Amateur Radio Manual/Atoms
Note that this information is presented in very simplistic form for clarity. Atomic theory is much more complicated than most radio amateurs need to know but those that wish to know more should explore the other wiki books or wikipedia entries on the subject.
Atoms
[edit | edit source]The smallest building block of all matter is the atom (from the Greek Atomos). An atom is the smallest little bit that retains all the characteristics of the element it represents. For instance, if you had an atom of copper, and you cut it further, it would no longer retain the characteristics of copper.
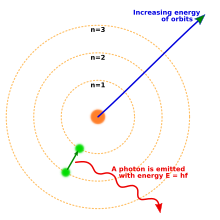
Atoms consist of smaller particles. In the nucleus, or centre of the atom, are neutrons and protons. Protons have a positive charge. Orbiting the nucleus of the atom are electrons that have a negative charge. [See Bohr atom. In simplest terms, electrons orbit the nucleus in orbits or shells. Each shell represents an energy state. The further from the nucleus, the more energetic the electron is. Each shell wants to contain a certain number of electrons to remain electrically stable. Neutral atoms have the same number of electrons and protons. This equal number is the atomic number of the element.
Note that the terms positive and negative do not mean more and less in quantity but refer to the type of charge.
What is interesting is that particles that have the same charge repel each other and particles that have opposite charges attract each other. If a proton meets an electron they join up to become a hydrogen atom, the simplest element.
If you add electrons and protons (and a scattering of neutrons which are neutral) you create the various elements of the world around us.
Now, lets look at the copper atom, because it is one of the best known conductors of electricity. Silver and platinum are better conductors, but cost too much for general use, unless you are building a satellite.
Copper has an atomic number of 29. This tells us that there are 29 protons in the nucleus and 29 electrons arranges in shells or orbits around the nucleus. The first shell contains 2 ... leaving us 27 electrons, the next shell wants 8 ... leaving us 19 electrons .... the 3rd shell contains eighteen ... leaving us 1 electron for the 4th shell. Whoops.. the fourth shell wants 32 electrons. Electrons in an outer unfilled shell are known as valence electrons. It is possible to supply enough energy to the atom to overcome the electron's attraction to the nucleus and break it free from the atom. Most metals fall into this category. Because it only takes a little energy to break the electrons free, most metals are considered to be conductors. Elements that require a great deal of energy to break their electrons free are considered to be insulators.