Ecology/Biogeochemical cycles
Chapter 14 | Chapter 15 >>
Chapter 15. Biogeochemical Cycles

In the previous chapter on environmental response, we considered the various ways that individual organisms respond to the physical, chemical and/or biological stimuli in their environment.In this chapter, we will go deeper into the interactions between the biotic and the abiotic elements of the biosphere, focusing on the nonliving components. To begin to understand this concept: the physical "home" of our biosphere, we first have to consider the physics of planet formation. From all we know about life in the universe, only planets can possibly offer conditions suitable for life. And indeed, since all life we know of occurs on just one planet, we need to develop an understanding of what is special or unique about our home planet. How is the earth constituted; what is the nature of this "stage" upon which life developed and upon which life continues to play out. Presently, the "ecology" of other planets, if such a thing exists, is a subject of study within the science of exobiology.
Nutrients move through the ecosystem in biogeochemical cycles. A biogeochemical cycle is a circuit/pathway by which a chemical element moves through the biotic and the abiotic factors of an ecosystem. It is inclusive of the biotic factors, or living organisms, rocks, air, water, and chemicals. The elements that are moving through the biotic or abiotic factors may be recycled, or they may be accumulated in a place called a sink/reservoir where they are held for a long period of time. The amount of time that a chemical is held in one place is called residence.
All the chemical elements in an organism are part of the biogeochemical cycle. The chemicals travel not only through the biotic and abiotic components of an ecosystem, but they also travel through an organism. The abiotic factors of an ecosystem include: (1) water (hydrosphere), (2) land (lithosphere), and (3) air (atmosphere). All of the living factors that are found on Earth make up the biosphere.
Nutrient Cycle Levels[edit | edit source]
Nutrient cycles are broken up into three levels. There are global nutrient cycles, which occur when ecosystems become linked on a global scale. This is now known as an Ecosphere. On a smaller scale, there are local nutrient cycles. These cycles consist of the cycling that occurs in just one ecosystem. The smallest level includes common nutrient budgets and fluxes; some examples of this are carbon, H2O, nitrogen, phosphorus, iron, and other trace elements. All of these cycles can be linked so that a community can remain at equilibrium. All of these cycles are driven by microbes. Microbes contain 350-550 giga tons of the worlds carbon. They also contain 85-130 giga tons of nitrogen, along with 9-14 giga tons of phosphorus. Nitrogen is only fixed by bacteria and archea, and they fix 85% of the 15 giga tons of nitrogen every year. Cyanobacteria are responsible for fixing 50% of the O2 in the Ecosphere. There are also huge reserves of carbon locked up in rocks. Rocks contain 81 million gig tons of carbon, which is in the form of calcium carbonate (CaCo3), commonly called limestone.
The processes that make up these cycles can be divided into two groups. The first group of processes all have an aerobic component. Through respiration, organic material can be converted into carbon dioxide. Respiration is done by animals, plants, and microbes. A second aerobic process is carbon fixation. This is the reverse of respiration where carbon dioxide is converted into organic material. The second group of processes are anaerobic processes. The first anaerobic process is fermentation. This is another way that organic material can be converted into carbon dioxide. Bacteria and fungi carry out this process when deprived of oxygen. A second form of an anaerobic process is carbon fixation. The only organisms that fix carbon anaerobically are archea. The last anaerobic process is methanogenesis. This process takes carbon dioxide and converts it into methane. This process is also only done by archea, but does not account for all methane because some methane can be made aerobically.

There are two main types of systems on Earth, a closed system and an open system. A closed system occurs when the chemicals or elements used in an ecosystem are recycled instead of being lost. An open system occurs when the sun provides Earth with energy in the form of light which is usually used and then lost in the form of heat as it travels through the various trophic levels on Earth.
Although the biogeochemical cycles are complex and differ between the needs of nutrients by heterotrophs and autotrophs, there are three shared components of nutrient cycles: inputs, internal cycling, and outputs. Precipitation is one form of input, which brings appreciable quantities of nutrients into a cycle (Patterson, 1975). Microbes recycling dead organic material back into the system to be reused is a form of internal cycling. The export of nutrients must be offset by the input of nutrients into a system if there is to be no net loss. Carbon is exported from a cycle in the form of CO2 via respiration of all living organisms.
