8th Grade Science/Section 1: Elements
Elemental Overview
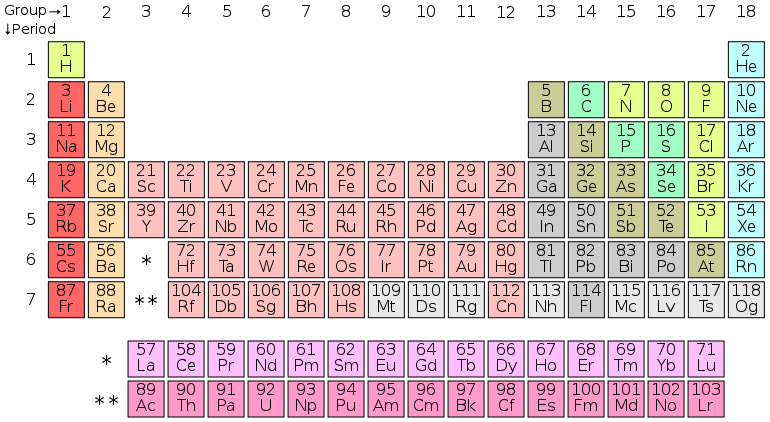
[edit | edit source]The elements of the periodic table are all different types of atoms that make up the structural basis of the physical universe. There are over 100 currently known elements. The elements are numbered according to the number of protons that they have, because this is what gives every atom its unique properties. Each atom contains a nucleus, which is surrounded by an orbiting cloud of negatively charged electrons. The nucleus is composed of protons, which are positively charged, and neutrons, which have no charge. For every proton, there is a mutual electron, which means that all elements have no electrical charge. For instance, the most common of all elements in the universe is hydrogen. Hydrogen has 1 proton, 1 electron, and either 1 or 2 neutrons. Since it has 1 proton, it is given the number 1 on the periodic table.
The electrons in orbit around each element dictates not only its location on the table, but also how it interacts with other elements. Each element has a set of orbits of electrons surrounding in it, and depending on the number of electrons and the orbitals they occupy, determines what it shares its electrons with. The periodic table is split this way to distinguish between elements that are electron deficient (left side; alkaline and earth metals), also deficient but all have an incomplete d-orbital (middle; transition metals), electron rich (right side; nonmetals). There is also the special group known as the noble elements. These are formed with a full outer electron ring, which makes them unable to react with anything except in special circumstances.
Electron Orbitals
[edit | edit source]As mentioned before, each element has a cloud of electrons surrounding it, and depending on that number dictates how it behaves and interacts with other elements. Every atom's first ring is stable when filled with two electrons. Every ring after that in general must have eight to be stable. Nature tries to get to balance in the least amount of effort possible. An atom with less than four electrons will try to get in any ring except the first ring will try to empty the outermost ring. An atom with more than four electrons, on the other hand, will try to fill the outermost ring. This is known as the Octahedral Rule, or the Rule of Eight. Hydrogen has an atomic number of 1, meaning it has one proton, and thus one electron. This one electron goes to fill to the s-orbital half way. Each pair of electrons that orbit an element requires one to be “spinning up” while the other electron “spins down” to create stability for the atom. An important rule of chemistry is that everything wants to find the state of most stability. An atom is stable when its outer electron ring, known as the Valence Ring, is full. Since hydrogen only has one electron, hydrogen atoms generally bond with other hydrogen atoms. The lements that naturally bond with themselves are like this are known as the Diatomic Elements, from the Greek "Di", which means two.
The next element after hydrogen is helium. Helium has an atomic number of two, with two electrons filling the s-orbital. Because the first ring needs only two electrons, helium is very stable. Not only is helium very stable but all the elements that share a column with it also share this full orbital configuration are stable too. These elements are the noble elements, which were mentioned above. As you travel down the periodic table (and up in atomic numbers) the more orbitals like p and d get filled. The electrons are distributed as such (with the leading number representing the orbital number and the last number showing the number of electrons): 1S2 2S2 2P6 3S2 3P6 4S2 3D10 4P6 4D10 4F14 There is an occasional exception along the way of how the electrons are distributed for an occasional element, otherwise the periodic table serves as an excellent way to quickly determine the orbital of an element based on trends in the table.