A-level Applied Science/The Role of the Pathology Service/Biochemistry
Clinical biochemistry (also known as chemical pathology or clinical chemistry) is the area of pathology that is generally concerned with analysis of bodily fluids.
The discipline originated in the late 19th century with the use of simple chemical tests for various components of blood and urine. Subsequently other techniques were applied including the use and measurement of enzyme activities, spectrophotometry, electrophoresis and immunoassay.
Most current laboratories are now highly automated and use assays that are closely monitored and quality controlled.
Tests that require examination and measurement of the cells of blood, as well as blood clotting studies, are not included as these are usually grouped under haematology.
Type of work
[edit | edit source]Biochemical tests are also known as 'chemical pathology'. The tests are performed on any kind of body fluid, but mostly on serum or plasma. Serum is the yellow watery part of blood that is left after blood has been allowed to clot and all blood cells have been removed. This is most easily done by centrifugation which packs the more dense blood cells and platelets to the bottom of the centrifuge tube, leaving the liquid serum fraction resting above the packed cells. Plasma is essentially the same as serum, but is obtained by centrifuging the blood without clotting. Plasma therefore contains all of the clotting factors, including fibrinogen. Biochemists test samples for different biochemical components.
- Immunology test for antibodies.
- DNA analysis is also done in large medical laboratories.
- Hormones
- Minerals
- Vitamins
- Metabolites
- Enzymes
A large laboratory will accept samples for up to about 700 different kinds of tests. Even the largest of laboratories rarely does all these tests themselves and some need to be referred to other labs. This large array of tests can be further sub-categorised into sub-specialities of:
- General or routine chemistry
- Endocrinology - the study of hormones
- Immunology - the study of the immune system and antibodies
- Pharmacology or Toxicology - the study of drugs
Chemical pathology tests
[edit | edit source]Common chemical pathology tests include:
- w:Sodium;
- w:Potassium;
- w:Chloride;
- w:Bicarbonate;
- w:Urea;
- w:Creatinine;
- w:Calcium;
- w:Phosphate;
- Albumin;
- w:Bilirubin;
- AST;
- ALT;
- w:GGT;
- w:Alkaline phosphatase;
- w:Magnesium;
- w:Osmolality;
- w:Urate;
- w:Iron;
- w:Transferrin;
- w:Total protein;
- w:Globulins;
- w:Glucose;
- w:C-reactive protein;
- w:HbA1c.
- w:Blood gases ( )
Automation
[edit | edit source]The biochemistry department uses more automated analysis than other departments. This is due to the:
- reliability and validity of testing procedures.
- volumes of samples required.
- numbers samples tested in one day.
Gel electrophoresis
[edit | edit source]
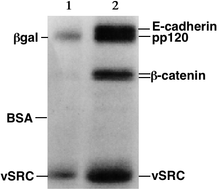
Gel electrophoresis is a group of techniques used by scientists to separate molecules based on physical characteristics such as size, shape, or isoelectric point. Gel electrophoresis is usually performed for analytical purposes, but may be used as a preparative technique to partially purify molecules prior to use of other methods such as mass spectrometry, PCR, cloning, DNA sequencing, or Western immuno-blotting for further characterisation.
Separation
[edit | edit source]"Gel", refers to the matrix used to separate the molecules. In most cases the gel is a crosslinked polymer whose composition and porosity is chosen based on the weight and composition of the target of the analysis. When separating proteins or small nucleic acids (DNA, RNA, or oligonucleotides) the gel is usually made with different concentrations of acrylamide and a cross-linker, producing different sized mesh networks of polyacrylamide. When separating larger nucleic acids (greater than a few hundred bases), the preferred matrix is purified agarose (a component of agar which is a red seaweed extract). In both cases, the gel forms a solid but porous matrix that looks and feels like clear jelly. Acrylamide, in contrast to polyacrylamide, is a neurotoxin and needs to be handled using Good Laboratory Practices (GLP) to avoid poisoning.
"Electrophoresis", refers to the electromotive force (EMF) that is used to push or pull the molecules through the gel matrix; by placing the molecules in wells in the gel and applying an electric current, the molecules will move through the matrix at different rates, towards the anode if negatively charged or towards the cathode if positively charged (note that gel electrophoresis operates as an electrolytic cell; the anode is positive and the cathode is negative).