The most important biogeochemical cycles are the carbon cycle, nitrogen cycle, oxygen cycle, phosphorus cycle, and the water cycle. The biogeochemical cycles always have a state of equilibrium. The state of equilibrium occurs when there is a balance in the cycling of the elements between compartments. Ecologists may also be interested in the sulfur cycle, the nutrient cycle, and the hydrogen cycle; however, ecologists are more interested in studying the carbon, nitrogen, oxygen, phosphorus, and water cycles.
The term biogeochemical gets its name from the previous cycles listed above (abiotiv factors). The prefix Bio refers to the biosphere. Geo refers to the lithosphere, atmosphere, and hydrosphere. Chemical refers to the various chemicals that go through/travel through the biogeochemical cycles.
Biogeochemical Cycles[edit | edit source]

A biogeochemical cycle or inorganic-organic cycle is a circulating or repeatable pathway by which either a chemical element or a molecule moves through both biotic ("bio-") and abiotic ("geo-") compartments of an ecosystem. In effect, an element is chemically recycled, although in some cycles there may be places (called "sinks") where the element accumulates and is held for a long period of time. In considering a specific biogeochemical cycle, we focus on a particular element and how that element participates in chemical reactions, moving between various molecular configurations. Of the 90-odd elements known to occur in nature, some 30 or 40 are thought to be required by living organisms (Odum, 1959). We will be considering only a few of these, mainly those utilized in fairly large quantities by living organisms. The principal elements of life are carbon, hydrogen, oxygen, and nitrogen. However, a number of others are certainly important to understand as well, notably phosphorus and sulfur. Some "non-essential" elements participate in biogeochemical cycles, entering organism tissues because of chemical similarity to essential elements. For example, strontium can behave like calcium in the body.
Hydrologic Cycle (Water Cycle)[edit | edit source]
A very significant molecule (on planet Earth) that cycles through ecosystems is the water molecule (H2O). One of the reasons that life is so dependent on water is that it is the medium of chemical reactions within cells. While it generally is the case that we discuss the water cycle in terms of the various states of water, at least some water molecules are taken up by plants and split apart (photolysed) into atoms of hydrogen and oxygen; the latter is released into the atmosphere as molecular oxygen (O2). Thus, by virtue of photosynthesizing organisms (photoautotrophs), the water cycle is an important part of both the oxygen and the hydrogen cycles. Note that hydrogen ends up as part of an organic molecule, and therefore a participant in the carbon cycle.
The majority of water in the water cycle is found within the oceans and the polar ice caps, although water is present in the bodies of organisms, in freshwater lakes and rivers, frozen in glaciers, and in the ground as groundwater. Water moves more or less freely between these storage reservoirs: by evaporation, by precipitation, and by runoff from the land.
The sedimentation cycle is an extension of the hydrological cycle. The water carries materials from the land to the ocean, where they are added as sediments. The sediment cycle includes the physical and chemical erosion, nutrient transport, and sediment formation from water flows. The sediment formed from water flows is mostly responsible for the buildup of sediments at the bottom of the ocean. The sediment cycle is tied in with the flow of six important elements, which are hydrogen, carbon, oxygen, nitrogen, phosphorus and sulfur. These elements, also known as macroelements, make up 95% of all living things. The balancing of these molecules is required to sustain life. These elements have to be recycled for life to continuously regenerate.
Carbon Cycle[edit | edit source]

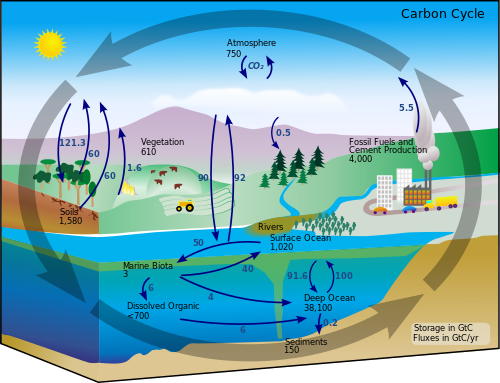
Carbon is required for the building of all organic compounds. Carbon in the form of carbon dioxide (CO2) is obtained from the atmosphere and transformed into a usable organic form by organisms. The reservoirs for the carbon cycle are the atmosphere, where carbon dioxide exists as a free gas, fossil organic deposits (such as oil and coal), and durable organic materials like cellulose. Mineral carbonates, such as limestone, are a significant geological sink for carbon. During the process of carbon fixation, carbon dioxide is taken up from the atmospheric reservoir (or from biocarbonates dissolved in water) by plants, photosynthetic bacteria, and algae and is "fixed" into organic substances. Animals obtain their requirements for carbon (as carbon-based molecules) by eating plants or other animals. For the biological links, the carbon cycle comes full cycle when carbon is released by either plants and animals as they respire or after life as they decompose. Organisms respire carbon dioxide as a waste product from the breakdown of organic molecules as their cells derive energy from oxidizing the molecules containing "fixed" carbon. The burning of organic material such as wood or fuels also results in the release of carbon dioxide from organic carbon.
