Biochemistry/Proteins/The chemistry of proteins
Amino acid structure and chemistry
[edit | edit source]General features
[edit | edit source]Amino acids consist of a primary amine bound to an aliphatic carbon atom (the so-called α-carbon), which in turn is bound to a carboxylic acid group. At least one hydrogen atom is bound to the α-carbon; in addition, the α-carbon bears a side chain, which is different for different amino acids. In a neutral aqueous solution, amino acids exist in two forms. A very small fraction of amino acid molecules will be neutral, with a deprotonated amino group and a protonated carboxylic acid group. However, the overwhelming majority of molecules will be in a Zwitterion tautomer, with a positive charge on the (protonated) amino group and a negative charge on the (deprotonated) carboxylate group.
Amino acids are linked via a Peptide bond. (From an organic chemistry perspective, a peptide bond is a type of amide group.) A peptide bond consists of a carbonyl group's carbon atom directly bound to the nitrogen atom of a secondary amine. A peptide chain will have an unbound amino group free at one end (called the N-terminus) and a single free carboxylate group at the other end (called the C-terminus).
The peptide bond is planar, because resonance between the carbonyl group and the amino nitrogen lends the C-N bond a partial double-bond character. (It is possible to draw a resonance structure with a double bond between the carbon atom and the nitrogen atom, with a formal negative charge on the oxygen atom and a formal positive charge on the nitrogen.) This prevents rotation around the C-N bond, locking the peptide bond in the trans conformation, and holding six atoms in a plane: the α-carbon of one amino acid, the carbonyl carbon and oxygen atoms, the amino nitrogen and hydrogen atoms, and the &alpha-carbon of the second amino acid are all co-planar.
The structure of a peptide chain's "backbone" can be described uniquely by the torsion angles between adjacent peptide units. The φ-angle is the torsion angle between the α-carbon and the carbonyl carbon of one amino acid; the ψ-angle is the torsion angle between the amino nitrogen and the α-carbon.
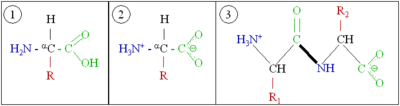
Two amino acids with different side chains react when the amino-terminus (red) of one joins the carboxy-terminus (blue) of another are linked by an amide bond (green). See also the mechanism of peptide bond formation.
It is important to note that the formation of this peptide bond is highly unfavorable under standard conditions. However, in the human body, there are enzymes that assist in facilitating this reaction, making peptide bond formation and proteins possible.
Chemical classification of aminoacids
[edit | edit source]The 20 amino acids encoded by the genetic code are:

They are not shown in their zwitterionic state for clarity. At physiological pH (~pH 6.8), all would be so, except proline, which is a five-membered ring. Charged side-chains are shown ionic when they exist as such at physiological pH.
Protein Electrophoresis
[edit | edit source]Protein Electrophoresis is a method in which a mixture of proteins can be separated and analyzed. Electrophoresis is based on the mobility of ions in an electric field. The charge distribution of the molecules is critical in the separation of all electrophoresis. In an electric field, electrophoresis is a passage of charged molecules in solution. Positively charged ions have tendency toward a negative electrode and inversely, negatively charged ions have tendency toward a positive electrode. The molecular weight results to a molecular friction which is directly proportional to the molecular charge and its voltage and inversely proportional to a molecule's mobility in an electric field.
Gel electrophoresis is performed to analyze the molecular weights and the charge of the protein and is mostly used in electrophoresis of the protein. The gel electrophoresis is carried out in a thin piece of polyacrylamide. The crosslinked of acrylamide and N,N'-methylene-bis-acrylamide forms the polyacrylamide by polymerization. The size has an important effect in the movement of the protein molecule. The smaller molecules of protein would result in a faster passage of the molecules through the gel pores.
The separation of the protein molecules in the gel affects the protein activity. In this process, first the protein reduces the disruption of disulfide bonding by heating which results in purification and denaturalization. Next, the sodium dodecyl sulfate, abbreviated as SDS, (and ionic detergent) is added. SDS is an anionic detergent which dissolve hydrophobic molecules and denatures protein molecules without breaking peptide bonds. This result in the dislocation of the structure of the protein changes the secondary, tertiary and quaternary to the primary structure with negative charge. Then the protein is passed through the gel. For denatured proteins, SDS can form a steady charge mass ratio in binding with proteins.
