Historical Geology/Chemistry for geologists

In this article we shall take a look at some of the chemistry necessary to follow this or any other introductory textbook on geology.
Chemistry is of course a large and complicated subject which can hardly be addressed in an article such as this one. I have omitted all inessential details and probably several that a chemist would think are in fact essential; I have recklessly sacrificed accuracy to simplicity in a way that would be quite unacceptable in the main body of this textbook. To get a full and accurate picture of chemistry, the reader will have to turn to a book devoted to chemistry; the objective of this appendix is to make sure that when chemical terms, notation, and concepts are introduced in this text, the reader will not find it written in a completely foreign language. More than that I have not attempted.
Some other less basic information on chemistry is introduced within the individual articles in this textbook as and when it becomes relevant.
Structure of an atom[edit | edit source]
An atom consists of protons, neutrons and electrons. Protons have an electrical charge of +1, neutrons have no charge, and electrons have a charge of -1. The number of protons in an atom is equal to the number of electrons, so the atom as a whole has no charge. The number of protons in an atom is called its atomic number, and atoms are classified into elements according to their atomic numbers: for example the element sodium consists of all atoms having an atomic number of 11.
The protons and neutrons in an atom sit together at the center of the atom in a cluster called the nucleus. The electrons form a cloud surrounding the nucleus. A proton or a neutron has a mass about 1836 times that of an electron. This means that the mass of an atom will be determined almost entirely by the sum of the protons and neutrons it contains; this is called the atomic weight of the atom.
The electrons don't just buzz randomly around the nucleus like flies around a jam jar. Rather, they arrange themselves around the nucleus according to the laws of quantum electrodynamics. The reader will find these discussed in chemistry textbooks; to drag quantum theory into an introductory textbook on geology would be a step too far in reductionist explanation.
Instead, we shall have to content ourselves with the sort of explanation that you would have been offered before the discovery of quantum mechanics, and which is still offered to middle-schoolers; this should be quite sufficient for our purposes.
According to this scheme, the electrons are arranged in electron shells around the nucleus. Two electrons can fit into the innermost shell, eight in the second, eight in the third, eighteen in the fourth, and so on in a pattern which we shall have to leave unexplained since we're not going to talk about quantum theory. The electrons arrange themselves in these shells from the inside working out, so that the first shell must have its full quota of electrons before any will go into the second; the second shell must be full before any electron go into the third shell; and so on.
Chemical bonds and molecules[edit | edit source]
Metaphorically, we might say that an atom is "happiest" when out of all its electron shells that contain electrons at all, the outermost one contains as many electrons as will fit in the shell.
This means that atoms can come to a mutually beneficial relationship in which they share the electrons in their outer shell; this is known as a covalent bond. For example, consider the diagram to the right, showing such a bond between two oxygen atoms, with the nucleus represented in red and the electrons in green. Each atom on its own has six electrons in its outer shell, but by sharing electrons they have eight electrons apiece in their outer shells. The two atoms bonded together in this way form a molecule: in this case the molecule O2 (this chemical notation will be explained below).
Alternatively, atoms can form ionic bonds in which instead of sharing electrons, one atom outright gives one or more of its electrons to another. So, for example, a sodium atom, with eleven electrons, would prefer to lose an electron, leaving it with eight electrons in what would then be its outermost shell, whereas a chlorine atom, with 17 electrons, would prefer to add an electron to its outermost shell, filling it up. If the sodium atom gives its unwanted electron to the chlorine atom, both are satisfied.
This leaves the sodium atom with a charge of +1, because it has one more proton than it has electrons, and the chlorine atom has one more electron than it has protons, giving it a charge of -1.
Chemical notation[edit | edit source]
Abbreviations for elements
Each element is represented either by a single capital letter (e.g. H for hydrogen, K for potassium, W for tungsten) or by a capital letter followed by a lower case letter (e.g. Cl for chlorine, Hg for mercury, Na for sodium).
Chemical formulas
We can use these abbreviations to describe the composition of molecules: for example the combination of sodium and chlorine described above can be written as NaCl. (By convention Na is written first because the sodium has a positive charge.)