CO2 is a trace gas and has huge effects on Earth’s heat balance by absorbing infrared radiation. During the growing season or summer, there is a decrease in atmospheric CO2 because increased sunlight and temperature helps plants increase their carbon dioxide uptake and growth. In the winter time, more CO2 enters the atmosphere than can be removed by plants. This happens because plant respirations and the death of plants happens faster than photosynthesis. [Life and Biogeochemical Cycles ]
Carbon Cycling Experiments[edit | edit source]
The impact of land-cover and land-use change on global carbon cycle has been the focus of various studies. The Brazilian Legal Amazon has been an area of particular interest because of rapid forest clearing. In determining such an impact, models are often developed and used. In a study focusing on the net carbon flux caused by deforestation and forest re-growth over the period of 1970-1998, a processed-based model of forest growth, carbon cycling, and land-cover dynamics was developed. This model was named CARLUC (for CARbon and Land-Use Change). This model, in particular, was used to estimate the size of terrestrial carbon pools in terra firme (unflooded) across the Brazilian Legal Amazon and the net flux of carbon resulting from forest disturbance and forest recovery from disturbance [1].
As previously mentioned, carbon cycling is driven by both abiotic and biotic factors. Net carbon fluxes can be partitioned into assimilatory and respiratory components. The complex interplay between assimilatory and respiratory sources and their responsiveness to abiotic changes such as drought and temperature are poorly understood. In a recent study, net carbon partitioning was used to disentangle abiotic and biotic drivers of all component influencing the overall sink strength of a Mediterranean ecosystem during a rapid spring to summer transition (between May and June 2006). It was ultimately determined that decreasing soil water availability rather than increasing air temperature largely affected both assimilation and respiration fluxes of understory plants and in consequence ecosystem respiration and soil respiration [2].
Global Carbon Cycle[edit | edit source]
Carbon is continuously cycled between the oceans, land, and the atmosphere. The atmospheric carbon is primarily carbon dioxide. Carbon on land occurs primarily in living biota and decaying organic matter. Dissolved carbon dioxide and small organisms like plankton that store carbon dioxide are major sources in the ocean. Carbon is measured in Gigatons, with the deep ocean containing almost 40,000 Gt, compared to about 2,000 Gt on land and 750 Gt in the atmosphere.
Carbon dioxide, a known "greenhouse gas," traps some of the radiation in the atmosphere that would be lost into space. This causes the atmosphere to be warmer than it would naturally be. Since pre-industrial times, man-made emissions have increased the amount of carbon dioxide in the atmosphere by about 30%. The increase in carbon dioxide causing global warming is important for us to understand so we can predict future implications concerning our planet.
Average global temperatures have increased this past century and are predicted to increase even more in the next century. In a study done by Parmesan et al, the distribution and population dynamics of several species of butterflies were monitored in response to the increased temperatures. Migratory patterns of each species were observed and compared to previous patterns of distribution. The butterflies showed a northern shift, which is thought to be a response to increased temperatures. Parmesan et al concluded that the current and future climate warming may be a big factor in shifting species distributions [3].
