Chemical Sciences: A Manual for CSIR-UGC National Eligibility Test for Lectureship and JRF/Named Reactions/Pinacol-Pinacolone Rearrangement
| This page was imported and needs to be de-wikified. Books should use wikilinks rather sparsely, and only to reference technical or esoteric terms that are critical to understanding the content. Most if not all wikilinks should simply be removed. Please remove {{dewikify}} after the page is dewikified. |
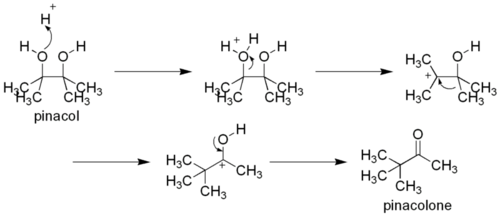
The pinacol rearrangement or pinacol-pinacolone rearrangement is a method for converting a 1,2-diol to a carbonyl compound in organic chemistry. This rearrangement takes place under acidic conditions. The name of the reaction comes from the rearrangement of pinacol to pinacolone.
This reaction was first described by Wilhelm Rudolph Fittig in 1860.[1]
An overview of mechanism
[edit | edit source]In the course of this organic reaction, protonation of one of the -OH groups occurs and a carbocation is formed. If both the -OH groups are not alike, then the one which yields a more stable carbocation participates in the reaction. Subsequently, an alkyl group from the adjacent carbon migrates to the carbocation center. The driving force for this rearrangement step is believed to be the relative stability of the resultant oxonium ion, which has complete octet configuration at all centers (as opposed to the preceding carbocation). The migration of alkyl groups in this reaction occurs in accordance with their usual migratory aptitude, i.e. hydride > Ph- > tertiary > secondary > methyl .
Stereochemistry of the rearrangement
[edit | edit source]In cyclic systems, the reaction presents more features of interest. In these reactions, the stereochemistry of the diol plays a crucial role in deciding the major product. An alkyl group which is situated trans- to the leaving -OH group alone may migrate. If otherwise, ring expansion occurs, i.e. the ring carbon itself migrates to the carbocation centre. This reveals another interesting feature of the reaction, viz. that it is largely concerted. There appears to be a connection between the migration origin and migration terminus throughout the reaction.
Moreover, if the migrating alkyl group has a chiral center as its key atom, the configuration at this center is retained even after migration takes place.
History
[edit | edit source]Although Fittig first published about the pinacol rearrangement, not he but Aleksandr Butlerov did correctly identify the reaction products involved.[2]
In a 1859 publication Wilhelm Rudolph Fittig described the reaction of acetone with potassium metal..[3]. Fittig wrongly assumed a molecular formula of (C3H3O)n for acetone, the result of a long standing atomic weight debate finally settled at the Karlsruhe Congress in 1860. He also wrongly believed acetone to be an alcohol which he hoped to prove by forming a metal alkoxide salt. The reaction product he obtained instead he called paraceton which he believed to be a acetone dimer. In his second publication in 1860 he reacted paraceton with sulfuric acid (the actual pinacol rearrangement).
Again Fittig was unable to assign a molecular structure to the reaction product which he assumed to be another isomer or a polymer. Contemporary chemists who had already adapted to the new atomic weight reality did not fare better. One of them, Charles Friedel, believed the reaction product to be the epoxide tetramethylethylene oxide [4] in analogy with reactions of ethylene glycol. Finally Butlerov in 1873 came up with the correct structures after he independently synthesised the compound trimethylacetic acid which Friedel had obtained earlier by oxidizing with a dichromate. [5]
Some of the problems during the determination of the structure are due to the fact that carbon skeletal rearrangements were unknown at that time and therefore the new concept had to be found. Butlerov theory allowed the structure of carbon atoms in the molecule to rearrange and with this concept a structure for pinacolone could be found.
References
[edit | edit source]- ↑ Wilhelm Rudolph Fittig (1860). "Ueber einige Derivate des Acetons". Annalen der Chemie und Pharmacie. 114 (1): 54–63. doi:10.1002/jlac.18601140107.
- ↑ Jerome A. Berson (2002). "What Is a Discovery? Carbon Skeletal Rearrangements as Counter-Examples to the Rule of Minimal Structural Change". Angewandte Chemie International Edition. 41 (24): 4655–4660. doi:10.1002/anie.200290007.
- ↑ W. R. Fittig (1859). "Ueber einige Metamorphosen des Acetons der Essigsäure". Annalen der Chemie und Pharmacie. 110 (1): 23–45. doi:10.1002/jlac.18591100104.
- ↑ Charles Friedel (1869). "Recherches sur les acétones et sur les aldéhydes". Annales de chimie et de physique Ser. 4. 16: 310.
- ↑ Aleksandr Butlerov (1873). "Ueber Trimethylessigsäure". Justus Liebigs Annalen der Chemie und Pharmacie. 170 (1–2): 151–162. doi:10.1002/jlac.18731700114.