Biochemistry/Catalysis
<< Biochemistry | << Thermodynamics | Catalysis | Metabolism and energy >>
Catalysis refers to the acceleration of the rate of a chemical reaction by a substance, called a catalyst, that is itself unchanged by the overall reaction. Catalysis is crucial for any known form of life, as it makes chemical reactions happen much faster than they would "by themselves", sometimes by a factor of several million times.
A common misunderstanding is that catalysis "makes the reaction happen", that the reaction would not otherwise proceed without the presence of the catalyst. However, a catalyst cannot make a thermodynamically unfavorable reaction proceed. Rather, it can only speed up a reaction that is already thermodynamically favorable. Such a reaction in the absence of a catalyst would proceed, even without the catalyst, although perhaps too slowly to be observed or to be useful in a given context.
Catalysts accelerate the chemical reaction by providing a lower energy pathway between the reactants and the products. This usually involves the formation of an intermediate, which cannot be formed without the catalyst. The formation of this intermediate and subsequent reaction generally has a much lower activation energy barrier than is required for the direct reaction of reactants to products.
Catalysis is a very important process from an industrial point of view since the production of most industrially important chemicals involve catalysis. Research into catalysis is a major field in applied science, and involves many fields of chemistry and physics.
Two types of catalysis are generally distinguished. In homogeneous catalysis the reactants and catalyst are in the same phase. For example acids (H+ ion donors) are common catalysts in many aqueous reactions. In this case both the reactants and the catalysts are in the aqueous phase. In heterogeneous catalysis the catalyst is in a different phase than the reactants and products. Usually, the catalyst is a solid and the reactants and products are gases or liquids. In order for the reaction to occur one or more of the reactants must diffuse to the catalyst surface and adsorb onto it. After reaction, the products must desorb from the surface and diffuse away from the solid surface. Frequently, this transport of reactants and products from one phase to another plays a dominant role in limiting the rate of reaction. Understanding these transport phenomena is an important area of heterogeneous catalyst research.
Enzymes
[edit | edit source]Enzyme (from Greek, in ferment) are special protein molecules whose function is to facilitate or otherwise accelerate most chemical reactions in cells. They are simply biological catalysts. Most enzymes are proteins, although a few are catalytic RNA molecules called ribozymes. Many chemical reactions occur within biological cells, but without catalysts most of them happen too slowly in the test tube to be biologically relevant.
Enzymes can also serve to couple two or more reactions together, so that a thermodynamically favorable reaction can be used to "drive" a thermodynamically unfavorable one. One of the most common examples is enzymes which use the dephosphorylation of ATP to drive some otherwise unrelated chemical reaction.
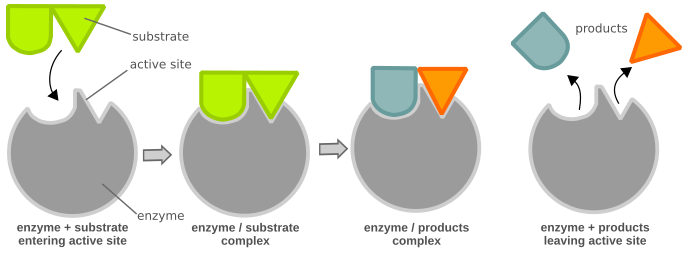
Chemical reactions need a certain amount of activation energy to take place. Enzymes can increase the reaction speed by favoring or enabling a different reaction path with a lower activation energy (Fig. 1), making it easier for the reaction to occur. Enzymes are large globular proteins that catalyze (accelerate) chemical reactions. They are essential for the function of cells. Enzymes are very specific as to the reactions they catalyze and the chemicals (substrates) that are involved in the reactions. Substrates fit their enzymes like a key fits its lock (Fig. 2). Many enzymes are composed of several proteins that act together as a unit. Most parts of an enzyme have regulatory or structural purposes. The catalyzed reaction takes place in only a small part of the enzyme called the active site, which is made up of approximately 2 - 20 amino acids.

The substrates (A and B) need a large amount of energy (E1) to reach the intermediate state A...B, which then reacts to form the end product (AB). The enzyme (E) creates a microenvironment in which A and B can reach the intermediate state (A...E...B) more easily, reducing the amount of energy needed (E2). As a result, the reaction is more likely to take place, thus improving the reaction speed.
