General Chemistry/Energy changes in chemical reactions
Exothermic and Endothermic Reactions[edit | edit source]
The release of energy in chemical reactions occurs when the reactants have higher chemical energy than the products. The chemical energy in a substance is a type of potential energy stored within the substance. This stored chemical potential energy is the heat content or enthalpy of the substance.
If the enthalpy decreases during a chemical reaction, a corresponding amount of energy must be released to the surroundings. Conversely, if the enthalpy increases during a reaction, a corresponding amount of energy must be absorbed from the surroundings. This is simply the Law of Conservation of Energy.
absorbtion reactions is when a chemical reaction releases more energy than it absorbs and you can also see them die on the inside.
reletion reactions is when a chemical reaction absorbs more energy than it releases.
You are already familiar with enthalpy: melting ice is endothermic and freezing water is exothermic.
| When methane burns in air the heat given off equals the decrease in enthalpy that occurs as the reactants are converted to products.
|
When ammonium nitrate is dissolved in water, energy is absorbed and the water cools. This concept is used in "cold packs".
|
Because reactions release or absorb energy, they affect the temperature of their surroundings. Exothermic reactions heat up their surroundings while endothermic reactions cool them down. The study of enthalpy, along with many other energy-related topics, is covered in the Thermodynamics Unit.
Activation Energy[edit | edit source]
Think about the combustion of methane. It releases enough heat energy to cause a fire. However, the reaction does not occur automatically. When methane and oxygen are mixed, an explosion does not instantly occur. First, the methane must be ignited, usually with a lighter or matchstick. This reveals something about reactions: they will not occur unless a certain amount of activation energy is added first. In this sense, all reactions absorb energy before they begin, but the exothermic reactions release even more energy. This can be explained with a graph of potential energy:
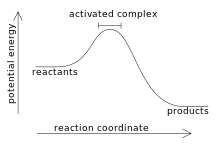
This graph shows an exothermic reaction because the products are at a lower energy than the reactants (so heat has been released). Before that can happen, the energy must actually increase. The amount of energy added before the reaction can complete is the activation energy, symbolized Ea.