When there is more than one atom of a given element in a molecule, this is written by means of a number written in subscript to the right of the symbol for the element. So, for example, a molecule of water contains two atoms of hydrogen (H) and one of oxygen (O), and is written H2O; a molecule of methane has one atom of carbon (C) and four of hydrogen (H), and is written CH4.
Chemical reactions
We can write chemical reactions, in which molecules form, break apart, and recombine, by using chemical formulas and the addition of a couple more symbols. For example, we can write:
- Na + Cl → NaCl
to indicate that sodium and chlorine, to the left of the arrow, will combine to produce NaCl, on the right.
Note that the same elements, in the same quantities, appear on both sides of the arrow; in a chemical reaction no atoms are created or destroyed, only the nature of their relationship with one another are changed.
Another convention used in describing chemical reactions is to use full-sized numbers in front of the name of an atom or molecule to indicate how many of them enter into the reaction. So for example we can write:
- 2HCl + Mg → MgCl2 + H2
Here the full-sized 2 indicates that there are two HCl molecules on the left-hand side. Note that the 2 refers to the entire formula HCl to the right of it, not just to the H immediately to the right of it.
Why don't we just write H2Cl2 instead of 2HCl? Because H2Cl2 is not a molecule; rather we have two molecules of HCl, and the notation reflects that. On the other hand we write H2 on the right hand side rather than 2H because H2 is a molecule, and it would be inaccurate to write 2H.
Ions
If an atom or a collection of atoms has lost an electron, as in the formation of ionic bonds, then it will have a positive charge, and is said to be a positively charged ion. We can represent this fact by a plus sign written as a superscript to the right. If instead it had gained an electron it would have a negative charge and would be described as a negative ion; we represent this by a minus sign written in the same place, above and to the right of the symbol for the element.
So for example we could if we wished write the combination of sodium and chlorine as Na+Cl-.
If an ion has lost or gained more than one electron, then we can represent this with a number written before the sign of the charge: so, for example, we could write Ca2+ to indicate an atom of calcium which has lost both of the electrons in its outer shell, giving it a charge of +2.
It is not compulsory to indicate the ionization. For example, no chemist would actually bother to write Na+Cl-, because any chemist would know that this is how sodium and chlorine must be ionized in order to form a molecule. However it is useful to have this notation as an option.
Chemical names
There are also conventional ways of giving molecules reasonably pronounceable names: so for example a chemist would look at the formula CaSO4 and know to call it calcium sulfate. For our purposes it is not necessary to explain the details of this system: in the articles in this textbook I have simply presented the chemical name alongside the chemical formula.
The periodic table[edit | edit source]
If two elements have a similar situation in their outermost shells, then chemically one will behave much the same as the other. For example, sodium and potassium both have one electron in their outermost shells. This means that just as one atom of sodium can combine with one atom of chlorine to make NaCl, so can one atom of potassium combine with one atom of chlorine to make KCl.
Similarly fluorine (F) is in the same situation as chlorine: each of them is short one electron in their outer shells. This means that fluorine will combine with sodium or potassium just the same as chlorine will, forming NaF or KF, respectively.
This information about elements can be summarized by arranging the elements in the periodic table, shown below.
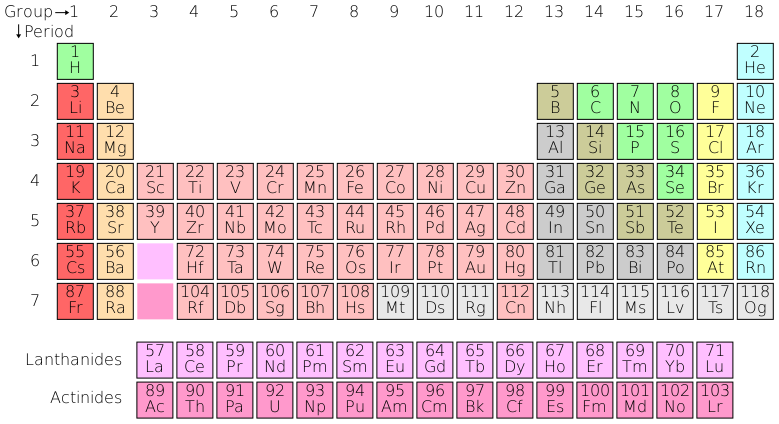
Elements in the same column are said to belong to the same group, and have similar situations in their outer electron shells. For example, the elements in the far left-hand column all have a single electron in their outer shells, which they are anxious to give away; meanwhile those in the far right-hand column all have full outer electron shells, and so will not form chemical bonds, since they are perfectly happy the way they are.