Visualisation
[edit | edit source]After the electrophoresis runs, when the smallest molecules have almost reached the anode, the molecules in the gel can be stained to make them visible. Ethidium bromide, silver, or coomassie blue dye can be used. Other methods can also be used to visualize the separation of the mixture's components on the gel. If the analyte molecules luminesce under ultraviolet light, a photograph can be taken of the gel under ultraviolet light. If the molecules to be separated contain radioactive atoms, an autoradiogram can be recorded of the gel (as in the example shown here).
If several mixtures have initially been injected next to each other, they will run parallel in individual lanes. Depending on the number of different molecules, each lane shows separation of the components from the original mixture as one or more distinct bands, one band per component. Incomplete separation of the components can lead to overlapping bands, or to indistinguishable smears representing multiple unresolved components.
Bands in different lanes that end up at the same distance from the top contain molecules that passed through the gel with the same speed, which usually means they are approximately the same size. There are special markers available - ladders - which contain a mixture of molecules of known sizes. If such a marker was run on one lane in the gel parallel to the unknown samples, the bands observed can be compared to those of the unknown in order to determine their size. The distance a band travels is approximately inversely proportional to the logarithm of the size of the molecule.
Applications
[edit | edit source]Gel electrophoresis is used in molecular biology, genetics, microbiology and biochemistry. The results can be analysed quantitatively by visualising the gel with UV light, or by staining the separated molecules. A gel imaging device records the image with a computer operated camera, and the intensity of the band or spot of interest is measured and compared against standard or markers loaded on the same gel. The measurement and analysis are mostly done with specialized software.
Nucleic acids
[edit | edit source]In the case of nucleic acids, the direction of migration, from negative to positive electrodes, is due to the natural negative charge carried on their sugar-phosphate backbone. Double-stranded DNA fragments naturally behave as long rods, so their migration through the gel is relative to their radius of gyration, or, for non-cyclic fragments, roughly size. Single-stranded DNA or RNA tend to fold up into molecules with complex shapes and migrate through the gel in a complicated manner based on their tertiary structure. Therefore, agents that disrupt the hydrogen bonds, such as sodium hydroxide or formamide, are used to renature the nucleic acids and cause them to behave as long rods again.
Gel electrophoresis of large DNA] or RNA molecules is usually done by agarose gel electrophoresis. See the "w:Chain termination method" page for an example of a polyacrylamide DNA sequencing gel.
Proteins
[edit | edit source]Proteins, on the other hand, can have different charges and complex shapes, therefore they may not migrate into the gel at similar rates, or at all, when placing a negative to positive EMF on the sample. Proteins therefore, are usually denatured in the presence of a detergent such as sodium dodecyl sulphate/sodium dodecyl phosphate (SDS/SDP) that coats the proteins with a negative charge. Generally, the amount of SDS bound is relative to the size of the protein (usually 1.4 g SDS per gram of protein), so that the resulting denatured proteins have an overall negative charge, and all the proteins have a similar charge to mass ratio. Since denatured proteins act like they were long rods instead of having a complex tertiary shape, the rate at which the resulting SDS coated proteins migrate in the gel is relative only to its size and not its charge or shape.
Proteins are usually analysed by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE), by native gel electrophoresis], by quantitative preparative native continuous polyacrylamide gel electrophoresis (QPNC-PAGE), or by 2-D electrophoresis.
Class experiments
[edit | edit source]Using simple clinical dipsticks, a mock urine sample can be analysed. Glucose, ketone and protein levels can be established to identify possible diseases.
Thin layer chromatography separations of carbohydrates, proteins and amino acids or lipids are simple to perform and introduce candidates to the concepts of separation of molecules being related to the chemical properties.
If access to electrophoresis equipment is not possible, teachers could approach the hospital biochemistry department or a local university biological sciences department and request a demonstration day, or arrange for someone to visit who can demonstrate the technique with samples gathered from the candidates. Protein electrophoresis equipment is available from a range of companies and the National Centre for Biotechnology Education offers a range of practical activities and resources for electrophoresis.
Paper chromatography
[edit | edit source]'Paper chromatography is an analytical technique for separating and identifying compounds that are or can be coloured, especially pigments. This method has been largely replaced by thin layer chromatography, however it is still a powerful teaching tool. Two-way paper chromatography, also called two-dimensional chromatography, involves using two solvents and rotating the paper 90° in between. This is useful for separating complex mixtures of similar compounds, for example, amino acids.
Technique
[edit | edit source]
A small spot of solution containing the sample is applied to a strip of chromatography paper about 1 cm from the base. This sample is adsorbed onto the paper and may form interactions with it. Any substance that reacts or bonds with the paper cannot be measured using this technique. The paper is then dipped in to a suitable solvent such as ethanol or water, and placed in a sealed container.