Nitrogen Cycle[edit | edit source]
Nitrogen is required for the manufacturing of all amino acids and nucleic acids; however, the average organism cannot use atmospheric nitrogen for these tasks and, as a result, is dependent on the nitrogen cycle to source usable nitrogen. The nitrogen cycle "begins" with nitrogen stored in the atmosphere as N2 (the largest nitrogen reservoir). Nitrogen is also stored in the soil as ammonium (NH4+), ammonia (NH3), nitrite (NO2−), and nitrate (NO3−). Nitrogen is assimilated into living organisms through three stages: nitrogen fixation, nitrification, and plant metabolism. Nitrogen fixation is a process that occurs in prokaryotes in which N2 is converted to NH4+. Atmospheric nitrogen can also undergo nitrogen fixation by lighting and UV radiation and become NO3-. Following nitrogen fixation, nitrification occurs. During nitrification, ammonia is converted into nitrite, and nitrite is converted into nitrate. Nitrification occurs in various bacteria. In the final stage, plants absorb ammonia and nitrate and incorporate it into their metabolic pathways. Once the nitrogen has entered the plant metabolic pathway, it may be transferred to animals when the plant is eaten. Nitrogen is released back into the cycle when denitrifying bacteria convert NO3- into N2 in the process of denitrification, when detrivorous bacteria convert organic compounds back into ammonia in the process of ammonification, or when animals excrete ammonia, urea, or uric acid.
A lot of environmental problems are caused by the disruption of the nitrogen cycle by human activity. Some of the problems caused range from the production of tropospheric (lower atmospheric) smog to the perturbation of stratospheric ozone and contamination of ground water. An example of one of the problems caused is the formation of greenhouse gas. Like carbon dioxide and water vapor, nitrous oxide traps heat near the earth’s surface, and it also destroys the stratospheric ozone. Nitrous oxide in the earth’s atmosphere is broken down by UV light into nitrogen dioxide and nitric oxide. These two products can reduce the ozone. Nitrogen oxides can be changed back into nitrates and nitrite compounds and recycled back into the earth’s surface.[Environmental Biology - Ecosystems ]
Phosphorus, Iron, and Trace Mineral cycles[edit | edit source]
The phosphorus cycle is one of the slowest biogeochemical cycles. Phosphorus mainly cycles through water, soil, and sediments. The little phosphorus in the atmosphere is carried by dust particles and such. Phosphate is most often in the form of phosphate salts, which are released from weathered rock and dissolved in ground water where plants take them up. Phosphorus is a limiting factor for plants on land and in water, including the ocean, because there is so little of it, and it is not very soluble in water. The cycle does speed up a little bit as it cycles through plants and animals. When the plants and animals die and decay, the phosphorus is returned to the soil and sediments and eventually locked back in the rock.
The iron cycle is similar to every other cycle. Iron is, however, much more abundant than phosphorus. One additional way iron gets cycled, besides rock weathering, is when a volcano erupts and sends an iron-rich dust into the atmosphere, where it eventually spreads to soil and water.
Other trace minerals, such as zinc, copper, and manganese, were once thought to be just as abundant as nitrogen, carbon, and oxygen. Their depletion is thought to be from water erosion of the soil and over cropping of land, so that they aren't able to replenish themselves quickly enough. To learn more about all the trace minerals essential to life visit:Nutrients - Trace Minerals
"Perfect" Cycles[edit | edit source]
Odum (1959) describes what are called more or less "perfect" cycles: biogeochemical cycles that involve equilibrium states. That is, there exists in nature a balance in the cycling of the element between various compartments, with the element or material moving into abiotic compartments about as fast as it moves into biotic compartments. Certain ecosystems may experience "shortages," but overall, a balance exists on a global scale.
II. Deserts are characterized by their limited precipitation. Deserts have a brief rainy season, which on average produces less than 30 cm rainfall per year. Deserts are mainly located at 30 degrees North and South latitude because of their dry air resulting from the Hadley cells (please read interlink below on a Hadley cell), which stretch between the equator and 20 to 35 degrees North and South latitude.
Deserts have the capacity to be very productive climates because they are in a high energy input area of the round earth (please refer to chapter 3 about solar irradiation and the earth being round), but they are limited by their low precipitation. Desert flora or plant life is located on only 10% of desert area. They consist of mainly annuals that grow and reproduce during the brief rainy season. The main flora are succulents that store water, have waxy cuticles to prevent water loss, may have few modified small leaves or needles, and have large, shallow root systems.
III. Chaparrals are desert types located in the Mediterranean area. Chaparrals are characterized by their distinct rainy seasons and more so by their extremes. Summer temperatures can reach a high of approximately 40 degrees Celsius, and winter temperatures can reach a low of approximately -15 degrees Celsius. Plant and animal inhabitants are drought adapted to aide survival. Plant life is similar in characteristics to desert plants, with mainly shrub/brush type species.
