Structural Biochemistry/Adderall

Intro[edit | edit source]
Adderall is a specific brand name drug that is used mostly for treatment of ADHD (Attention Deficit Hyperactivity Disorder) but can also be used to treat certain forms of narcolepsy. Dextroamphetamine-amphetamine otherwise known as Adderall, is a drug available by prescription that aids in the concentration of patients who suffer from attention disorders such as Attention Deficit Hyperactivity Disorder (ADHD) and sleeping disorders like narcolepsy. Adderall is a schedule II drug in the US, meaning that it has the ability to cause physical and psychological dependence, has a high potential of being abused (due to the fact that it is a combination out of four amphetamine salts) but has been approved for medical usage[1]. Adderall is a stimulant meaning that it is a psychoactive drug that can improve mental and physical functions. It is also a relative to the phenethylamine and amphetamine families.</g>
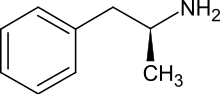
Medical use[edit | edit source]
As stated above, Adderall is a prescription medication used to treat many forms of ADHD (increased or more difficulty focusing, controlling actions, and remaining still or quiet than other people who are the same age) and narcolepsy (a sleep disorder that causes excessive daytime sleepiness and sudden attacks of sleep),[1] although its primary role is to treat ADHD patients. There are two forms commonly found on the market: Adderall XR (extended release) and Adderall IR (instant release). Both are used to treat ADHD, but only the IR form is used in treatment of narcolepsy. Adderall is thought to stop the reuptake of chemicals such as norepinephrine and dopamine, which are thought to better aid persons with ADHD as different levels of the chemicals should cause a better ability to focus, to control actions and finally to remain silent, or still in relation to persons from their same age group.
Side effects[edit | edit source]
Many side effects include : nervousness, restlessness, difficulty falling asleep, or staying asleep, uncontrollable shaking of a part of the body, headache, changes in sex drive or ability, dry mouth, stomach pain, nausea, vomiting, diarrhea, loss of appetite, weight loss. Other more adverse side effects many occur.
Chemistry[edit | edit source]
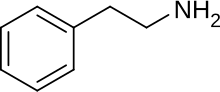

As previously stated, Adderall is a class of both the phenethylamine and amphetamine families. Phenethylamine is an organic compound that is part of many drugs that have psychoactive and stimulant effects [2] and is typically found in stimulants, psychedelics, entactogens, anorectics, bronchodilators, decongestants and anti-depressants [3]. Amphetamines are psychostimulants that are a branch of the phenethylamine class that induces focus and wakefulness. More specifically, dextroamphetamine is a stereoisomer of amphetamine. The dextro- prefix signifies the clockwise rotation of a polarized light plane. Since Adderall is composed of dextroamphetamine and amphetamine; it is composed of both enantiomers of amphetamine.
Biochemical Interactions/Composition[edit | edit source]
Adderall is an amphetamine, which can be described as non-catecholamine sympathomimetic amines with central nervous system stimulant effects. In essence, it is a mixture of amphetamine based salts which are thought to stop the reuptake of norepinephrine and dopamine back into the neurons. In the Adderall XR form, the composition of the drug includes a three to one ratio of d-amphetamines and l-amphetamines respectively. The four ingredients that are a part of Adderall XR include Dextroamphetamine saccharate, Amphetamine Aspartate, Dextroamphetamine Sulfate, and Amphetamine Sulfate, each of which are in equal proportions.
Pharmacology[edit | edit source]
The exact mechanism of dextroamphetamine (d-AMP) in treatment of ADHD is not yet completely understood, however it is known that it plays an important role in the efficiency of dopamine neurotransmission. The stereoisomer of dextroamphetamine, amphetamine is known for releasing dopamine from the nerve terminal by using dopamine active transporter (DAT) as a carrier [4]. Despite how Adderall is used to enhance concentration, studies have shown that amphetamines do not optimize short-term memory nor does it improve “cognitive flexibility.” In fact there is evidence that it may be impairing to short-term memory and cognitive flexibility [5]. D-AMP is also known to be an reuptake inhibitor because it is an substrate analog, therefore it competes with neurotransmitters (chatecholamine) such as dopamine with similar monoamine, groups for uptake [6]. Since d-AMP induces a dopamine effect by increasing its concentration, a user feels the effects of the excitatory neurotransmitter. At really high doses, d-AMP is known for causing euphoria by triggering a cascade of catecholamines that release large amounts of neurotransmitters and an overstimulation of receptors [8]. It is postulated that d-AMP tends to interact with the dopamine systems while its enantiomer tends to interact with the norepinephringernic systems [9].
Another way dextroamphetamine chemically affects the body was postulated through research in bipolar disorder by Silverston et. al.(2002). Studies suggest that dextroamphetamine increases the activity of the phosphoinositol cycle through the dopamine and noradrenaline. The phosphoinositol cycle was discovered by Lowell and Hopkins, and is known to generate phosphatidylinosol which is a precursor to lipid signaling molecules in eukaryotes to help regulate specific functions [10].
