: condensation
- Gas to solid: deposition
- Solid to gas: sublimation
The diagram on the right also shows the plasma state of matter. A plasma is simply a gas that has been completely ionized, so that there is a mixture of positive ions and electrons. It has interesting electrical properties, but it is not important in the scope of General Chemistry.
Energy Changes
[edit | edit source]| Helpful Hint! | |
|---|---|
| The kinetic energy of a molecule is directly proportional to its temperature. Potential energy is used to break bonds. |
If you boil water, it never goes above 100 degrees Celsius. Only after it has completely evaporated will it get any hotter. This is because once water reaches the boiling point, extra energy is used to change the state of matter and increase the potential energy instead of the kinetic energy. The opposite happens when water freezes. To boil or melt one mole of a substance, a certain amount of energy is required. These amounts of energy are the molar heat of vaporization and molar heat of fusion. If that amount of energy is added to a mole of that substance at boiling or freezing point, all of it will melt or boil, but the temperature won't change.
The graph shows the temperature of ice as heat is added. Temperature increases linearly with heat, until the melting point. Then, the heat added does not change the temperature. That heat energy is instead used to break intermolecular bonds and convert ice into water. At this point, there is a mixture of both ice and water. Once all ice has been melted, the temperature again rises linearly with heat added. At the boiling point, temperature no longer rises with heat added because the energy is once again being used to break intermolecular bonds. Once all water has been boiled to steam, the temperature will continue to rise linearly as heat is added.