Soil[edit | edit source]
Soilis the foundation for terrestrial systems. It is a mixture of organic (living and nonliving) and inorganic material. The organic part is contributed by plants, animals, and microbes. The inorganic part is a result of weathering.
Soil Layering[edit | edit source]
Soil is a composition of many different layers. The first(top) layer is the O Horizon which is then compsed of 2 sublayers the Oi and Oa layers. The Oi is the intact organic layer which is made up of dead organic matter and leaf litter. The Oa is contains mostly humus and is situated next to the A Horizon. The entire O Horizon is about 10cm deep. Deserts usually lack the O Horizon.
The next layer is the A Horizon. This is what most consider the topsoil, it is a mixture of weathered rock(clay, sand, and silt) from lower layers and organic material from the O Horizon. This is the area where there are the most roots, microbes, and invertebrates. This horizon usually has a high respiration rate, and has most of the nutrients leeched to lower layers by water. The top 2 layers combined (O and A Horizons) are on average about 0.5m deep.
The next layer is the E Horizon (or Eluviation Horizon). This layer has the least amount of dissolved nutrients. This layer can have a clay pan formed at the bottom which is highly compacted clay which prevents weathering of rock below. The also prevents tap roots from reaching the C Horizon. This may also cause the area above to become water-logged.
Below the E horizon is the Illuviation Horizon of B Horizon. This is the area where leechates collect, and most tap roots exist. It is usually mineral rich and contains little organic material. It is usually about 0.5m deep. Below the B Horizon is the C Horizon, which is the lowest soil, layer is commonly called the subsoil. This is mostly weathered parent material. This is usually about 0.5m deep also.
Lastly is the inorganic material of the soil or the R Horizon. This is also called bedrock which is a result of weathering from wind, water, temperature, and plants.
Particle Size[edit | edit source]

The particle size of soil is correlated to the amount of surface area of the particles and the charge of the surface area. A decrease in particle size equals an increase in surface area which in turn equals an increase in a negative charge of the surface area. The United States Department of Agriculture (USDA) designates types of soil by the size of the particles in the soil. Clay consists of particles smaller than 0.002mm and has the largest water capacity and highest nutrient capacity. Silt consists of particles smaller than 0.05mm and has a relatively median water capacity and a median nutrient capacity. Sand consists of particles smaller than 2mm and has the smallest water capacity and the lowest nutrient capacity.
Acidic Soil[edit | edit source]
Acidic soil is any type of soil that has a low pH. It is created whenever soil is very low in minerals and may be created by conifer needles. Amino acids also breaks down clay. The breakdown of clay is done by leeching the minerals out of the soil. This leeching of minerals is due to a high Hydronium ion [H+] concentration which results of in less productive soil. Acid rain or precipitation that is unusually acidic because of emissions of sulfur and nitrogen compounds which react with the atmosphere is widely believed to be responsible for acidifying soil.
References[edit | edit source]
- ^ Hirsh, Adam I. et al. 2004. "The Net Carbon Flux Due to Deforestation and Forest Re-growth in the Brazilian Amazon: Analysis using a Processed-Based Model". Global Change Biology. Vol. 10, pgs 908-924.
- ^ Unger, Stephan et al. 2009. "Partitioning Carbon Fluxes in a Mediterranean Oak Forest to Dientangle Changes in Ecosystem Sink Strength During Drought". Agricultural and Forest Meterology. Vol. 149, Iss. 6-7, pgs 949-961.
- ^ Hanski, Ilkka. "Metapopulation Dynamics." Nature 396 (1998): 41-49.
- ^ Odum, E. P. 1959. Fundamentals of ecology. W. B. Saunders Co., Philadelphia and London. 546 p.
- ^ Patterson, D. T. 1975. Nutrient return in stemflow and throughfall of individual trees in the Piedmont deciduous forest. Mineral Cycling in Southeastern Ecosystems. National Technical Information Service, U.S. Deot. of Commerce: 800-812.
- ^ Parmesan, Camille, Nils Ryrholm, Constanti Stefanescus, etal. 1991. Poleward shifts in geographical ranges of butterfly species associated with regional warming. Nature Vol.399.