Chemical Sciences: A Manual for CSIR-UGC National Eligibility Test for Lectureship and JRF/Vertico SMI
| This page was imported and needs to be de-wikified. Books should use wikilinks rather sparsely, and only to reference technical or esoteric terms that are critical to understanding the content. Most if not all wikilinks should simply be removed. Please remove {{dewikify}} after the page is dewikified. |
Vertico-SMI is currently the fastest light microscope for the 3D analysis of complete cells in the nanometer range.

Configuration[edit | edit source]
SMI stands for a special type of laser optical illumination (Spatially Modulated Illumination) and Vertico reflects the vertical arrangement of the microscope axis which renders possible the analysis of fixed cells but also of living cells with an optical resolution below 10 nanometers (1 nanometer = 1 nm = 1 × 10−9 m).
Basic Facts[edit | edit source]
Vertico-SMI was developed by Christoph Cremer, Professor of Applied Optics and Information Processing at Heidelberg University and is based on the combination of light optical techniques of localization microscopy (SPDM, Spectral Precision Distance Microscopy) and structured illumination (SMI, Spatially Modulated Illumination). A particularity of this technology compared with focusing techniques such as 4Pi microscopy, is the wide field exposures which allow entire cells to be depicted at the nano scale. Such a 3D exposure of a whole cell with a typical object size of 20 µm × 20 µm require only 2 minutes. Wide field exposures signify that the entire object is illuminated and detected simultaneously.
Spatially Modulated Illumination (SMI)[edit | edit source]
SMI microscopy is a light optical process of the so-called Point Spread Function-Engineering. These are processes which modify the Point Spread Function (PSF) of a microscope in a suitable manner to either increase the optical resolution, to maximize the precision of distance measurements of fluorescent objects that are small relative to the wavelength of the illuminating light, or to extract other structural parameters in the nanometer range.
The SMI microscope currently being developed at the Kirchhoff Institute for Physics at Heidelberg University achieves this in the following manner: The illumination intensity within the object range is not uniform, unlike conventional wide field fluorescence microscopes, but is spatially modulated in a precise manner by the use of two opposing interfering laser beams along the axis. The principle of the spatially-modulated wave field was developed in 1993 by Bailey et al. The SMI microscopy approach used in the Heidelberg application moves the object in high-precision steps through the wave field, or the wave field itself is moved relative to the object by phase shift. This results in an improved axial size and distance resolution.
Spectral Precision Distance Microscopy (SPDM)[edit | edit source]
SPDM (Spectral Precision Distance Microscopy) is a light optical process of fluorescence microscopy which allows position, distance and angle measurements on "optically isolated" particles (e.g. molecules) well below the theoretical limit of resolution for light microscopy. "Optically isolated" means that at a given point in time, only a single particle/molecule within a region of a size determined by conventional optical resolution (typically approx. 200-250 nm diameter) is being registered. This is possible when molecules within such a region all carry different spectral markers (e.g. different colors or other usable differences in the light emission of different particles).
The structural resolution achievable using SPDM can be expressed in terms of the smallest measurable distance between two in their spatial position determined punctiform particle of different spectral characteristics ("topological resolution“). Modeling has shown that under suitable conditions regarding the precision of localization, particle density etc., the "topological resolution" corresponds to a "space frequency" which in terms of the classical definition is equivalent to a much improved optical resolution.
SPDM is a localization microscopy which achieves an effective optical resolution several times better than the conventional optical resolution (approx. 200-250 nm), represented by the half-width of the main maximum of the effective point image function. By applying suitable laser optical precision processes, position and distances significantly smaller than the half-width of the point spread function (conventionally 200-250 nm) can be measured with nanometer accuracy between targets with different spectral signatures. An important area of application is genome research (study of the functional organization of the genome). Another important area of use is research into the structure of membranes.
Resolution[edit | edit source]
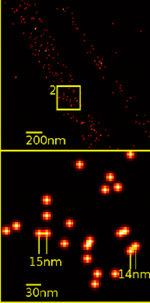
The effective optical resolution of this optical nanoscope has reached the vicinity of 10 nm in 2D and 40 nm in 3D and is therefore substantially better than the physical limit of 200 nm predicted by Abbe‘s law. Abbe postulated in 1873 the theoretical limit of resolution of optical microscopy [1].
The 3D images using Vertico-SMI are made possible by the combination of SMI and SPDM, whereby first the SMI and then the SPDM process is applied. The SMI process determines the center of particles and their spread in the direction of the microscope axis. While the center of particles/molecules can be determined with a 1-2 nm precision, the spread around this point can be determined down to an axial diameter of approx. 30-40 nm.
Subsequently, the lateral position of the individual particles/molecules is determined using SPDM, achieving a precision of a few nanometers. At present, SPDM achieves 16 frames/sec with an effective resolution of 10 nm in 2D (object plane); approximately 2000 such frames are combined with SMI data (currently ca. 10 sec acquisition time) to achieve a three dimensional image of highest resolution (effective optical 3D resolution ca. 40-50 nm). With a faster camera, one can expect even higher rates (up to several hundred frames/sec, currently under development). Using suitable dyes, even higher effective optical 3D resolutions should be possible[2][3]
GFP Superresolution "Blinking Dyes" instead of photo-switchable molecules[edit | edit source]
Only in the past two years have molecules been used in nanoscopic studies which emit the same spectral light frequency (but with different spectral signatures based on the flashing characteristics) but which can be switched on and off by means of light as is necessary for spectral precision distance microscopy. By combining many thousands of images of the same cell, it was possible using laser optical precision measurements to record localization images with significantly improved optical resolution. The application of these novel nanoscopy processes appeared until recently very difficult because it was assumed that only specially manufactured molecules could be switched on and off in a suitable manner by using light. Professor Christoph Cremer’s team discovered in 2008 that this was also possible for many standard fluorescent dye like GFP, Alexa dyes and fluorescein molecules, provided certain photo-physical conditions are present.[4]
The GFP gene has been introduced and expressed in many procaryotic and eucaryotic cells and the Nobel Prize in Chemistry 2008 was awarded to Martin Chalfie, Osamu Shimomura, and Roger Y. Tsien for their discovery and development of the green fluorescent protein. The finding that these standard fluorescent molecules can be used extends the applicability of the SPMD method to numerous research fields in biophysics, cell biology and medicine.
Literature[edit | edit source]
- ↑ Reymann J, Baddeley D, Gunkel M, Lemmer P, Stadter W, Jegou T, Rippe K, Cremer C, Birk U.: High-precision structural analysis of subnuclear complexes in fixed and live cells via spatially modulated illumination (SMI) microscopy. in: Chromosome Research 2008;16(3):367–82. PMID 18461478 Template:DOI
- ↑ Lemmer P, Gunkel M, Baddeley D, Kaufmann R, Urich A, Weiland Y, Reymann J, Müller P, Hausmann M, Cremer C: SPDM – Light Microscopy with Single Molecule Resolution at the Nanoscale. In: Applied Physics B 2008; 93: 1–12
- ↑ Baddeley D, Batram C, Weiland Y, Cremer C, Birk UJ.: Nanostructure analysis using Spatially Modulated Illumination microscopy. In: Nature Protocols 2007; 2: 2640–2646
- ↑ Manuel Gunkel, Fabian Erdel, Karsten Rippe, Paul Lemmer, Rainer Kaufmann, Christoph Hörmann, Roman Amberger and Christoph Cremer: Dual color localization microscopy of cellular nanostructures. In: Biotechnology Journal, 2009, 4, 927-938. ISSN 1860-6768