Enzymes can perform up to several million catalytic reactions per second. To determine the maximum speed of an enzymatic reaction, the substrate concentration is increased until a constant rate of product formation is achieved (Fig. 3). This is the maximum velocity (Vmax) of the enzyme. In this state, all enzyme active sites are saturated with substrate. This was proposed in 1913 by Leonor Michaelis and Maud Menten. Since the substrate concentration at Vmax cannot be measured exactly, enzymes are characterized by the substrate concentration at which the rate of reaction is half its maximum. This substrate concentration is called the Michaelis-Menten constant (KM). Many enzymes obey Michaelis-Menten kinetics.
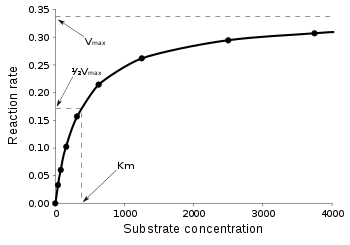
The speed V means the number of reactions per second that are catalyzed by an enzyme. With increasing substrate concentration [S], the enzyme is asymptotically approaching its maximum speed Vmax, but never actually reaching it. Because of that, no [S] for Vmax can be given. Instead, the characteristic value for the enzyme is defined by the substrate concentration at its half-maximum speed (Vmax/2). This KM value is also called Michaelis-Menten constant.
Several factors can influence the reaction speed, catalytic activity, and specificity of an enzyme. Besides de novo synthesis (the production of more enzyme molecules to increase catalysis rates), properties such as pH or temperature can denature an enzyme (alter its shape) so that it can no longer function. More specific regulation is possible by posttranslational modification (e.g., phosphorylation) of the enzyme or by adding cofactors like metal ions or organic molecules (e.g., NAD+, FAD, CoA, or vitamins) that interact with the enzyme. Allosteric enzymes are composed of several subunits (proteins) that interact with each other and thus influence each other's catalytic activity. Enzymes can also be regulated by competitive inhibitors (Fig. 4) and non-competitive inhibitors and activators (Fig. 5). Inhibitors and activators are often used as medicines, but they can also be poisonous.

A competitive inhibitor fits the enzyme as well as its real substrate, sometimes even better. The inhibitor takes the place of the substrate in the active center, but cannot undergo the catalytic reaction, thus inhibiting the enzyme from binding with a substrate molecule. Some inhibitors form covalent bonds with the enzyme, deactivating it permanently (suicide inhibitors). In terms of the kinetics of a competitive inhibitor, it will increase Km but leave Vmax unchanged.

Non-competitive inhibitors/activators (I) do not bind to the active center, but to other parts of the enzyme (E) that can be far away from the substrate (S) binding site. By changing the conformation (the three-dimensional structure) of the enzyme (E), they disable or enable the ability of the enzyme (E) to bind its substrate (S) and catalyze the desired reaction. The noncompetitive inhibitor will lower Vmax but leave Km unchanged.
An uncompetitive inhibitor will only bind to the enzyme-substrate complex forming an enzyme-substrate-inhibitor (ESI) complex and cannot be overcome by additional substrate. Since the ESI is nonreactive, Vmax is effectively lowered. The uncompetitive inhibitor will in turn lower the Km due to a lower concentration of substrate needed to achieve half the maximum concentration of ES.
Several enzymes can work together in a specific order, creating metabolic pathways (e.g., the citric acid cycle, a series of enzymatic reactions in the cells of aerobic organisms, important in cellular respiration). In a metabolic pathway, one enzyme takes the product of another enzyme as a substrate. After the catalytic reaction, the product is then passed on to another enzyme. The end product(s) of such a pathway are often non-competitive inhibitors (Fig. 5) for one of the first enzymes of the pathway (usually the first irreversible step, called committed step), thus regulating the amount of end product made by the pathway (Fig. 6).

- The basic feedback inhibition mechanism, where the product (P) inhibits the committed step (A⇀B).
- Sequential feedback inhibition. The end products P1 and P2 inhibit the first committed step of their individual pathway (C⇀D or C⇀F). If both products are present in abundance, all pathways from C are blocked. This leads to a buildup of C, which in turn inhibits the first common committed step A⇀B.
- Enzyme multiplicity. Each end product inhibits both the first individual committed step and one of the enzymes performing the first common committed step.