The solvent moves up the paper by capillary action, which occurs as a result of the attraction of the solvent molecules to the paper and to one another. As the solvent rises through the paper it meets and dissolves the sample mixture, which will then travel up the paper with the solvent. Different compounds in the sample mixture travel at different rates due to differences in solubility in the solvent, and due to differences in their attraction to the fibres in the paper. Paper chromatography takes anywhere from several minutes to several hours.
In some cases, paper chromatography does not separate pigments completely; this occurs when two substances appear to have the same Rf values in a particular solvent. In these cases, two-way chromatography is used to separate the multiple-pigment spots. The chromatogram is turned by ninety degrees, and placed in a different solvent in the same way as before; some spots separate into multiple spots, showing the presence of more than one pigment. As before the Rf value is calculated, and the two pigments identified.
Thin-layer chromatography
[edit | edit source]
Thin layer chromatography (TLC) is a widely-used chromatography technique used to separate chemical compounds. It involves a stationary phase consisting of a thin layer of adsorbent material, usually silica gel, alumina, or cellulose immobilised onto a flat, inert carrier sheet. A liquid phase consisting of the solution to be separated dissolved in a solvent is drawn through the plate via capillary action, separating the experimental solution. It can be used to determine the pigments a plant contains, to detect pesticides or insecticides in food, in forensics to analyse the dye composition of fibres, or to identify compounds present in a given substance, among other uses.
Plate preparation
[edit | edit source]TLC plates are made by mixing the adsorbent, such as silica gel, with a small amount of inert binder like calcium sulphate (gypsum) and water. This mixture is spread as a thick slurry on an inert carrier sheet, usually glass, thick aluminium foil, or plastic, and the resultant plate is dried and activated by heating in an oven. The thickness of the adsorbent layer is typically around 0.1–0.25 mm for analytical purposes and around 1–2 mm for preparative TLC.
Technique
[edit | edit source]The process is similar to paper chromatography with the advantage of faster runs, better separations, and the choice between different stationary phases. Because of its simplicity and speed TLC is often used for monitoring chemical reactions and for the qualitative analysis of reaction products.
A small spot of solution containing the sample is applied to a plate, about one centimeter from the base. The plate is then dipped in to a suitable solvent, such as ethanol or water, and placed in a sealed container. The solvent moves up the plate by capillary action and meets the sample mixture, which is dissolved and is carried up the plate by the solvent. Different compounds in the sample mixture travel at different rates due to differences in solubility in the solvent, and due to differences in their attraction to the stationary phase.
Analysis of chromatograms
[edit | edit source]After development, the spots corresponding to different compounds may be located by their colour, by ultraviolet light, ninhydrin or by treatment with iodine vapours. The final chromatogram can be compared with other known mixture chromatograms to identify sample mixes using the Rf value.
As in most other forms of chromatography, paper chromatography uses Rf values to help identify compounds. Rf values are calculated by dividing the distance the pigment travels up the paper by the distance the solvent travels (the solvent front). Because Rf values are standard for a given compound, known Rf values can be used to aid in the identification an unknown substance in an experiment.
As the chemicals being separated may be colourless, several methods exist to visualise the spots:
- Often a small amount of a fluorescent compound, usually Manganese-activated Zinc Silicate, is added to the adsorbent that allows the visualisation of spots under a blacklight(UV254). The adsorbent layer will thus fluoresce light green by itself, but spots of analyte quench this fluorescence.
- Iodine vapours are a general unspecific colour reagent
- Specific colour reagents exist into which the TLC plate is dipped or which are sprayed onto the plate
Once visible, the Rf value of each spot can be determined by dividing the distance travelled by the product by the total distance travelled by the solvent (the solvent front). These values depend on the solvent used, and the type of TLC plate, and are not physical constants.
See also
[edit | edit source]- w:Reference ranges for common blood tests
- w:Capillary electrophoresis
- w:DNA electrophoresis
- w:Zymography
- w:Gel isolation
- w:Protein electrophoresis
- w:Electrofocusing
- w:Southern blotting
- w:Northern blotting
- w:Western blotting
- w:Two-dimensional gel electrophoresis
- w:Temperature gradient gel electrophoresis
References
[edit | edit source]
External links
[edit | edit source]- Biotechniques Laboratory electrophoresis demonstration, from the University of Utah's Genetic Science Learning Center
- Protein electrophoresis and Western Blotting
![{\displaystyle [H^{+}],P_{{\mathrm {CO} }_{2}},P_{{\mathrm {O} }_{2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d1570dae6464e073170e7b9b8759243c27e93350)