The polyacrylamide gel electrophoresis is a very sensitive method capable of a bearing a high resolution and it is analytically used in the studies in the separation techniques.
Isoelectric point
[edit | edit source]The isoelectric point (pI) is the pH-value in which a protein is neutral, that is, has zero net charge. To be clear, it is not the pH value where a protein has all bases deprotonated and all acids protonated, but rather the value where positive and negative charges cancel out to zero.
Calculating pI: An aminoacid with n ionizable groups with their respective pKa values pK1, pK2, ... pkn will have the pI equal to the average of the group pkas:
pI=(pK1+pK2+...+pkn)/n
Most proteins have many ionizable sidechains in addition to their amino- and carboxy- terminal groups. The pI is different for each protein and it can be theoretically calculated according Henderson-Hasselbalch equation if we know amino acids composition of protein.
In order to experimentally determine a protein's pI 2-Dimensional Electrophoresis (2-DE) can be used. The proteins of a cell lysate are applied to a pH immobilized gradient strip, upon electrophoresis the proteins migrate to their pI within the strip. The second dimension of 2-DE is the separation of proteins by MW using a SDS-gel. To clearly understand isoelectric point, you have to keep in mind that the positively charged groups are balanced by the negatively charged groups. For simple amino acid " alanine", the isoelectric point is an average of the PKa of the carboxyl with PK1 of (2.34) and ammonium with PK2 of (9.69) groups. So, the PI for the simple amino acid " alanine" is calculated as:(2.34+9.69)/2 which is equal to 6.02. When additional basic or acidic groups are added as a side-chain functions, the isoelectric point pI will be the average of the pKa's of the most similar acids. Example for this concept could be aspartic acid in which the similar acids are alpha-carboxyl function with pKa of 2.1 and the side chain carboxyl function with a pKa of 3.9. Thus, the pI for aspartic acid is (2.1+3.9)/2=3.0. Another example is arginine, its similar acids are guanidinium on the side chain with pKa of 12.5 and the alpha-ammonium function with pKa of 9.0. Thus the calculated pI for arginine= (12.5+9.0)/2=10.75. pI does not has a unite.
The peptidic bond
[edit | edit source]Two amino acid molecules can be covalently joined through a substituted amide linkage, termed a peptide bond, to yield a dipeptide. this link is formed by dehydration (removal of the water molecules - one hydrogen atom from one amino acid and an OH group from the other). This process can then continue to join other amino acids and yield in an amino acid chain. When there are few amino acids in a chain, it is called an oligopeptide, when there are many it is called a polypeptide. although the terms "protein" and "polypeptide" are sometimes used to describe the same thing, the term polypeptide is generally used when the molecular weight of the chain is below 10,000. An amino acid unit in a peptide is often called a residue.
Disulfide bonds
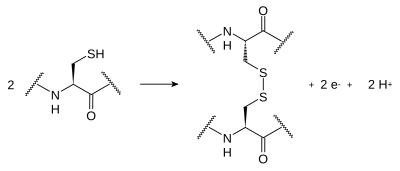
[edit | edit source]Disulfide bonds form between the sulfur atoms of two cysteine side chains in a protein. The side chains undergo a reversible oxidation of the sulfhydryl groups of the cysteine, which results in covalent bonding of the sulfur atoms (S-S). This bonding is called a "disulfide bridge". Typically, disulfides don't form on the surface of proteins because of the presence of reducing agents in the cytoplasm. These bonds are of great importance concerning the shaping of protein structure; their formation guides the folding of peptide chains as the proteins are produced. Structural proteins that have to be rigorously stable (for example, Keratin, which is found in nail, horn and crustacean shell) often contain a large number of disulfide bonds.
Post-translational modifications
[edit | edit source]After a protein is synthesized inside the cell, it is usually modified by the addition of extra functional groups to the polypeptide chain. These can be sugar or phosphate groups and may confer to the protein special functions such as: the ability to recognize other molecules, to integrate in the plasma membrane, to catalyze biochemical reactions, and various other processes. It is in the interest of the biochemist to understand what proteins are modified, what the modification is, and where it is located. An easy way to do this is by using mass spectrometry. In a sample of protein submitted to mass spectrometry you will see both modified and unmodified protein signals. The change in mass between these signals will correspond to the change in mass of your protein due to your post-translation modification.