- Concerted feedback inhibition. Each end product inhibits the first individual committed step. Together, they inhibit the first common committed step.
- Cumulative feedback inhibition. Each end product inhibits the first individual committed step. Also, each end product partially inhibits the first common committed step.
Enzymes are essential to living organisms, and a malfunction of even a single enzyme out of approximately 2,000 present in our bodies can lead to severe or lethal illness. An example of a disease caused by an enzyme malfunction in humans is phenylketonuria (PKU). The enzyme phenylalanine hydroxylase, which usually converts the essential amino acid phenylalanine into tyrosine does not work, resulting in a buildup of phenylalanine that leads to mental retardation. Enzymes in the human body can also be influenced by inhibitors in good or bad ways. Aspirin, for example, inhibits an enzyme that produces prostaglandins (inflammation messengers), thus suppressing pain. But not all enzymes are in living things. Enzymes are also used in everyday products such as biological washing detergents where they speed up chemical reactions, (to get your clothes clean).
Digestive and Metabolic Enzymes
[edit | edit source]In the previous section we have been talking about the digestive enzymes, both the ones produced by the body, such as salivary amylase, and the food enzymes. Their primary role is for the digestion of food. Another class of enzymes is called metabolic enzymes. Their role is to catalyze chemical reactions involving every process in the body, including the absorption of oxygen. Our cells would literally starve for oxygen even with an abundance of oxygen without the action of the enzyme, cytochrome oxidase. Enzymes are also necessary for muscle contraction and relaxation. The fact is, without both of these classes of enzymes, (digestive and metabolic,) life could not exist. Digestive enzymes function as biological catalysts in which it helps to breakdown carbohydrates, proteins, and fats. On the other hand, metabolic enzymes function as a remodel of cells. Digestion of food has a high priority and demand for enzymes; digestive enzymes get priority over metabolic enzymes. Any deficiency in metabolic enzyme can lead to over work, which could lead to enlarge organs in order to perform the increased workload. The result is unhealthy and could cause enlarged heart or pancreas. The deficiencies of metabolic enzymes can have a tremendous impact on health. As we grow older enzyme level decline and the efficiency in the body decline.
Enzyme naming conventions
[edit | edit source]By common convention, an enzyme's name consists of a description of what it does, with the word ending in "-ase". Examples are alcohol dehydrogenase and DNA polymerase. Kinases are enzymes that transfer phosphate groups. The International Union of Biochemistry and Molecular Biology has developed a nomenclature for enzymes, the EC numbers; each enzyme is described by a sequence of four numbers, preceded by "EC". The first number broadly classifies the enzyme based on its mechanism:
- EC 1 Oxidoreductases: catalyze oxidation/reduction reactions
- EC 2 Transferases: transfer a functional group (e.g., a methyl or phosphate group)
- EC 3 Hydrolases: catalyze the hydrolysis of various bonds
- EC 4 Lyases: cleave various bonds by means other than hydrolysis and oxidation
- EC 5 Isomerases: catalyze isomerization changes within a single molecule
- EC 6 Ligases: join two molecules with covalent bonds
Some other important enzymes are: Protease: breaks the protein into amino acids in high acidity environments such as stomach, pancreatic and intestinal juices. Act on bacteria, viruses and some cancerous cells. Amylase: Break complex carbohydrates such as starch into simpler sugars (dextrin and maltose). It found in the intestines, pancreas and also in salivary glands. Lipase: breaks down fats and some fat soluble vitamins (A,E,K, and D). helpful in treating cardiovascular diseases. Cellulase: break down cellulose that found in fruits, grains, and vegetables. It increases the nutritional values of vegetables, and fruits. Pectinase: break down pectin that found in citrus fruits, carrots, beets, tomatoes, and apple. Antioxidants: protect from free radical negative effect that can damage cell in the body. Cathepsin: break animal protein down. Lactase: break down lactose that found in milk products. the production of lactase decrease with age. Invertase: assimilate sucrose that can contribute to digestive stress if not digested properly. Papain: break down protein and help the body in digestion. Bromelain: Break proteins that found in plants and animals. it could help the body to fight cancer and treat inflammation. Glucoamylase: break down maltose that found in all grains in to two glucose molecules.
<< Biochemistry | << Thermodynamics | Catalysis | Metabolism and energy >>