Pharmacokinetics[edit | edit source]
According to a medication of pamphlet of Adderall XR®, the mean elimination half-life for d-AMP is 10 hours in adults; 11 hours in persons less than or equal to 165 lbs and 9 hours in children 6-12 years of age. In the enantiomer, l-amphetamine, the mean elimination half-life is 13 hours, 13-14 hours in persons less than or equal to 165 lbs and 11 hours in children 6-12 years [11].
Metabolism[edit | edit source]
All enzymes for the metabolism of amphetamine has not yet been identified. However, CYP2D6 is known to be responsible for the formation of 4-hydroxyamphetamine which in turn oxidizes into α-hydroxyamphetamine and undergoes further unknown metabolism. Amphetamine is also known to be an inhibitor of CYP2D6 enzyme [11].
Secretion[edit | edit source]
Amphetamines are sensitive to pH with a pKa of 9.9 and are therefore dependent on the pH of the urine for proper disposal. “Acidic pHs and high flow rates result in increased renal elimination…indicating the involvement of active secretion [11].”
Adderall Abuse[edit | edit source]
Adderall is abused as:
- A study drug: Its nicknames include “college crack” or “cognitive steroid.” Students who take Adderall for studying report that they can focus on their book well, and can do better on the following exam than they would without the drug. - A party drug: Adderall contains dextroamphetamine, which creates a feeling of well-being, confidence and enhanced libido, and enables users to go without sleep for extended periods. - A weight-loss drug: Appetite conquest is often a side effect of Adderall and is mostly loved by young women looking for an easy way to lose weight.
Why are Adderall and abused?
The same properties that make Adderall effective in treating many cases of ADHD have made it a drug of choice for non-ADHD students, who find it tempting for other reasons.
Adderall works by changing the brain’s chemistry in a way that helps ADHD patients pay attention to the task at hand despite distractions. In non-ADHD users, Adderall produces a flow of focus and energy and an associated loss of appetite. Although casual Adderall consumption is considered as acceptable among students at many schools, use of any prescription drug for a purpose other than its prescribed use, or by a person for whom it hasn’t been prescribed, constitutes abuse.
The Results of Adderall Abuse
Misusing or abusing Adderall can cause a range of problems including difficulty sleeping, feelings of hostility, anxiety or paranoia, and a hyper-energetic, aroused state. Some users may experience undesirable or unhealthy weight loss. Especially when snorted, Adderall can cause a potentially dangerous increase in heart rate, body temperature and blood pressure. Caregivers screen ADHD patients to avoid prescribing Adderall to those with conditions that could make the drug dangerous for them. Abusers who take the drug without a prescription don’t have the benefit of such screening. Ironically, although the primary reason for Adderall’s abuse on campus is to enhance academic performance, students in whom the drug causes a manic state may not be able to exercise good judgment about the quality of the papers or exams they produce while influenced by Adderall. After abusing Adderall, users often experience a “crash” and suffer from exhaustion, nausea, depression or irritation. Some Adderall abusers smoke marijuana as an antidote for these distressing symptoms. After long-term abuse, your body depends on Adderall for normal function and trying to discontinue the drug can result in withdrawal symptoms including panic, suicidal thoughts and nightmares.
Reference[edit | edit source]
http://adhd.emedtv.com/adderall/adderall-abuse.html http://adderallabuse.com/ http://www.webmd.com/drugs/drug-63163-Adderall+Oral.aspx?drugid=63163&drugname=Adderall+Oral http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000166/ http://www.drugs.com/pro/adderall.html 1. 21 USC § 812 – Schedules of controlled substances | LII / Legal Information Institute http://www.law.cornell.edu/uscode/text/21/812
2. Glen R. Hanson, Peter J. Venturelli, Annette E. Fleckenstein (2005-11-03). "Drugs and society (Ninth Edition)". Jones and Bartlett Publishers. ISBN 978-0-7637-3732-0
3. Phenethylamine. http://en.wikipedia.org/wiki/Phenethylamine
4. Kuczenski, R., and D. S. Segal. 1997. Effects of methylphenidate on extracellular dopamine, serotonin, and norepinephrine: comparison with amphetamine. J. Neurochem. 68:2032–2037.
5. Lackhan S. et al. (15 June 2012). " Prescription stimulants in individuals with and
without attention deficit hyperactivity disorder: misuse, cognitive impact, and adverse effects". Brain and Behavior.
6. Kuczenski R et al. (1 February 1995). "Hippocampus Norepinephrine, Caudate Dopamine and Serotonin, and Behavioral Responses to the Stereoisomers of Amphetamine and Methamphetamine". The Journal of Neuroscience 15 (2): 1308–1317.
7. Patrick, and Markowitz; Markowitz, John S. (1997). "Pharmacology of Methylphenidate, Amphetamine Enantiomers and Pemoline in Attention-Deficit Hyperactivty Disorder".Human Psychopharmacology 12 (6): 527–546 (Page:530).
8. InterScience | Dextroamphetamine increases phosphoinositol cycle activity in volunteers: an MRS study
9. http://en.wikipedia.org/wiki/Adderall
10. Heck, J. et.al, 'A Conspicuous Connection: Structure Defines Function for the Phosphatidylinositol-Phosphate Kinase Family', Critical Reviews in Biochemistry and Molecular Biology, 42:1, 15 – 39
11. FDA (2007). Medication guide adderall xr. pp. 1–14. Archived from the original on 30 January 2011.