The facts summarized in the periodic table are important to our understanding of various aspects of geology; for example in our discussion of paleoclimatology we shall make use of the fact that both magnesium and strontium can substitute for calcium; and in discussing U-Pb radiometric dating, it is important to know that uranium can substitute for zirconium in the formula ZrSiO4 but that lead cannot.
The reader will note by looking at the periodic table that in that last example uranium (U) does not exactly lie in the same column as zirconium (Zr). However, for reasons which can hardly be explained without reference to quantum theory, there is more flexibility for elements colored pink in the table to substitute for one another: so uranium can stand in for zirconium, but (for example) calcium could not substitute for sodium despite the fact that they're closer together in the table than uranium is to zirconium.
Solvents and solutes[edit | edit source]
A substance (a solute) is said to be dissolved in another substance (a solvent) if it is mixed with it in such a way as to acquire the phase of the solvent (i.e. whether it is solid, liquid or gas, and its crystal structure if it is a solid).
Compare, for example, what happens when we add to water a substance which is not soluble in water, such as sand, and a substance which is soluble in water, such as table salt (NaCl). The sand remains a solid; all we have done is mix a solid with a liquid. What's more, it will not be evenly distributed in the water, but will sink to the bottom, being more dense than water; if it was less dense, it would float to the top.
By contrast, salt does dissolve in water, so what we get is not a solid (salt) in a liquid (water), but rather is simply a liquid, salty water; and the salt rapidly becomes evenly distributed throughout the water rather than sinking to the bottom.
Salt (NaCl) is an example of a molecule held together by an ionic bond, as we have noted above. Dissolved in water, however, the bond is also dissolved, as the positively charged sodium is attracted to the negative charge on the oxygen atoms in the water molecules, and the chlorine atoms are attracted to the positive charge on the hydrogen atoms. Instead of describing the dissolved salt as NaCl, it is more accurate to describe it as Na+ + Cl-; this is one occasion on which the notation for ions is useful.
Isotopes[edit | edit source]
Because the chemical behavior of atoms is determined by the interaction between their electrons, the key figure that determines the chemical properties of an atom is its atomic number; this tells us how many electrons it has, and the number of electrons determines their arrangement in the electron shells. This is why to chemists the right way to classify atoms is into elements according to their atomic number.
However, we can make a finer distinction between atoms: two atoms which have the same number of electrons (and therefore protons) can have different numbers of neutrons and therefore different atomic weights. So for example we can distinguish between uranium-235 (uranium with 143 neutrons, 92 protons, 92 electrons, and an atomic weight of 235) and uranium-238 (uranium with 146 neutrons, 92 protons, 92 electrons, and an atomic weight of 238).
The notation used for isotopes is to write the atomic weight as a superscript to the left of the symbol for the element: so for example uranium-235 would be written as 235U. (Some texts also write the atomic number as a subscript to the left of the atomic symbol; in this textbook I have not followed this convention.)
Because two different isotopes of the same element still are the same element, with the same arrangement of electrons, they have the same chemical properties. So just as one element can substitute for another if they are in the same group, it is even more the case that two isotopes of the same element will be chemically interchangeable. This fact is particularly useful to geologists, as many forms of radiometric dating would not be possible without it.
At this point it would be usual to explain nuclear decay and radioactivity; however, this textbook contains an article exclusively devoted to the subject in the chapter on absolute dating.